AP Syllabus focus: ‘Explain how absorption or emission in different electromagnetic regions corresponds to specific molecular motions or electronic transitions.’
Spectroscopy links macroscopic measurements of absorbed or emitted light to microscopic energy changes in molecules. Different regions of the electromagnetic spectrum deliver different photon energies, so they drive different kinds of molecular transitions.
Big idea: spectrum region tells you the type of transition
Absorption vs emission
When matter interacts with electromagnetic radiation, it can change energy only in specific allowed steps.
Absorption: a molecule takes in a photon and moves to a higher-energy state.
Emission: a molecule releases a photon when dropping to a lower-energy state.
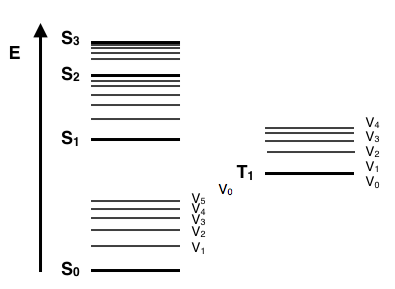
Jablonski-style energy-level diagram showing quantized electronic states (e.g., ) and transitions between them. Upward arrows represent absorption to an excited electronic state, while downward radiative pathways represent emission as the system returns toward the ground state. Source
Spectroscopic transition: a change between quantised energy states of an atom or molecule caused by absorbing or emitting a photon.
Quantised energy levels and why region matters
Molecules have multiple “layers” of energy structure, and the spacings differ in size. Smaller spacings require lower-energy photons (longer wavelength); larger spacings require higher-energy photons (shorter wavelength).
Quantised energy levels: discrete (not continuous) allowed energies for molecular rotation, vibration, or electronic structure.
In AP Chemistry, the key connection is that microwave photons match rotational spacings, infrared photons match vibrational spacings, and visible/UV photons match electronic spacings.
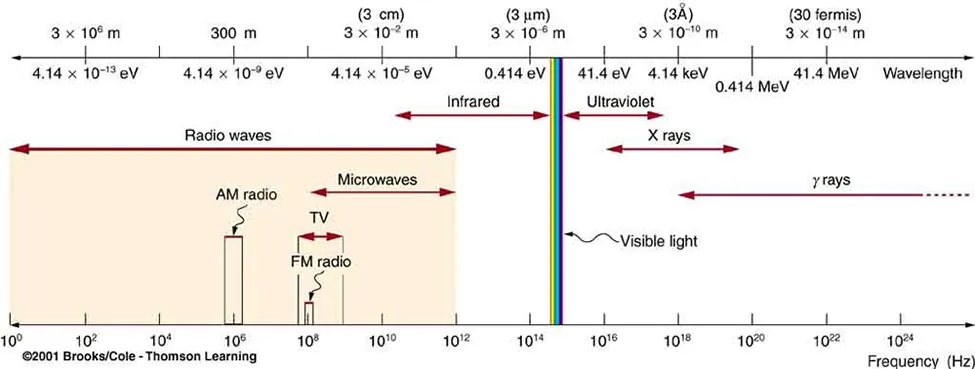
Electromagnetic spectrum overview showing how wavelength, frequency, and photon energy change across regions (radio → microwave → IR → visible → UV → X-ray → gamma). This provides the physical basis for why microwaves typically access smaller rotational energy spacings, while visible/UV photons are energetic enough to promote electronic transitions. Source
Key regions and what they probe
Microwave radiation → molecular rotation
Microwave radiation drives transitions between rotational energy levels.
Most relevant for gas-phase molecules (rotations are freer than in liquids/solids).
Typically associated with changes in the way a molecule spins as a whole.
Requires an effective dipole change during rotation; many homonuclear diatomics (e.g., , ) are not microwave-active.
Infrared radiation → molecular vibration
Infrared (IR) radiation drives transitions between vibrational energy levels.
Vibrations include stretching and bending motions of bonds.
IR absorption is strongest when a vibration involves a changing dipole moment (common in polar bonds).
Because different bond types have different stiffness and atomic masses, IR spectra can help identify functional groups through characteristic absorption regions.
Visible/ultraviolet radiation → electronic transitions
Visible and UV radiation drives transitions between electronic energy levels.
Involves promotion of electrons to higher-energy orbitals or excited electronic states.
Visible absorption often leads to colour (the transmitted/reflected light is missing the absorbed wavelengths).
UV–Vis transitions are commonly discussed for species with delocalised electrons or easily excited electrons, which can produce strong absorption bands.
Interpreting spectra qualitatively (AP emphasis)
You should be able to connect an observed absorption/emission region to the likely molecular change:
Microwave signal change ⇒ rotation changed.
IR absorption peak(s) ⇒ bond vibrations changed.
Visible/UV absorption/emission ⇒ electronic state changed.
A useful qualitative trend is:
Higher frequency (shorter wavelength) radiation corresponds to larger energy gaps in the molecule, so it tends to cause electronic rather than vibrational/rotational transitions.
Common AP-level connections and pitfalls
Do not treat “spectroscopy” as one process; the region determines the molecular motion or electronic change being probed.
A molecule can have rotational, vibrational, and electronic energy simultaneously; spectroscopy often isolates which spacing best matches the photon energy.
Absorption features reflect allowed transitions; “no absorption” in a region can indicate the transition is not energetically matched or not spectroscopically active.
FAQ
IR absorption requires the vibration to produce a change in dipole moment.
Symmetric vibrations can leave the net dipole unchanged.
Non-polar molecules may have vibrations that do not create a temporary dipole change strong enough for IR absorption.
Changing an atom to a heavier isotope increases the reduced mass, typically lowering the vibrational frequency.
Heavier isotope → slower vibration → absorption shifts to lower wavenumber.
Bond strength is similar, so the shift is mainly mass-related.
Electronic transitions are accompanied by many closely spaced vibrational/rotational sublevels.
In condensed phases, collisions and solvent interactions broaden features further.
The result is a band representing many nearly overlapping transitions.
In liquids and solids, rotational motion is hindered by strong intermolecular interactions.
Rotational energy levels become less distinct.
Collisions and restricted rotation smear out or suppress clear microwave transitions.
UV–Vis absorption is stronger when electrons are easily promoted.
Conjugation (delocalised $\pi$ systems) often lowers the energy required.
Certain functional groups act as chromophores, increasing absorption intensity at specific wavelengths.
Practice Questions
(2 marks) Which type of molecular transition is primarily associated with absorption of infrared radiation? State the transition and one brief description of what changes.
1 mark: Identifies vibrational transition (between vibrational energy levels).
1 mark: Describes bond stretching/bending (bond vibration changes).
(5 marks) A substance shows strong absorption in the ultraviolet region but little to no absorption in the microwave region.
(a) Identify the type of transition responsible for UV absorption. (1)
(b) Explain what this indicates about the relative size of the energy gap involved compared with IR and microwave transitions. (2)
(c) Give one reason a molecule might show little or no microwave absorption. (2)
(a)
1 mark: Electronic transition.
(b)
1 mark: UV corresponds to higher frequency/shorter wavelength than IR/microwave.
1 mark: Therefore UV involves a larger energy spacing than vibrational (IR) and rotational (microwave) spacings.
(c) Any one reason, with explanation:
1 mark: Example reason: molecule has no permanent dipole / rotation does not produce a changing dipole.
1 mark: Links this to microwave rotational transitions being inactive/weak (selection rule/dipole requirement).

