AP Syllabus focus: ‘Elements in the same column of the periodic table tend to form analogous compounds because they have similar valence electron structures.’
Elements in the same periodic-table family often behave alike because their outer electrons follow the same pattern. Recognising these patterns lets you anticipate common formulas, ions formed, and the kinds of substances each family typically produces.
Families (Groups) and Valence Electron Similarity
What “family” implies chemically
Elements are arranged in vertical columns (families) that share similar valence electron arrangements, so they tend to show similar bonding and compound-forming behaviour.
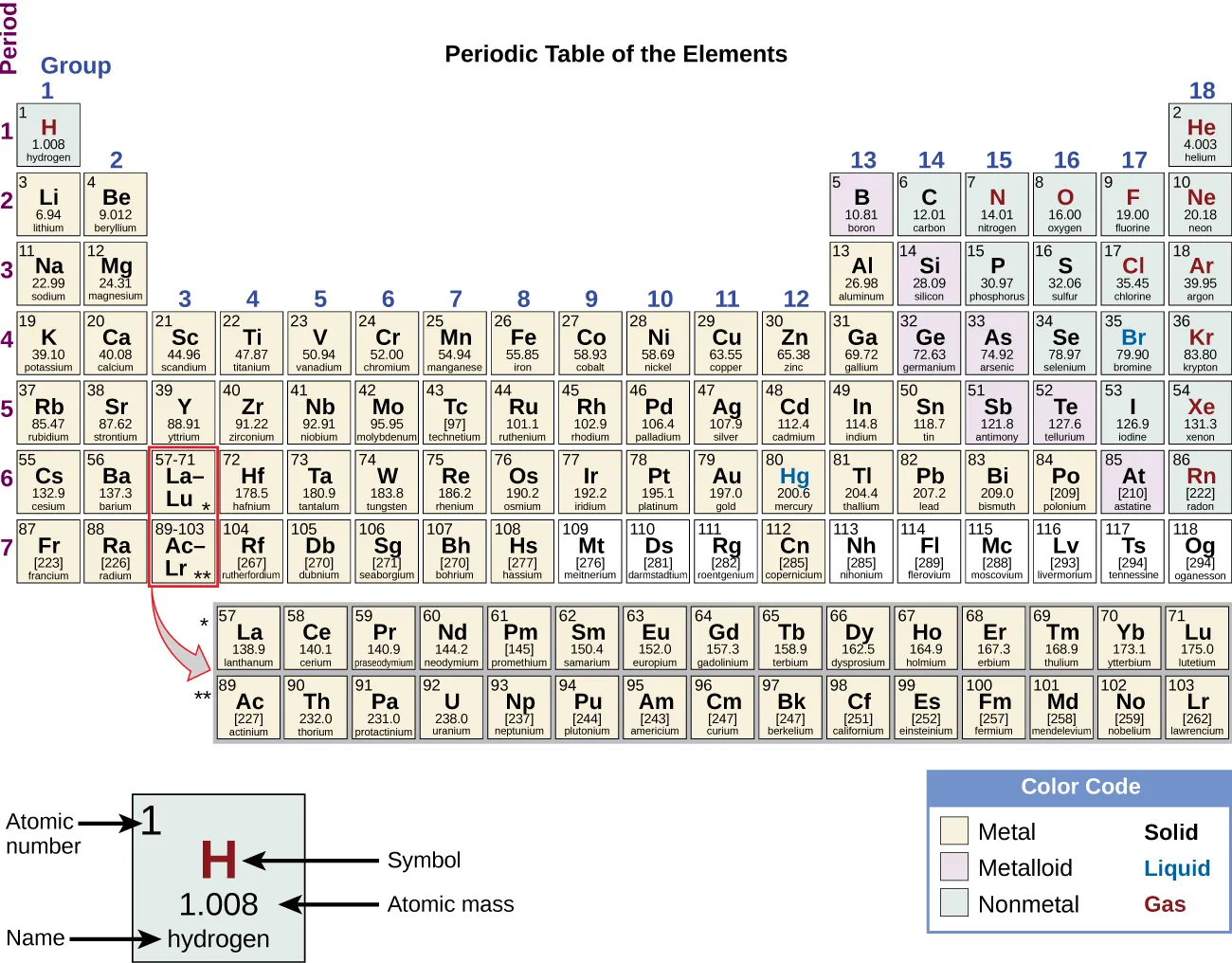
A labeled periodic table showing how elements are organized into groups (vertical columns) and periods (horizontal rows). Reading down a group highlights the “family” concept: elements align in the same column because their outer-shell electron patterns repeat in a way that drives similar bonding and compound-forming behavior. Source
Valence electrons: the outermost electrons of an atom that are most directly involved in chemical bonding and typically control the atom’s reactivity and common compound patterns.
A Bohr-style shell diagram illustrating electrons arranged in discrete energy levels, with the valence electrons located in the outermost shell. The picture-level separation between core and outer electrons helps explain why atoms with the same outer-shell electron count (same group) tend to show similar bonding and compound patterns. Source
Because members of a family have the same number of valence electrons, they often:
form the same types of ions (especially main-group metals and nonmetals)
make compounds with similar element ratios (analogous formulas)
show similar preferences for bonding with particular partners (e.g., oxygen, halogens, hydrogen)
How Similar Valence Structures Lead to Analogous Compounds
Repeating formulas within a group
For many main-group families, the shared valence structure means a repeated “template” of compound stoichiometry across the column.
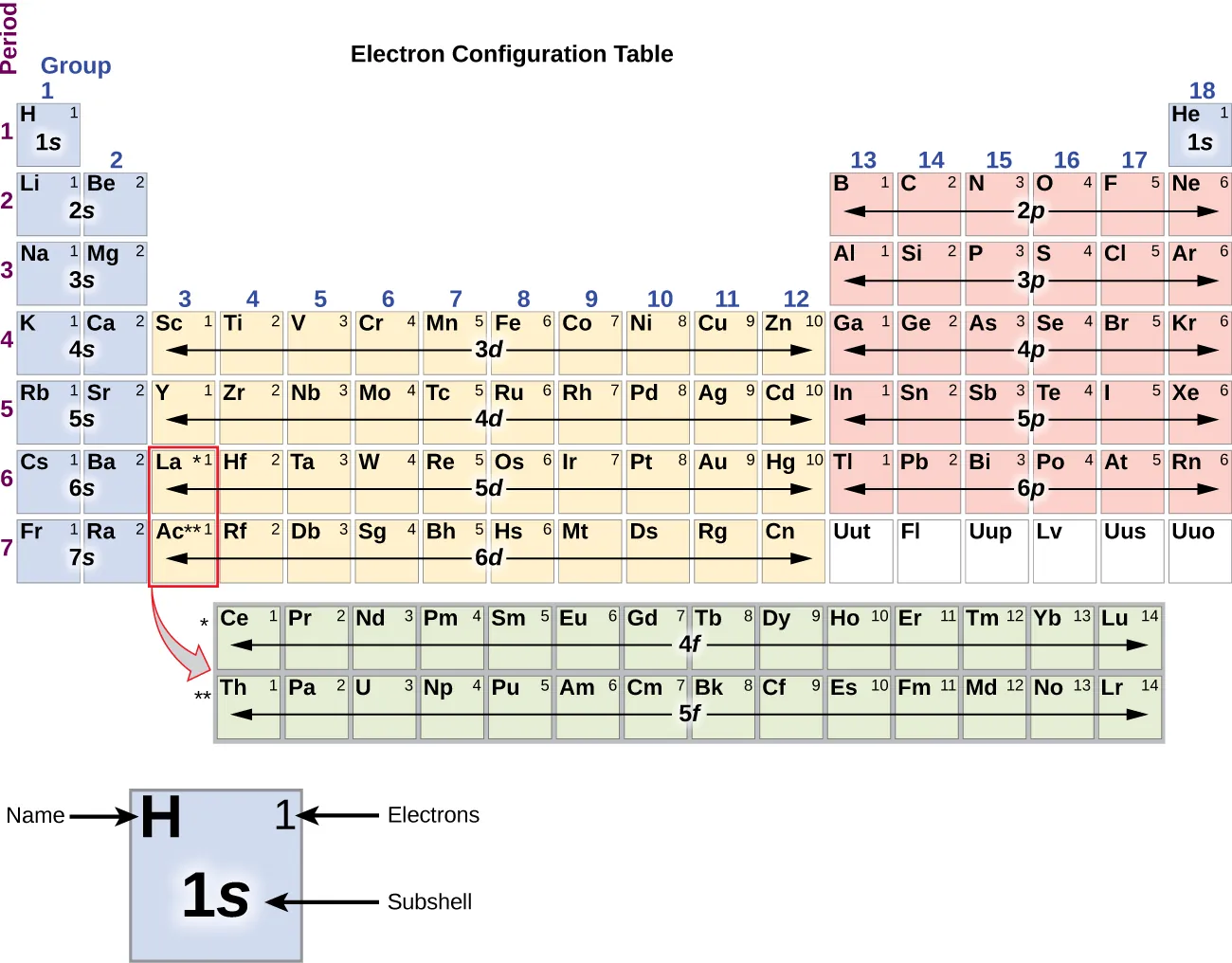
A periodic-table overlay that labels which subshell (s, p, d, f) is being filled across different blocks of elements. This makes the electron-configuration basis of periodic trends explicit, helping you see why main-group elements in the same column share similar outer-electron structures and therefore tend to form analogous compounds. Source
For example, a family may commonly form:
chlorides with the same general atom ratio across the group
oxides with a repeating ratio pattern across the group
hydrides that follow a consistent pattern within that family
These analogous formulas reflect that each element in the family tends to reach a similar stable electron arrangement when bonding, so the number of bonding interactions (or electron transfers) is often comparable.
Similar reaction “types,” not identical reactivity
Families form similar compounds, but not necessarily at the same rate or under the same conditions. Down a group, changes in atomic size and electron shielding can alter how readily compounds form, even when the compound type stays similar. Practically, this means:
the set of common compounds is similar across the family
the ease of forming those compounds can vary among members
Recognisable Family Patterns (High-Utility for AP Chemistry)
Alkali metals (Group 1)
Group 1 elements share a one-valence-electron pattern, so their compounds frequently show parallel formulas and properties:
salts with nonmetals that commonly share similar stoichiometries across the family
basic compounds with oxygen-containing anions that often behave similarly in water
Alkaline earth metals (Group 2)
Group 2 members commonly form analogous ionic compounds with consistent ratios across the family:
related halide and oxide formulas that repeat by element
many compounds that are characteristically ionic and form crystalline solids
Halogens (Group 17)
Halogens share a seven-valence-electron pattern and often form analogous compounds:
salts with metals that are frequently similar in formula type across the group
hydrogen halides that share related molecular structures and many parallel chemical features
Noble gases (Group 18)
Noble gases share filled valence shells, so they are comparatively unreactive and form far fewer compounds. The family similarity here is that:
most members do not commonly form compounds under standard conditions
when compounds do exist (special cases), they tend to be limited to specific circumstances
Using Family Similarity as a Predictive Tool
What you can infer from “same column”
When you know an element’s family, you can often predict:
the kinds of binary compounds it is likely to form (e.g., with oxygen, halogens, hydrogen)
whether its compounds are more likely to be ionic (metals) or molecular (nonmetals)
whether it tends to form a consistent set of analogous compound formulas like its neighbors in the same group
Limits of the idea
Family similarity is strongest for the main-group elements. As you apply it, keep in mind:
elements may form multiple compounds, but a small set tends to be “most typical” for that family
heavier elements in a family can show more varied behaviour, yet the valence-structure link still provides a strong first prediction
FAQ
Less reliably. Transition metals often have multiple accessible oxidation states and can form complexes with different geometries and ligand sets. Group trends exist, but “analogous” is a weaker prediction than for main-group families.
Larger atoms have more inner-shell shielding and more energetically similar subshells available for bonding. This can broaden the range of stable bonding arrangements and oxidation states, so the “family template” becomes less strict.
Identify whether the family is metallic or nonmetallic. Metals in a family commonly form ionic compounds with nonmetals, while nonmetals in a family commonly form molecular compounds with other nonmetals.
Often, but not always. Some families show strong repeating patterns for certain compound classes (like halides), while other classes can vary due to stability differences, bonding preferences, or the ability to form multiple oxidation states.
Compare the role each element plays (e.g., same family member substituting for another) and the overall bonding type and stoichiometry pattern. If swapping one group member for another keeps the structural/bonding “template,” they are likely analogous.
Practice Questions
(1–3 marks) Explain why elements in the same group of the periodic table tend to form analogous compounds.
States that elements in the same group have similar numbers/arrangements of valence electrons (1).
Links similar valence electrons to similar bonding/ion formation and hence similar compound stoichiometries/formulas (1).
Mentions that this leads to similar chemical behaviour/compound types across the group (1).
(4–6 marks) A student claims that if two elements are in the same group, they will always form compounds with identical properties. Evaluate this claim using the idea of valence electron structure and periodic trends within a group.
Explains that same-group elements have similar valence electron structures, so they tend to form analogous compounds (1).
Connects analogous compounds to similar bonding patterns/stoichiometries (1).
States the claim is not fully correct: properties need not be identical (1).
Gives a valid reason for differences down a group, e.g. changing atomic size/shielding affecting bond strength, lattice energy, polarity, or reactivity (1).
Makes a clear distinction between “similar compound types/formulas” and “identical properties/conditions” (1).
Uses correct chemical language throughout (1).

