AP Syllabus focus: ‘Photoelectron spectroscopy measures electron energies in shells; each peak position relates to the energy required to remove an electron from a subshell.’
Photoelectron spectroscopy (PES) provides experimental evidence for quantized electron energies. By ejecting electrons with high-energy light and measuring their kinetic energies, PES reveals how tightly electrons are held in different atomic subshells.
What PES Measures
PES is designed to probe electron energies in shells/subshells by converting an electron’s attraction to the nucleus into a measurable energy quantity.
Photoelectron spectroscopy (PES): An experimental technique that uses photons to eject electrons from atoms (or ions) and measures the electrons’ kinetic energies to determine the energy required to remove them.
In AP Chemistry, interpret PES conceptually as an energy measurement: electrons in different subshells require different energies to remove, so PES distinguishes them.
Key measurable quantity: energy to remove an electron
Removing an electron requires overcoming electrostatic attraction between the negatively charged electron and the positively charged nucleus; this required energy depends on which subshell the electron occupies.
Binding energy: The energy required to remove an electron from a specific subshell of an atom (or ion), typically reported in electronvolts (eV).
Binding energy is the central “electron energy” PES reports; larger binding energy means the electron is held more strongly.
How PES Works (Conceptual Procedure)
A PES experiment connects light energy to measured electron kinetic energy.
Step-by-step idea
A beam of monochromatic photons (single frequency) with known energy strikes gaseous atoms.
If a photon has enough energy, it ejects an electron, producing a photoelectron.
An electron energy analyzer measures the kinetic energy of the ejected electrons.
Using energy conservation, the instrument determines the binding energy for electrons originating from each subshell.
The results are displayed as a spectrum with peaks, where each peak position corresponds to a particular electron-removal energy (subshell).
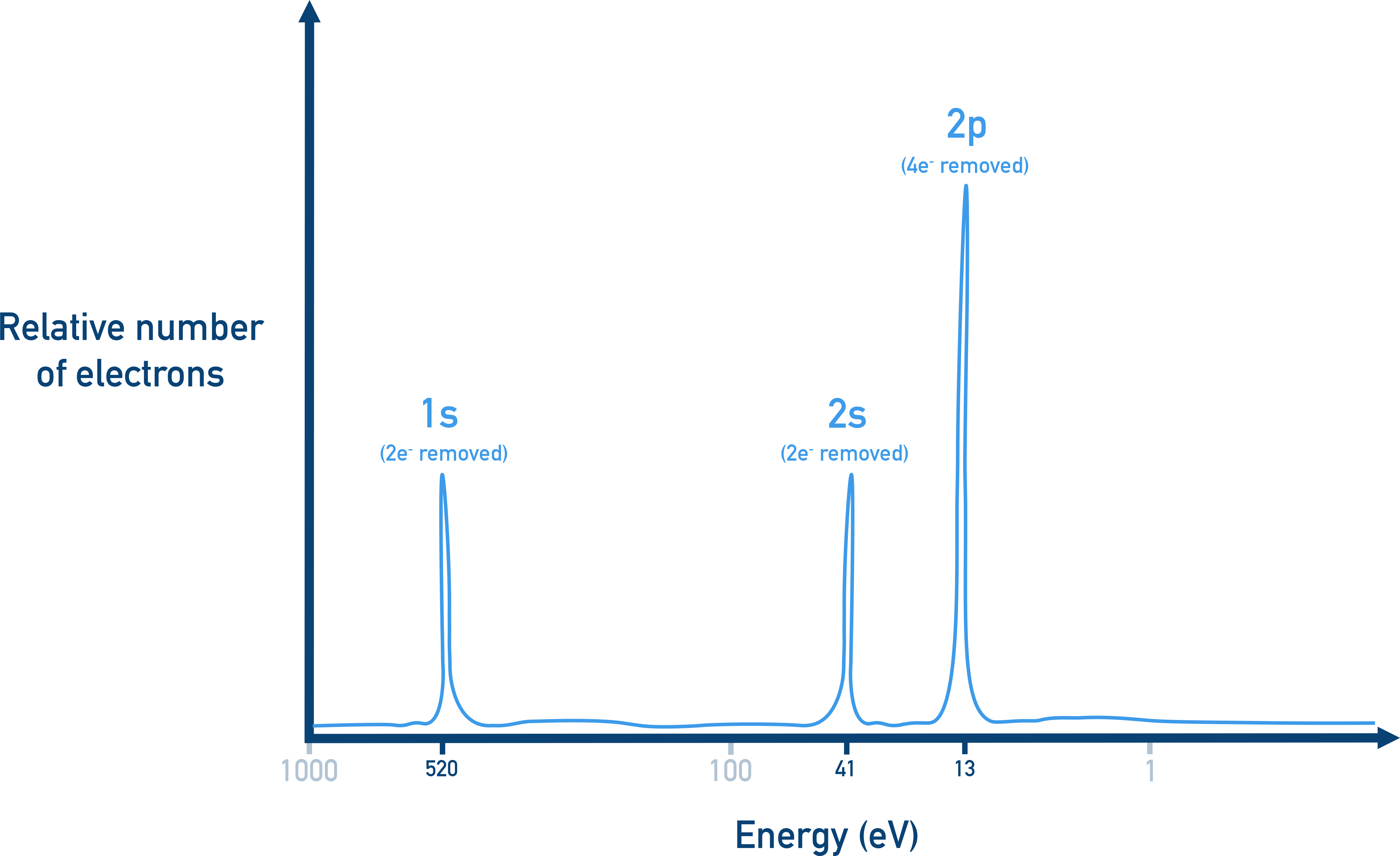
Example photoelectron spectrum with labeled 1s, 2s, and 2p peaks. The x-axis shows electron binding energy (in eV), so peak position indicates how tightly electrons in each subshell are held. Peak height reflects the relative number of electrons detected/removed from that subshell. Source
A single subshell produces a characteristic binding energy, so PES can separate electron groups by subshell energy.
Energy Relationship Behind PES
PES relies on conservation of energy: photon energy becomes electron kinetic energy plus the energy needed to detach the electron.
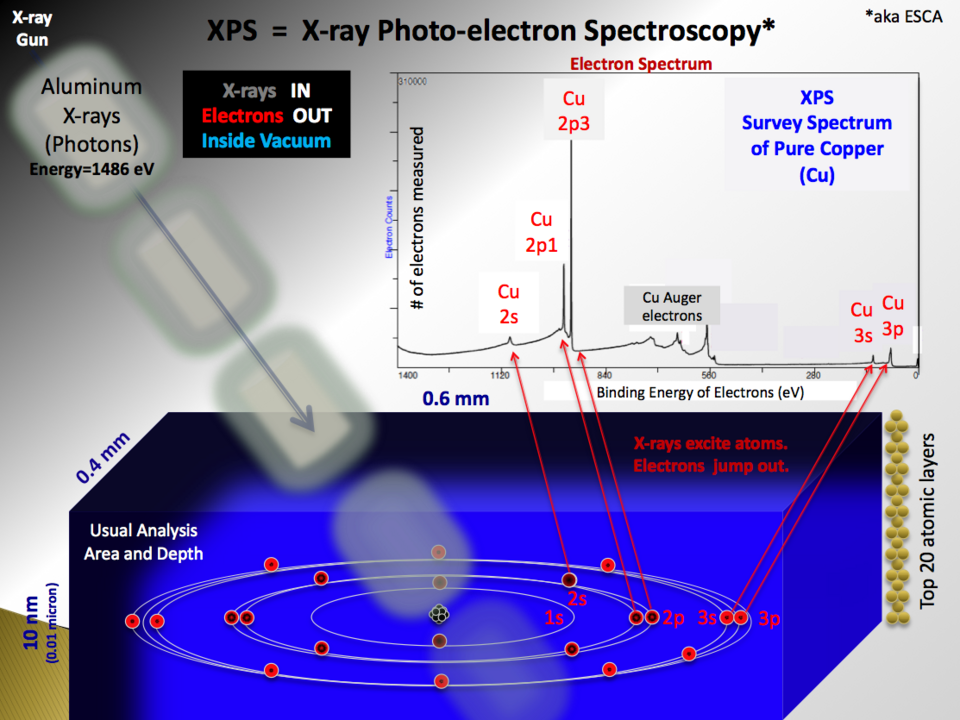
Schematic of photoelectron spectroscopy illustrating electrons being ejected by incoming photons and then analyzed to produce a spectrum. It emphasizes the energy accounting behind the method: known photon energy and measured electron kinetic energy allow inference of binding energy for each peak. The resulting plot shows intensity versus binding energy, linking subshell-specific electrons to distinct features in the spectrum. Source
= photon energy (J or eV)
= Planck’s constant ( J·s)
= photon frequency (s)
= binding energy (eV or J)
= kinetic energy of the ejected electron (eV or J)
Because is known and is measured, can be found for electrons coming from different subshells, matching the syllabus idea that peak position relates to the energy required to remove an electron from a subshell.
What “Peak Position” Means in PES (Only the Essential Interpretation)
A PES spectrum plots a signal versus binding energy (or an equivalent energy axis). For this subsubtopic, the key point is the meaning of where peaks appear:
Peaks at higher binding energy correspond to electrons that require more energy to remove (more tightly held).
Peaks at lower binding energy correspond to electrons that are easier to remove.
This provides direct experimental access to electron energy differences among shells and subshells without assuming electron energies are continuous.
Why Electron Energies Differ Among Subshells (PES Context)
Within an atom, electrons occupy different regions and experience different nuclear attraction:
Inner-shell electrons are closer on average and experience stronger attraction, so they have larger binding energies.
Outer-shell electrons are farther and more shielded, so they have smaller binding energies.
PES therefore supports the shell/subshell model by showing distinct energies rather than a smear of values.
FAQ
Gas-phase atoms minimise interactions between particles that would otherwise shift measured electron energies.
In solids or liquids, neighbouring atoms and bonding can broaden or shift features, making it harder to assign a measured energy to a specific atomic subshell.
The photon must supply at least the binding energy for that electron.
If $E_{\text{photon}}$ is too small, no photoelectron is produced from that subshell, so that subshell will not appear in the spectrum for that photon energy.
Electron energies at the atomic scale are conveniently sized in eV.
Conversion: $1\ \text{eV} = 1.602\times10^{-19}\ \text{J}$, which makes typical binding energies (tens to thousands of eV) easier to display and compare.
Common approaches include:
Electrostatic energy analysers that only transmit electrons of selected kinetic energy.
Time-of-flight methods that infer kinetic energy from how long electrons take to reach a detector.
Both methods convert electron motion into an experimentally measurable signal.
Peak width can be affected by instrumental resolution and intrinsic effects such as:
Slight energy variations from electron–electron interactions,
Finite lifetimes of electron vacancies (leading to energy uncertainty),
Thermal motion and detector broadening.
Sharper peaks indicate better energy resolution for distinguishing nearby subshell energies.
Practice Questions
(1–3 marks) In a PES experiment, photons of energy eject electrons from a gaseous element. Explain how measuring the kinetic energy of the ejected electrons allows the binding energy to be determined.
States that photon energy is known and kinetic energy is measured (1).
Uses conservation of energy to relate them: (1).
Explains that binding energy is the energy required to remove the electron from a subshell / peak position corresponds to this removal energy (1).
(4–6 marks) A student is told that a PES spectrum contains several peaks at different binding energies. Using PES principles, explain what information the positions of these peaks provide about electron energies in the atom, and why electrons from different subshells give different peak positions.
Identifies that each peak position corresponds to a particular binding energy (energy needed to remove an electron) (1).
Links different peaks to electrons in different shells/subshells with different energies (1).
States that higher binding energy means electrons are more strongly attracted to the nucleus / harder to remove (1).
States that lower binding energy means electrons are less strongly held / easier to remove (1).
Explains subshell differences in terms of distance from nucleus and/or shielding affecting nuclear attraction (1).
Makes clear that PES provides evidence for quantised electron energy levels (1).

