AP Syllabus focus: ‘Typical charges in ionic compounds are governed by valence electron count and can be predicted from an element’s location in the periodic table.’
Predicting ion charges quickly is a core skill for writing correct ionic formulas and interpreting chemical behavior. The periodic table encodes valence electron patterns, letting you infer the most common monatomic ion charges for many elements.
Linking periodic position to ionic charge
Valence electrons drive typical charges
Main-group elements (representative elements) tend to form ions that achieve a noble-gas electron configuration by losing or gaining valence electrons.

Lewis-symbol diagrams and examples showing how atoms form ions by losing or gaining valence electrons. The figure contrasts cation formation (fewer dots after electron loss) with anion formation (a filled octet after electron gain), reinforcing the “noble-gas configuration” idea at the particle level. Source
Ion (monatomic): An atom with a net electric charge because it has lost or gained one or more electrons.
Metals usually form cations (positive ions) by losing electrons; nonmetals usually form anions (negative ions) by gaining electrons. The magnitude of the charge often equals the number of electrons lost or gained to reach the nearest noble gas configuration.
Group trends for common monatomic ion charges
Use an element’s group (column) for fast predictions (especially for Groups 1, 2, 13, 15–17):
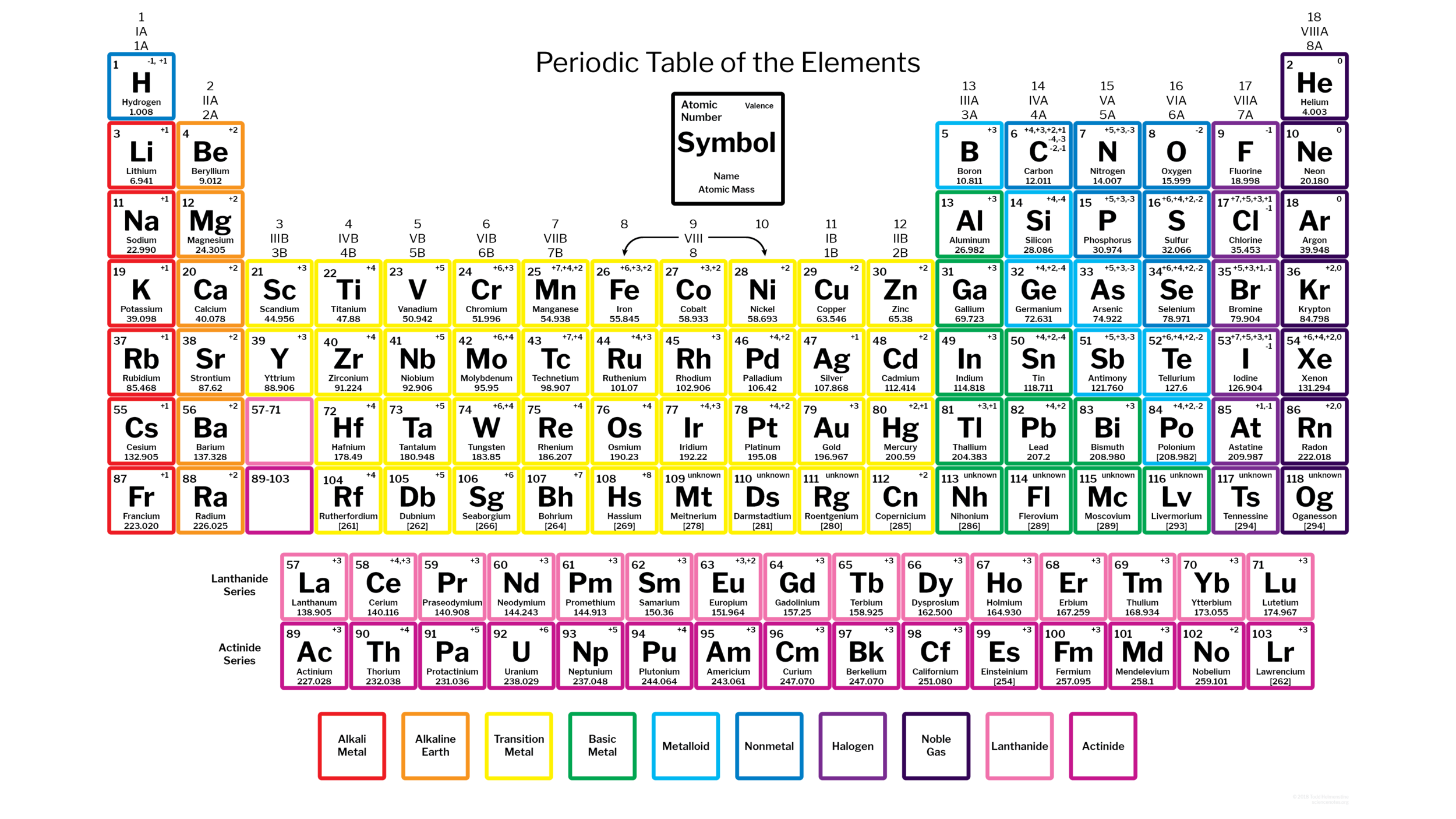
Periodic table annotated with common ionic charges/oxidation states for many elements, making the main-group charge patterns (, , , , , ) visually easy to spot by column. It also highlights that several metals (especially transition metals) can exhibit multiple common charges rather than a single fixed value. Source
Group 1 (alkali metals): form ions (lose 1 e)
Group 2 (alkaline earth metals): form ions (lose 2 e)
Group 13: form ions (lose 3 e)
Group 15: form ions (gain 3 e)
Group 16: form ions (gain 2 e)
Group 17 (halogens): form ions (gain 1 e)
Group 18 (noble gases): typically (rarely form ions)
These are “typical” charges because they are the most common in ionic compounds for those families.
Using predicted charges to build neutral ionic formulas
Charge neutrality requirement
Ionic compounds are electrically neutral overall: total positive charge balances total negative charge.
= charge contribution of each ion (in units of the elementary charge)
To apply this idea when predicting formulas:
Identify each element’s most likely ion charge from its periodic position.
Choose subscripts so the total charge sums to zero.
Reduce subscripts to the lowest whole-number ratio.
Practical cues and common exceptions to “simple” patterns
Hydrogen commonly forms (when bonded to nonmetals) but can form in metal hydrides; rely on context when it appears in ionic compounds.
Group 14 elements do not have a single reliable monatomic ion charge in typical ionic compounds; predicting or from the group alone is often misleading.
Transition metals (d-block) can form multiple cation charges (for example, and are common). Periodic position alone is less definitive here, so charge is often given explicitly in names or formulas.
Heavier p-block metals (post-transition metals) may show more than one common cation charge; treat “group charge” as a starting guess, not a guarantee.
What to prioritise on the AP Chemistry exam
Be fluent with main-group typical charges (Groups 1, 2, 13, 15–17).
Justify predictions using valence electron count and periodic-table location.
Use predicted charges to enforce overall neutrality when writing ionic formulas.
FAQ
Forming $4+$ or $4-$ can require removing or adding too many electrons, which is often energetically unfavourable.
Many Group 14 elements prefer covalent bonding or form ions with different charges depending on bonding and structure.
Look for information provided in the compound name (e.g., iron(III)) or the formula.
If neither is provided, common possibilities include $+2$ and $+3$, but you generally cannot deduce a single charge from position alone.
Hydrogen’s charge depends on whether it loses electron density (with nonmetals) or gains it (with very electropositive metals).
In metal hydrides, hydrogen is present as hydride, $H^-$.
For monatomic ions in ionic compounds, the ion charge and oxidation number typically match.
In more complex substances (especially covalent compounds), oxidation numbers are accounting tools and may not correspond to actual ion charges.
They may show multiple stable charges due to stabilisation of certain electron configurations in heavier atoms.
When multiple charges are plausible, you usually need the compound’s name, formula, or experimental context to decide between them.
Practice Questions
(3 marks) Using the periodic table, state the most likely charge on the ions formed by: a) potassium b) oxygen c) aluminium
a) (1)
b) (1)
c) (1)
(6 marks) A compound forms between magnesium and nitrogen. a) Predict the charge on a magnesium ion and explain your reasoning using periodic-table position. (2 marks) b) Predict the charge on a nitride ion and explain your reasoning using periodic-table position. (2 marks) c) Write the empirical formula of the compound formed, showing how charge neutrality determines the subscripts. (2 marks)
a) Mg is in Group 2 so forms (1); explanation: loses 2 valence electrons to reach noble gas configuration (1)
b) N is in Group 15 so forms (1); explanation: gains 3 electrons to reach noble gas configuration (1)
c) Correct formula (1); charge balance shown: or equivalent reasoning (1)

