AP Syllabus focus: ‘Explain trends in elemental reactivity using periodicity and valence electron interactions, connecting patterns to positions on the periodic table.’
Elemental reactivity is not random: it follows periodic patterns because valence electrons experience different attractions to the nucleus across periods and down groups. These attractions control how easily atoms lose or gain electrons during reactions.
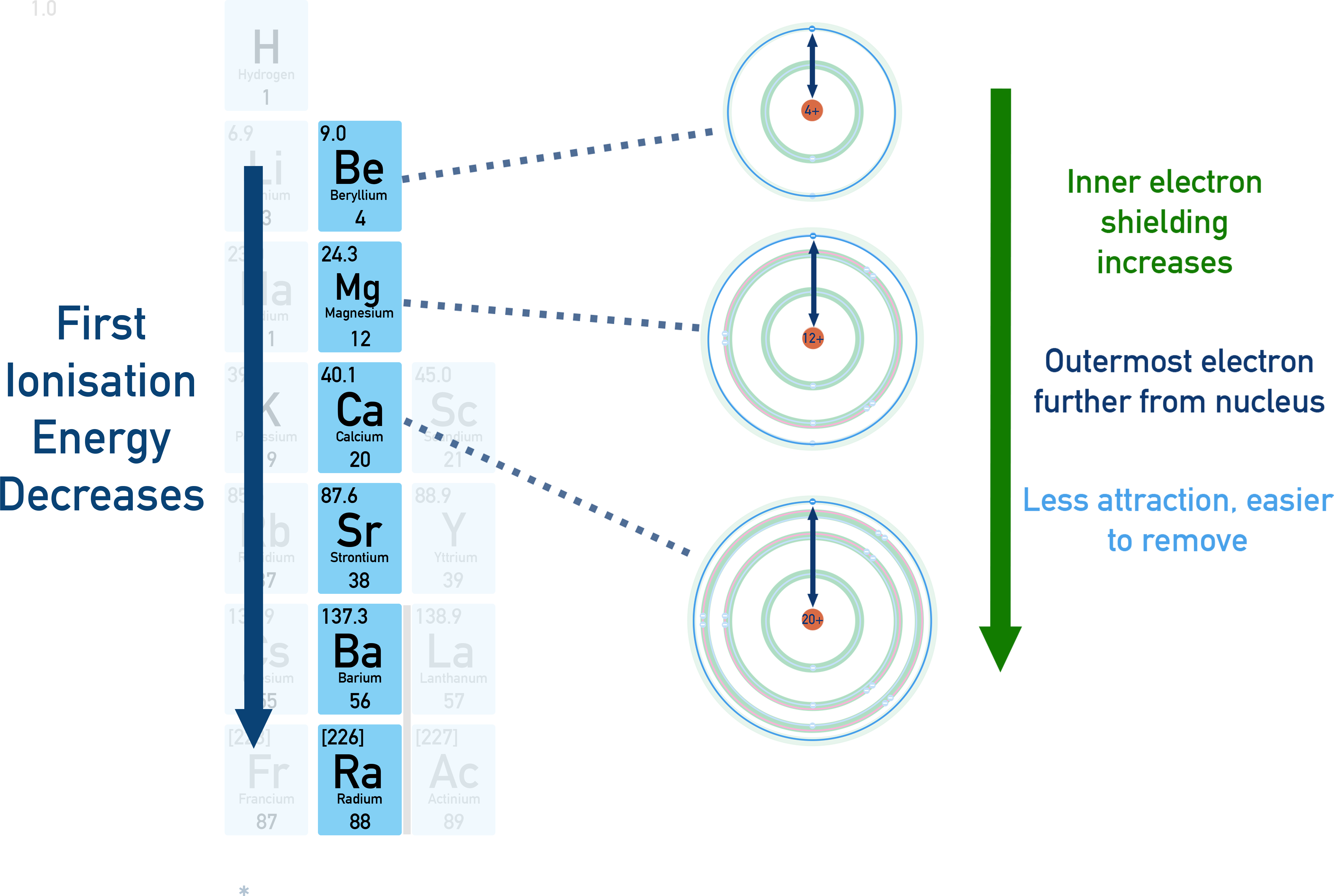
Diagram showing why first ionization energy decreases down a group: added energy levels increase shielding and place the valence electron farther from the nucleus, reducing electrostatic attraction. Lower ionization energy corresponds to easier electron loss, which helps explain why metallic reactivity increases down groups such as the alkali metals. Source
What “reactivity” means in periodic trends
Reactivity depends on how strongly an atom’s valence electrons are held and how favorable it is to reach a more stable valence electron arrangement (often a noble-gas-like configuration).
Reactivity: the tendency of an element to undergo chemical change, often by losing or gaining electrons in redox processes.
In general:
Metals react by losing valence electrons (forming cations).
Nonmetals react by gaining valence electrons (forming anions) or by sharing electrons in covalent bonds, but periodic reactivity trends are commonly explained using electron gain/loss tendencies.
Valence electrons and nuclear attraction
Periodic trends in reactivity are explained by the competition between:
Attraction between the nucleus and valence electrons (stronger attraction makes electron loss harder but electron gain easier)
Distance of valence electrons from the nucleus (greater distance weakens attraction)
Shielding by core electrons (more shielding reduces the nucleus’ pull on valence electrons)
These factors vary predictably with an element’s position on the periodic table.
Metallic reactivity trends (electron loss)
For metals, higher reactivity means easier oxidation (easier loss of electrons).
Oxidation: loss of electrons by a species (often forming a cation).
Down a group: metals become more reactive
Down a group (e.g., alkali metals, alkaline earth metals):
Valence electrons occupy higher energy levels farther from the nucleus
Increased shielding reduces nuclear attraction to valence electrons
Electrons are lost more easily, so metal reactivity increases
This is why Group 1 metals become more reactive from Li to Cs in typical chemical reactions.
Across a period: metals become less reactive
Across a period from left to right (within the metallic region):
Nuclear charge increases while shielding changes less dramatically
Valence electrons are attracted more strongly
Electron loss becomes harder, so metal reactivity generally decreases
So, within a given period, a Group 1 metal is typically more reactive than a Group 2 metal.
Nonmetal reactivity trends (electron gain)
For nonmetals, higher reactivity often means greater tendency to gain electrons (be reduced) or attract electron density.
Reduction: gain of electrons by a species (often forming an anion).
Up a group: nonmetals become more reactive
Up a group (especially the halogens):
Valence electrons are closer to the nucleus and experience less shielding
The nucleus attracts incoming electrons more strongly
Nonmetal reactivity increases upward
Thus, F is generally more reactive than I as an oxidizing agent in many contexts.
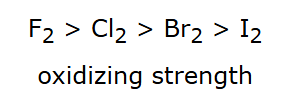
Figure giving the halogen oxidizing-strength trend , indicating decreasing tendency to gain electrons (be reduced) down Group 17. This ordering is commonly used to justify why fluorine is the strongest oxidizing agent among the common halogens while iodine is much weaker. Source
Across a period: nonmetals become more reactive
Across a period (toward the upper right, excluding noble gases):
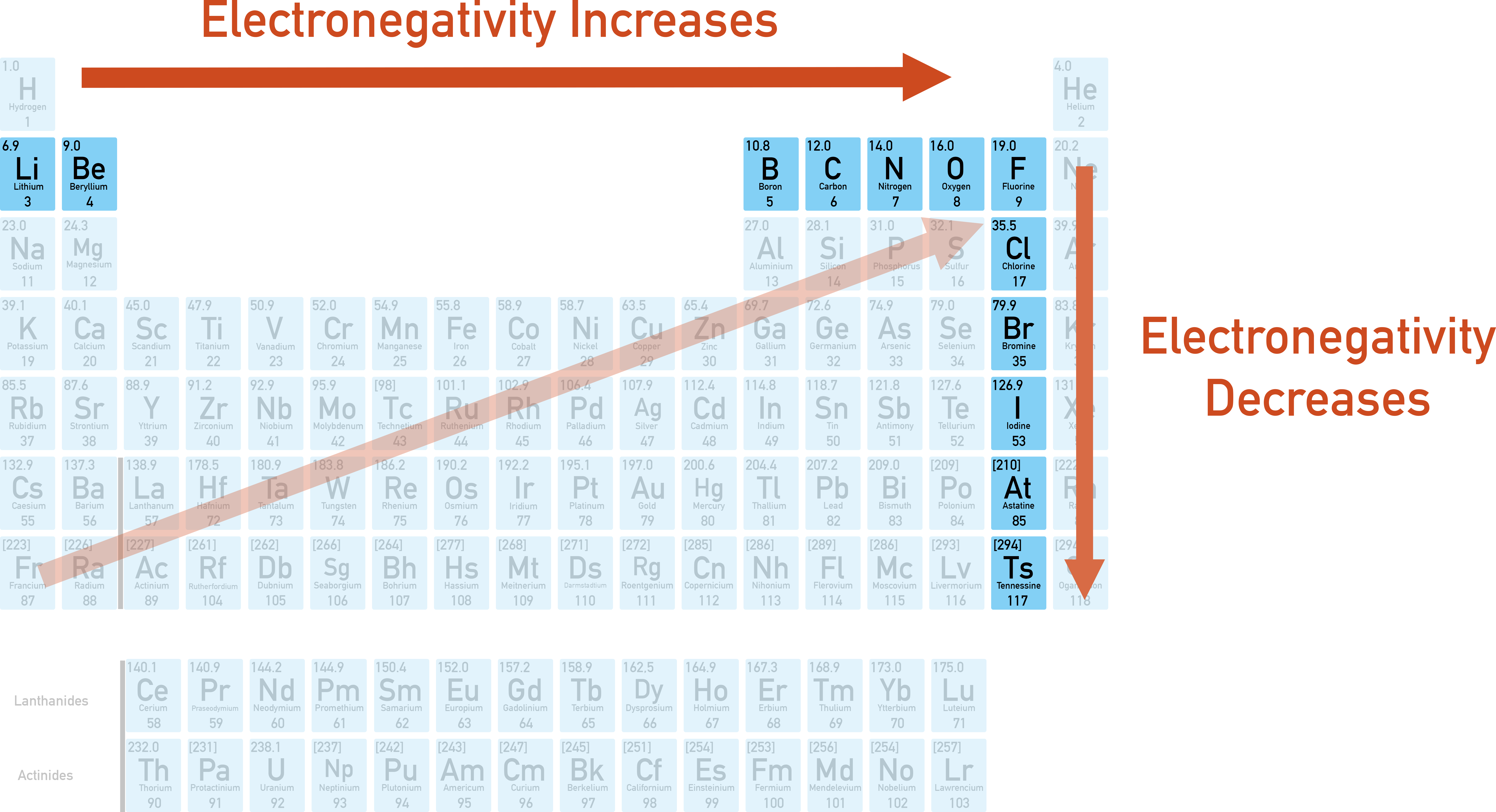
Periodic-table trend map showing electronegativity increasing up and to the right, peaking near fluorine. Higher electronegativity indicates stronger attraction for shared or incoming electron density, aligning with the increasing nonmetal reactivity described for the upper-right region (excluding noble gases). Source
Effective nuclear attraction for added electrons increases
Atoms more strongly attract electrons to complete their valence shell
Nonmetal reactivity increases left to right within the nonmetal region
Noble gases are a key exception in trend discussions because a filled valence shell makes electron gain/loss unfavorable, so they are typically very unreactive under normal conditions.
Connecting periodic position to reaction tendencies
When justifying reactivity trends, use position-based reasoning tied to valence electrons:
Elements on the far left: few valence electrons; react by losing them readily (more metallic reactivity, especially down a group)
Elements near the top right (nonmetals): many valence electrons; react by gaining electrons readily (more nonmetal reactivity, especially up a group)
Middle regions: trends are less extreme; reactivity arguments still rely on valence electron attraction and likelihood of electron transfer
What a strong explanation includes
Identifies whether the element reacts mainly by electron loss (metal) or electron gain (nonmetal)
Links reactivity to changes in distance, shielding, and nuclear attraction across/down the periodic table
Explicitly connects the change to the ease of forming ions with stable valence electron arrangements
Common pitfalls to avoid
Treating “reactivity” as a single trend for all elements; metals and nonmetals trend in opposite directions
Claiming atoms “want” electrons; instead, state that electron transfer becomes more energetically favorable due to nuclear attraction and electron configuration stability
Forgetting that periodic trends explain tendencies, not absolute outcomes in every possible reaction system
FAQ
Solvent effects can stabilise certain ions more than others.
Hydration can favour formation of smaller, more highly charged ions.
Side reactions (e.g., with water) can change the observed products, altering “apparent” reactivity.
Some metals form protective oxide layers that slow further reaction.
This can make an element seem less reactive in practice even if electron-loss trends predict higher reactivity.
Their valence shells are filled, so both electron gain and electron loss tend to be energetically unfavourable.
Reactivity can occur under special conditions, but it is not well captured by basic periodic electron-transfer arguments.
Not reliably.
Variable oxidation states and involvement of $d$ electrons can complicate predictions, so position-based valence arguments are less straightforward than for Groups 1, 2, 17, and 18.
“More reactive” is broad, while redox strength is specific.
A strong reducing agent is easily oxidised (common for very reactive metals).
A strong oxidising agent is easily reduced (common for very reactive halogens).
Practice Questions
(2 marks) Explain, using periodic trends, why potassium is more reactive than sodium.
1 mark: Down Group 1, valence electron is further from the nucleus and/or more shielded.
1 mark: Weaker attraction makes electron loss easier (oxidation more likely), so reactivity increases.
(5 marks) Compare the reactivity trends of Group 1 metals and Group 17 nonmetals. In your answer, relate the trends to valence electrons and attraction to the nucleus.
1 mark: Group 1 metals react by losing one valence electron (forming ions).
1 mark: Down Group 1, increased distance/shielding reduces attraction; electron loss becomes easier; reactivity increases.
1 mark: Group 17 nonmetals react by gaining one electron (forming ions).
1 mark: Up Group 17, decreased distance/shielding increases attraction; electron gain becomes easier; reactivity increases.
1 mark: Clear comparison noting opposite vertical trends (Group 1 increases down; Group 17 increases up).

