AP Syllabus focus: ‘You do not need to assign quantum numbers to electrons, and you will not be tested on writing electron configurations for Aufbau exceptions.’
These notes clarify the boundaries of what you are and are not responsible for on the AP Chemistry exam regarding electron configurations, helping you focus study time on tested skills and avoid unnecessary memorisation.
What the AP Exam Explicitly Excludes
AP Chemistry assessments sometimes reference atomic structure language, but they intentionally avoid two advanced tasks that can become overly rules-based rather than concept-based:
Assigning quantum numbers to specific electrons
Writing electron configurations that require Aufbau exceptions (configuration “anomalies”)
Exclusion 1: Assigning Quantum Numbers
You may see the terms “quantum numbers” in some textbooks, but AP Chemistry does not require you to generate them for any electron in an atom or ion.
Quantum numbers: a set of labels (commonly , , , ) used in quantum mechanics to specify an electron’s energy level and orbital properties.
Instead of quantum-number assignments, AP Chemistry expects you to reason with the shell/subshell/orbital model at a qualitative level (for example, recognising that electrons occupy different energy levels and that subshells differ in energy).
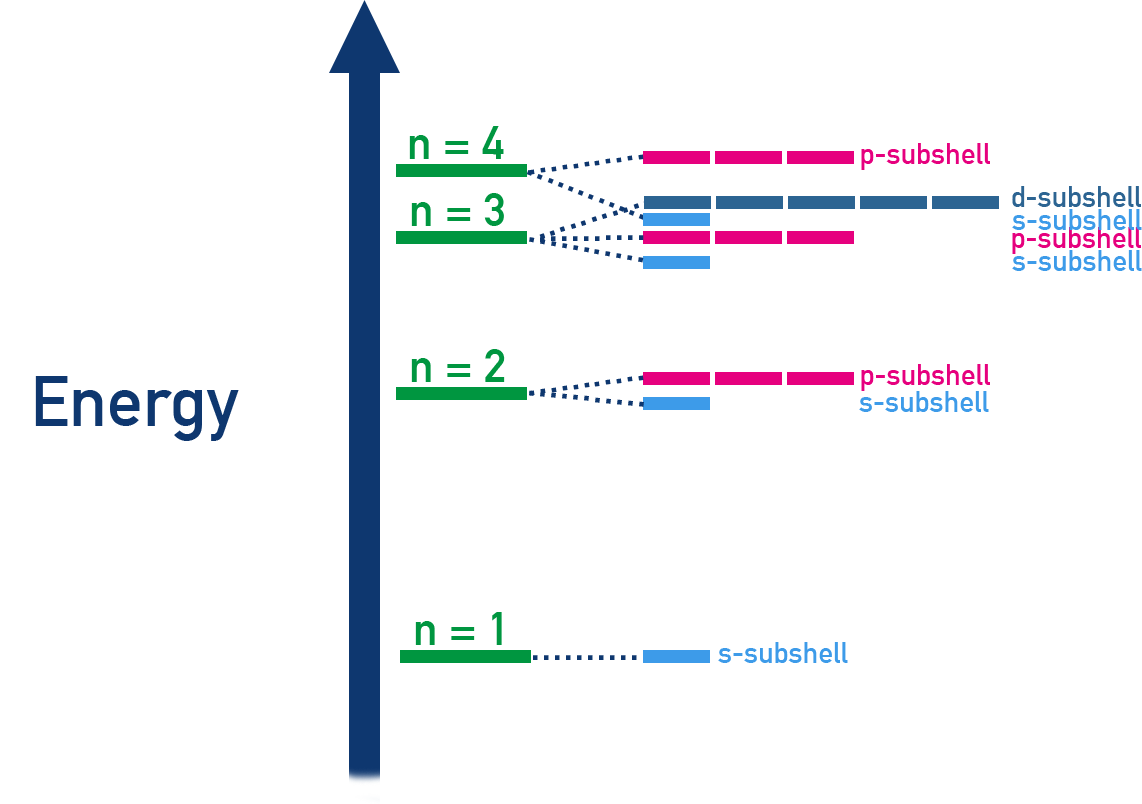
Energy-level diagram showing the relative energies of common subshells (e.g., , , , , , , , ). The vertical axis indicates increasing energy, and the layout highlights that subshell energies can overlap (notably, is lower in energy than for neutral atoms). Source
What this means for you:

Schematic linking shells (energy levels labeled by ) to subshell types (, , ) and their typical electron capacities. The diagram helps interpret electron-configuration language conceptually—focusing on levels and subshells—without needing to compute specific quantum-number sets. Source
Do not memorise the full quantum-number rules or allowed-value sets.
Do not practise “find , , , for the last electron” style problems for AP purposes.
If a question mentions orbitals, focus on broad ideas (relative energy, filling order), not quantum-number bookkeeping.
Exclusion 2: Aufbau Exceptions (“Anomalous” Configurations)
The AP exam expects you to write ground-state electron configurations using the standard Aufbau filling pattern presented in most introductory chemistry courses. You will not be tested on the small set of real-world cases where observed configurations deviate from that simple pattern.
Aufbau exception: a species whose experimentally observed ground-state electron configuration differs from the configuration predicted by the simple Aufbau filling order (often involving and subshell rearrangements).
Between these two excluded topics, AP is signalling that you should prioritise consistent, periodic-table-based filling patterns over exceptions that require special memorisation.
Practical Implications for Studying and Test-Taking
What you should do on the AP Exam
Use the expected Aufbau order (the one you learn from the periodic table block structure).
When a multiple-choice option set includes an “exception” configuration, the test will not require you to know it as an exception; the correct answer will be obtainable without that specialised knowledge.
If you are provided with a configuration in the question stem (for instance, as given information), you may be asked to interpret it conceptually, but not to “fix” it using exception rules.
Common ways these exclusions show up
Distractor answers may include quantum numbers or anomalous configurations; treat them as outside the intended skill set unless explicitly provided as information.
Classroom resources sometimes overemphasise exceptions (e.g., select transition metals). For AP, you should recognise that this is enrichment, not required mastery.
What not to spend time memorising
Lists of “special-case” elements with irregular configurations
Rule sets for promoting electrons to create half-filled or filled subshell patterns
Any procedure that assigns four quantum numbers to an electron
FAQ
Yes, terminology may appear for context, but you will not be required to generate sets of four quantum numbers.
If such symbols appear, treat them as background labels rather than something to compute.
Using an exception will typically not be necessary to earn marks.
If you choose to write an exception, ensure it does not conflict with the question’s instructions or any provided information.
No. The mechanistic or stability-based justification is beyond what AP expects to assess directly for this exclusion.
You can view it as enrichment rather than examinable reasoning.
Occasionally, a prompt might provide information that implies a configuration. In that case, use the provided information.
You are not expected to independently recall which elements deviate from Aufbau.
The exclusion is about being tested on exception-writing and quantum-number assignment.
If an ion configuration question is asked, it will be solvable with the standard approach and any information supplied in the prompt.
Practice Questions
State two topics related to electron configuration that are explicitly excluded from the AP Chemistry exam.
Mentions that students do not need to assign quantum numbers to electrons (1)
Mentions that students will not be tested on writing electron configurations for Aufbau exceptions (1)
A student revises by memorising quantum-number rules and a list of Aufbau exceptions (e.g., cases where an electron shifts between and subshells).
(a) Explain why this revision focus is not aligned with AP Chemistry expectations. (2 marks)
(b) Describe an exam-appropriate approach to writing electron configurations given this exclusion. (3 marks)
(a)
Quantum-number assignment is excluded (1)
Aufbau exceptions are excluded / not assessed (1)
(b)
Use the standard Aufbau filling pattern / periodic-table-based order (1)
Do not apply memorised exception shifts between subshells (1)
If unusual information is supplied, interpret it only as given rather than relying on exception recall (1)

