AP Syllabus focus: ‘Microwave radiation is associated with transitions between molecular rotational energy levels, linking photon energies to changes in rotational motion.’
Microwave spectroscopy connects the electromagnetic spectrum to molecular motion by showing how molecules absorb low-energy photons to change rotational states. This topic explains what rotational transitions are and what molecular features control them.
What microwave radiation probes
Microwaves have relatively low photon energies, so they typically do not change electronic structure or vibrate bonds. Instead, they can change how a molecule rotates in space.
Rotational energy is quantized
Molecules do not take on any rotation energy continuously; they occupy discrete rotational energy levels. Absorbing a microwave photon can promote a molecule from one rotational level to another.
Rotational energy level: A discrete (quantized) amount of rotational energy a molecule can possess, so that changes in rotation occur as jumps between allowed levels.
These transitions are the key physical meaning behind “microwave radiation is associated with transitions between molecular rotational energy levels.”
Linking photon energy to rotational motion
A microwave photon has energy that must match the energy gap between two rotational levels for absorption to occur. When absorption happens, the molecule’s rotational motion changes (a higher rotational state corresponds to faster or differently oriented rotation, depending on the molecule).
= energy difference between rotational levels, in J
= Planck constant, in J·s
= microwave frequency, in s (Hz)
Because microwave frequencies are much lower than visible/UV frequencies, for rotational transitions is comparatively small, consistent with changing rotation rather than vibration or electronic arrangement.
Which molecules show microwave rotational spectra
Not every molecule absorbs microwaves for rotational transitions. A common requirement is that the molecule has a permanent dipole moment so that the electromagnetic wave can interact with the molecule’s charge distribution during rotation.
Polar molecules (for example, many heteronuclear diatomics) can often absorb microwaves via rotational transitions.
Nonpolar molecules with no permanent dipole generally do not show strong pure rotational microwave absorption because the radiation cannot efficiently couple to the rotating molecule’s charge distribution.
This idea links structure (polarity) to whether rotational transitions are observable.
What controls the spacing of rotational transitions
The photon frequency needed depends on how widely spaced the rotational energy levels are. While AP Chemistry treats this qualitatively, the key molecular factors are:
Mass distribution and size (moment of inertia)
How hard it is to spin a molecule depends on its moment of inertia—how mass is distributed relative to the axis of rotation.
Larger, “more spread out” molecules tend to have larger moments of inertia.
Larger moments of inertia generally lead to more closely spaced rotational energy levels.
More closely spaced levels correspond to lower-frequency microwave photons being sufficient for transitions.
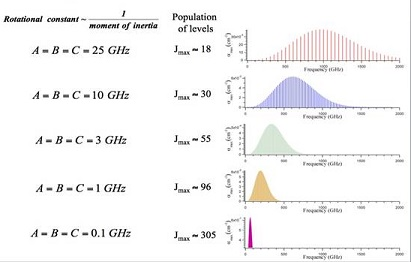
Plot of rotational absorption strength versus frequency for different rotational constants (which are inversely related to moment of inertia). It shows that as molecules get larger (smaller rotational constants, larger moments of inertia), rotational lines crowd together at lower frequencies—exactly the qualitative trend emphasized in AP Chemistry. Source
Molecular structure and rotational axes
Molecules can rotate about different axes, and their shape affects which rotations are possible and how energy levels are arranged.
Linear molecules have simpler rotational behaviour than many nonlinear molecules.
Structural differences change the pattern of absorption lines, which is why microwave spectra can be used as evidence of molecular identity and geometry (at a conceptual level).
Interpreting absorption in words (what you should be able to say)
When you are asked to “link photon energies to changes in rotational motion,” a complete explanation is typically:
Rotational energies are quantized, so only specific rotational energy levels exist.
A molecule absorbs microwave radiation only when the photon energy matches the energy gap between two rotational levels.
Absorption causes a transition to a higher rotational energy level, meaning the molecule’s rotational motion changes.
The microwave frequency required depends on molecular properties (especially mass distribution/size), which determine the spacing of rotational levels.
These points directly restate the syllabus expectation using particle-level reasoning without drifting into unrelated spectroscopy topics.
FAQ
Changing isotopes changes mass distribution and the moment of inertia.
That shifts rotational level spacings and therefore the line positions.
Selection rules are constraints on which quantised transitions are allowed.
For pure rotational absorption, only certain changes in rotational state produce a changing dipole that couples to the radiation.
Rotational levels are discrete, giving discrete transition energies.
At low pressures, fewer collisions occur, so individual lines remain relatively sharp.
Temperature changes the population of molecules in each rotational level.
More molecules in the starting level gives stronger absorption for transitions out of that level.
Line positions depend sensitively on the moment of inertia.
Moment of inertia depends on bond lengths and how atoms are arranged in space, allowing very precise structural inference.
Practice Questions
(2 marks) Explain why microwave radiation can cause a molecule to change its rotational energy but usually does not change its electronic energy.
Mentions microwave photons have relatively low energy compared with electronic transitions. (1)
States absorption promotes a transition between quantised rotational energy levels (change in rotational motion). (1)
(5 marks) A student compares two gases: A is polar and small; B is non-polar and larger. Discuss which gas is more likely to show microwave absorption due to rotational transitions, and how molecular size affects the frequencies absorbed.
States rotational energy levels are quantised and absorption occurs when photon energy matches . (1)
Identifies polar molecule A as more likely to absorb microwaves because a permanent dipole allows interaction with the radiation. (1)
Identifies non-polar molecule B as unlikely to show strong pure rotational microwave absorption. (1)
Explains larger molecules tend to have larger moment of inertia/mass further from axis. (1)
Links larger moment of inertia to smaller energy spacing and thus lower frequencies required. (1)

