AP Syllabus focus: ‘Ultraviolet and visible radiation are associated with transitions between electronic energy levels, explaining many UV–Vis absorption and emission phenomena.’
Ultraviolet–visible (UV–Vis) spectroscopy connects light absorption or emission to changes in a molecule’s electronic structure. Understanding which transitions occur, and what shifts their energies, explains observed spectra and colours.
What UV–Vis Radiation Does
UV (higher energy) and visible (lower energy) photons can promote electrons to higher-energy arrangements within atoms, ions, or molecules. The result is an electronic transition that changes the species’ electronic energy.
UV–Vis measurements commonly track:
Absorption: a decrease in transmitted light at selected wavelengths because photons are taken up by the sample.
Emission: light released when an excited species relaxes to a lower electronic energy state.
Key Energy-State Language
Electronic energy level: a quantized (discrete) allowed energy for electrons in an atom, ion, or molecule; electrons can change levels only by absorbing or emitting specific energies.
Electronic energy levels are separated by gaps that often match UV or visible photon energies, making UV–Vis ideal for probing electronic structure.
Electronic Transitions in Molecules
In molecular substances, UV–Vis absorption often involves electrons in bonding and nonbonding regions moving into higher-energy antibonding regions. Common qualitative transition types include:
: nonbonding (lone-pair) electron promoted to an antibonding pi orbital (often lower-energy than many other UV transitions).
: pi-bonding electron promoted to an antibonding pi orbital (common in double bonds and aromatic rings).
Which transitions are possible depends on:
The presence of lone pairs (e.g., O, N, halogens).
Pi systems (double bonds, aromatic rings).
The relative spacing between available electronic energy levels.
Relating Wavelength to Transition Energy
UV photons correspond to larger energy gaps than visible photons, so species with small electronic energy gaps can absorb in the visible region and appear coloured.
= photon energy (J per photon)
= Planck’s constant (J·s)
= frequency (s)
= speed of light (m·s)
= wavelength (m)
If the photon energy matches the gap between two electronic energy levels, absorption can occur and an electron is promoted to an excited state.
Absorption Spectra and Observed Colour
An absorption spectrum plots how strongly a sample absorbs as a function of wavelength.
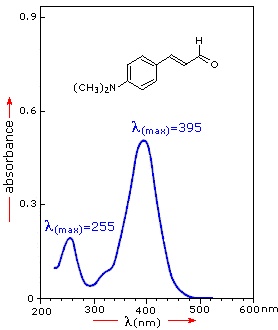
A representative UV–Vis absorption spectrum showing absorbance as a function of wavelength, with the peak maximum labeled as . This visual reinforces that spectra are read by locating peak positions (transition energies) and comparing relative intensities (how probable/strong a transition is). Source
Peaks occur where electronic transitions are most probable.
Visible colour is tied to selective absorption:
If a substance absorbs mainly in the visible region, the transmitted or reflected light is missing those wavelengths.
The observed colour is typically the complement of the absorbed colour (because the remaining light reaches your eye).
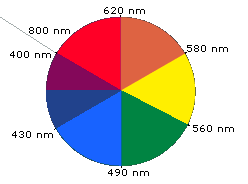
A complementary-color wheel that pairs absorbed wavelengths with the perceived (transmitted/reflected) color. This makes the ‘observed color is complementary to absorbed color’ rule concrete and helps you predict appearance from an absorption band in the visible region. Source
If absorption occurs only in the UV, the substance may appear colourless to humans.
The breadth of UV–Vis absorption peaks (rather than razor-thin lines) is typical for molecules because electronic transitions are accompanied by many closely spaced vibrational/rotational possibilities.
Emission Phenomena (Fluorescence and Related Effects)
After absorption, an excited species can lose energy by:
Non-radiative relaxation: energy transferred into molecular motion or collisions (no light produced).
Radiative relaxation (emission): a photon is released as the electron returns to a lower electronic energy level.
Because some energy may be lost before emission, emitted light is often lower energy (longer wavelength) than the absorbed light.
What Shifts UV–Vis Absorption to Longer or Shorter Wavelengths
Anything that changes the spacing between electronic energy levels can shift absorption.
More conjugation (alternating multiple bonds) typically decreases the energy gap, shifting absorption toward longer wavelengths (sometimes into the visible).
Different substituents can donate or withdraw electron density, altering orbital energies and shifting peak positions.
Solvent and environment can stabilize certain electronic states more than others, changing the transition energy and shifting the spectrum.
FAQ
Selection rules (based on symmetry and changes in electron distribution) affect transition probability.
Allowed transitions give intense bands
Forbidden transitions can still appear weakly due to vibrations/solvent effects
Electronic excitation occurs alongside many closely spaced vibrational states.
This produces a cluster of transitions that merge into a broad band, especially in liquids where collisions further blur energies.
Conjugation delocalises electrons, reducing the energy gap between relevant orbitals.
A smaller gap means lower-energy photons are absorbed, corresponding to longer wavelengths that can fall in the visible range.
Solvents stabilise ground and excited states differently (polarity, specific interactions).
If the excited state is stabilised more, the energy gap decreases and $\lambda_{\max}$ shifts to longer wavelength; the reverse gives a shorter-wavelength shift.
Fluorescence is fast emission after excitation; phosphorescence is delayed.
The delay typically arises from a change to a different type of excited state that returns to the ground state more slowly.
Practice Questions
(2 marks) Explain why UV–Vis absorption occurs only at certain wavelengths for a given molecule.
Electronic energy levels are quantised/discrete. (1)
Absorption occurs when photon energy matches the gap between two electronic energy levels (electronic transition). (1)
(5 marks) A dye appears blue in white light. Use ideas about UV–Vis electronic transitions to explain this observation and what it implies about the dye’s absorption.
The dye absorbs light via electronic transitions between electronic energy levels. (1)
Absorption must occur in the visible region (not only UV) for a coloured appearance. (1)
The absorbed wavelengths are not predominantly blue; blue light is transmitted/reflected to the eye. (1)
The observed blue corresponds to the complementary relationship between absorbed and observed colours. (1)
The absorption peak(s) correspond to photon energies matching the relevant electronic energy gap(s). (1)

