AP Syllabus focus: ‘Infrared radiation is associated with transitions between molecular vibrational energy levels, used to probe bond vibrations within molecules.’
Infrared (IR) spectroscopy connects molecular-scale bond vibrations to measurable absorption of IR light. In AP Chemistry, focus on how quantized vibrational energy changes produce characteristic absorptions that reveal which bonds are present.
What IR Radiation Probes
Infrared photons have energies that match vibrational energy level spacings in molecules. When a molecule absorbs IR radiation, it undergoes a transition to a higher vibrational state for a particular bond motion.
Vibrational transition: absorption of IR radiation that promotes a molecule from one quantized vibrational energy level to a higher one for a specific vibrational mode.
IR spectroscopy is therefore used to probe bond vibrations within molecules, linking absorption features to the presence of particular bond types (and sometimes aspects of molecular environment).
Vibrational Modes: How Bonds Can Move
Molecules do not vibrate in arbitrary ways; they vibrate in specific normal modes. The two broad classes of modes emphasized at this level are:
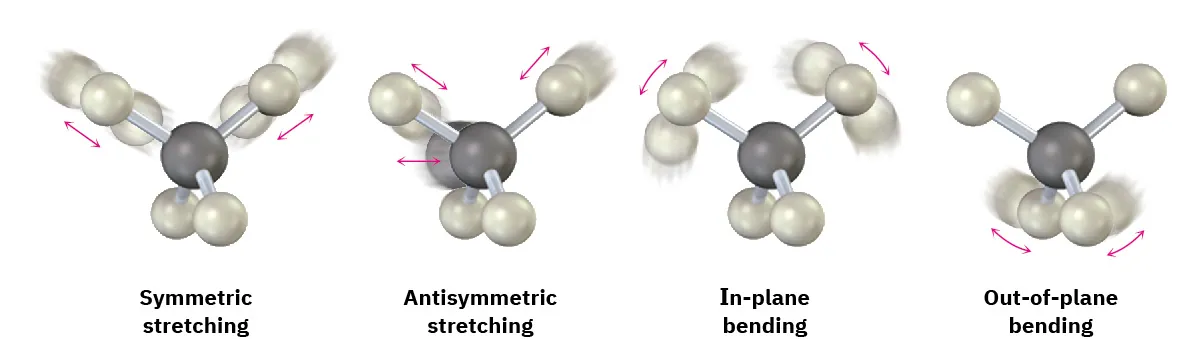
Examples of common IR vibrational modes are shown, separating stretching motions (symmetric vs. antisymmetric changes in bond length) from bending motions (in-plane vs. out-of-plane changes in bond angles). The arrows emphasize that each “peak” in an IR spectrum corresponds to a specific, quantized pattern of atomic motion rather than random vibration. Source
Stretching vibrations
Change the bond length
Commonly higher in energy than bending
Includes:
Symmetric stretching (bonds lengthen/shorten together)
Asymmetric stretching (one bond lengthens as another shortens)
Bending vibrations
Change bond angles
Often occur at lower energies than stretching
Includes motions such as scissoring, rocking, wagging, and twisting (naming details are less important than recognising “angle change” vs “length change”).
What Makes a Vibration IR-Active
Not every vibration leads to IR absorption. To interact strongly with IR radiation, the vibrating molecule must create a changing electrical imbalance during the vibration.
A vibration is typically IR-active when it involves a change in dipole moment during the motion.
Vibrations that do not change the dipole moment are weak or absent in an IR spectrum.
This explains why some symmetric vibrations can be less visible: if the bond polarity changes cancel out during a symmetric motion, there may be little net dipole change, reducing absorption.
How IR Spectra Are Commonly Displayed
IR spectra are often plotted with the x-axis as wavenumber (related to frequency/energy) rather than wavelength.
= wavenumber (commonly in )
= wavelength (commonly in cm)
Higher wavenumber corresponds to higher-energy vibrations (typically stronger bonds and lighter atoms). A spectrum usually shows peaks (or dips in % transmittance) at wavenumbers where IR light is absorbed.
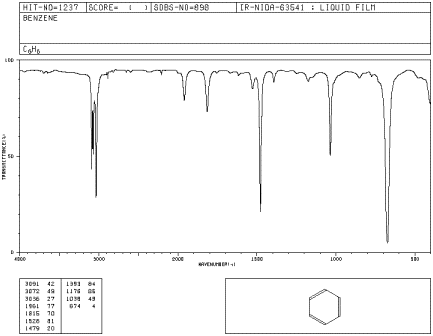
A representative IR spectrum (benzene) illustrates the standard IR plotting convention: wavenumber () along the x-axis and percent transmittance on the y-axis. Absorptions appear as downward features because lower %T means more IR light was absorbed at that vibrational frequency. Source
What Controls the Position and Appearance of Absorptions
Bond strength and atomic masses (qualitative trends)
The vibrational frequency depends on how hard it is to stretch/bend the bond and on the masses of the atoms involved.
Stronger bonds (e.g., multiple bonds) generally vibrate at higher wavenumber
Bonds to hydrogen often appear at relatively high wavenumber because hydrogen is very light
Heavier atoms in a bond generally shift vibrations to lower wavenumber (slower vibration)
These trends help justify why different bond types appear in different regions of an IR spectrum.
Intensity and peak shape
Peak intensity increases when the vibration causes a larger change in dipole moment.
Broader peaks can occur when a range of slightly different environments exists in the sample (common in condensed phases).
Some absorptions can overlap, especially in complex molecules, because many vibrations may occur in similar energy ranges.
Using IR to Identify Bond Types (Conceptual Use)
IR spectroscopy is used to infer the presence of certain bonds by matching observed absorptions to expected vibrational frequencies.
When interpreting qualitatively:
Treat each absorption as evidence of a bond vibration consistent with that frequency.
Recognise that a molecule has many modes; the observed spectrum is a combined “fingerprint” of its vibrations.
Use the idea that IR involves vibrational energy level transitions, not electronic transitions, to stay focused on bond motion.
FAQ
Splitting can occur when similar bonds are in slightly different chemical environments, giving slightly different vibrational energies.
It can also arise from coupling between vibrational modes that are close in energy.
Hydrogen bonding weakens and lengthens the O–H bond on average, typically shifting the O–H stretch to lower wavenumber.
It also creates a distribution of bond strengths, often broadening the absorption substantially.
Overtones are transitions to higher excited vibrational levels beyond the first, such as $v=0 \rightarrow 2$.
They are typically weak because their transition probabilities are much lower than the fundamental transitions.
Replacing an atom with a heavier isotope increases the effective mass in the vibration, lowering the vibrational frequency.
As a result, the absorption usually shifts to lower wavenumber, often noticeably for H/D substitution.
Neighbouring atoms and electron distribution alter bond strength (and thus vibrational energy), shifting absorptions.
Conjugation, inductive effects, and local polarity can all slightly change the bond’s vibrational frequency.
Practice Questions
(2 marks) Explain what happens at the molecular level when a sample absorbs infrared radiation.
Mentions absorption promotes molecule between vibrational energy levels / causes a vibrational transition (1)
Links absorption to bond vibrations (stretching/bending) within the molecule (1)
(5 marks) A student states: “All bond vibrations appear in an IR spectrum, and stronger bonds always give more intense peaks.” Evaluate this statement using IR spectroscopy principles.
Not all vibrations appear: absorption requires a change in dipole moment / some modes are IR-inactive (1)
Identifies that vibrations are quantised and IR causes transitions between vibrational energy levels (1)
Stronger bonds typically absorb at higher wavenumber (position), not necessarily higher intensity (1)
Peak intensity relates to the magnitude of dipole moment change during vibration (1)
Notes that symmetric vibrations may be weak/absent due to little/no net dipole change (1)

