AP Syllabus focus: ‘Whether two elements form a bond depends on interactions between their valence electrons and the nuclei of the atoms involved.’
Chemical bonding is best understood by focusing on valence electrons, the electrons most exposed to other atoms. Bonds form when electrostatic attractions and repulsions between nuclei and valence electrons lead to a lower-energy, more stable arrangement.
Valence electrons as the drivers of chemical change
Valence electrons are the outermost electrons of an atom and are the ones directly involved in bonding because they experience the weakest attraction to their own nucleus (relative to core electrons) and the strongest interaction with other atoms.
Valence electrons: electrons in the highest occupied energy level (outer shell) of an atom that are available for bond formation.
Core (inner) electrons are usually not involved in bonding because:
they are much closer to the nucleus and are held more strongly
they are shielded from other atoms by the valence shell
moving them would require large amounts of energy without providing comparable stabilisation
Electrostatic interactions: why atoms attract (or repel)
Bond formation depends on the balance of attractions and repulsions among:
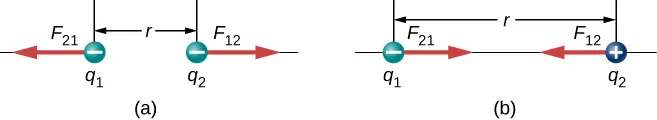
Coulomb’s-law force diagram for two point charges separated by distance , showing repulsion for like charges and attraction for unlike charges. The equal-and-opposite force vectors emphasize that attractions/repulsions act along the internuclear line, matching the qualitative bonding argument that smaller strengthens interactions. Source
positively charged nuclei of both atoms
negatively charged valence electrons on each atom
electron–electron repulsions and nucleus–nucleus repulsions
The key stabilising interaction is the attraction between a nucleus of one atom and the valence electrons of the other atom. As atoms approach, attractions increase, but so do repulsions; a bond forms when attractions dominate at an optimum distance.
= electrostatic force between charges (N)
= charge 1 (C)
= charge 2 (C)
= distance between charges (m)
= Coulomb constant ()
In bonding explanations, Coulomb’s law is mainly used qualitatively:
shorter distance ( smaller) increases both attraction and repulsion
greater charge magnitude (more positive nucleus or more negative electron density) strengthens interactions
a stable bond length corresponds to a minimum in potential energy (net attraction without excessive repulsion)
How valence electrons enable lower-energy arrangements
Atoms form bonds when rearranging valence electrons can decrease the overall potential energy of the system. This typically occurs by:
increasing attractive nucleus–electron interactions between atoms
achieving a more stable valence-shell arrangement (often described by “filled” outer shells, though this is a model, not a universal rule)
Because valence electrons are the electrons being redistributed or shared, they are central to determining:
whether a bond can form at all
what type of bond is favoured (transfer vs sharing)
bond strength trends within similar types of bonding (stronger attraction when electrons are held tightly between nuclei)
Bond formation pathways focused on valence electrons
Electron transfer: forming ions through valence electron loss/gain
If one atom’s valence electrons are weakly held while another atom strongly attracts additional electrons, valence electron transfer can occur. This produces oppositely charged ions, and their electrostatic attraction holds the substance together.
atom losing valence electron(s) becomes a cation
atom gaining valence electron(s) becomes an anion
attraction is between ion charges, ultimately arising from nucleus–electron interactions across the crystal
Electron sharing: concentrating valence electron density between nuclei
If neither atom can easily lose or gain electrons completely, atoms can form a bond by sharing valence electrons. Shared electron density between the nuclei increases nucleus–electron attraction from both atoms to the same electrons, which can lower the system’s energy.
shared electrons are still valence electrons, now influenced by two nuclei
bond formation reflects a balance: nucleus–electron attractions vs electron–electron and nucleus–nucleus repulsions
the shared pair is stabilising because it increases attractive interactions in the internuclear region
Using valence electron patterns to anticipate bonding tendency
A practical AP-level perspective is that bond formation likelihood increases when:
an atom has valence electrons that are relatively easy to redistribute (less tightly held)
another atom provides a strong electrostatic “pull” on those electrons
the resulting arrangement allows stronger attractions at a stable internuclear distance
Thus, whether two elements form a bond depends on how their nuclei interact electrostatically with each other’s valence electrons, and whether those interactions can outweigh the repulsions that appear as the atoms get close.
FAQ
Lewis dots show only valence electrons as dots around an element symbol.
They do not show 3D shape, orbital overlap, or relative energies; they are a bookkeeping model for valence electron counting rather than a physical picture.
Their valence shells are already low in energy and relatively stable.
As a result, there is little energetic benefit to rearranging their valence electrons, and many approaches by other atoms do not produce a net lowering of energy.
For elements in period 3 and beyond, additional available orbitals and electron density distribution can permit “expanded” bonding descriptions.
In practice, many such cases are better treated as models of electron density rather than literal extra electron pairs in fixed locations.
Formal charge is a valence-electron accounting tool used in structures:
assign bonding electrons equally between atoms
compare assigned electrons to the atom’s valence count
It helps evaluate which electron distributions are more plausible, even though it is not a direct measurement of charge.
In metals, valence electrons are often described as delocalised across many nuclei rather than localised between a specific pair.
This collective attraction between positive metal centres and mobile valence electrons helps explain conductivity and malleability.
Practice Questions
(3 marks) Explain why valence electrons, rather than core electrons, are involved in chemical bond formation.
Identifies valence electrons as outer-shell electrons that interact with other atoms (1)
Explains core electrons are closer to the nucleus and more strongly attracted/less available (1)
Links bonding to redistribution or sharing of these outer electrons to lower energy / increase attractions (1)
(6 marks) Using electrostatic ideas, describe how interactions between nuclei and valence electrons can lead to either electron transfer or electron sharing when two different atoms form a bond. You may refer to Coulomb’s law qualitatively.
States that bonding depends on attractions between nuclei and valence electrons of the other atom (1)
Notes repulsions also exist (electron–electron and nucleus–nucleus) and a bond forms when net attraction is achieved at an optimum distance (1)
Explains electron transfer as movement of valence electron(s) from an atom holding them weakly to an atom attracting them strongly, forming ions (1)
States resulting cation–anion attraction is electrostatic and holds the bonded structure together (1)
Explains electron sharing as valence electron density located between nuclei, increasing attraction of both nuclei to the shared electrons (1)
Uses Coulomb’s law qualitatively: stronger interaction with higher charge magnitude and/or shorter distance (1)

