AP Syllabus focus: ‘Particulate-level representations that show multiple interacting species help communicate how intermolecular interactions establish macroscopic properties of a substance.’
Particulate representations translate invisible particle-scale attractions into visible models. In AP Chemistry, you must interpret and critique these diagrams to justify macroscopic properties using evidence about what particles are present and how they interact.
What particulate representations are showing
A useful particulate diagram does more than “draw dots.” It identifies what the particles are (atoms, molecules, ions) and communicates how strongly and in what way they attract.
Particulate-level representation. A drawing or model that depicts particles (and often their spacing, orientation, and interactions) to explain or predict macroscopic properties.
Between two correct diagrams, the better one makes the interaction story clearer: which attractions dominate, where contact occurs, and how many interacting neighbors each particle has.
Features that communicate intermolecular interactions
Particle identity and composition
Use different shapes/colours/sizes for different species to show multiple interacting species (e.g., solute and solvent, or two molecular types).
For molecules, show key structural features that control attractions (overall shape, polar regions, sites of high electron density).
Spacing, packing, and contact
Shorter average distances between particles typically indicate stronger attractions and/or more efficient packing.
Dense, regular packing suggests many simultaneous attractions per particle, which often correlates with “stronger” macroscopic behavior (less compressible, less volatile).
Diagrams should distinguish touching/nearby particles (meaningful attractions) from far-apart ones (weak net attraction).
Orientation (especially for polar species)
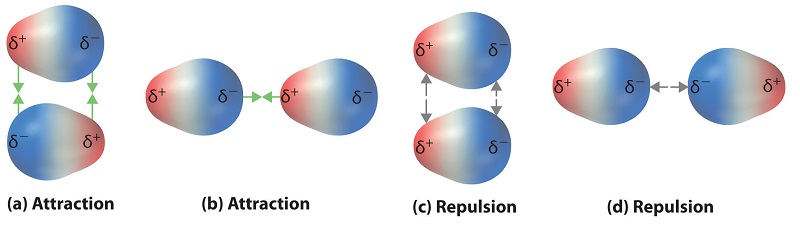
Dipole–dipole interactions depend strongly on molecular orientation. This diagram contrasts attractive alignments (opposite partial charges adjacent) with repulsive alignments (like partial charges adjacent), making the “interaction story” in a particulate representation unambiguous. Source
For polar particles, relative orientation matters: the model should show alignment of opposite partial charges when attractions are important.
Random orientations can communicate weaker directional effects or thermal disorder; consistent alignment can communicate stronger directional attractions.
Explicit interaction cues
Dashed lines, arrows, or labels can indicate attractive interactions; these should connect the correct regions (e.g., positive end to negative end).

Hexagonal ice (ice Ih) can be represented as an extended network where dashed lines mark hydrogen bonds between water molecules. The diagram emphasizes that each molecule participates in multiple directional attractions, helping explain why hydrogen bonding can produce rigid structures and unusual macroscopic behavior compared with substances dominated by weaker forces. Source
For ionic species, include full charges and show that attractions occur between opposite charges; for polar molecules, indicate partial charges where relevant.
Linking particle diagrams to macroscopic properties
The AP skill is to use what the diagram shows—particle type, spacing, and interaction patterns—to justify an observed property.
Volatility and phase-change tendencies (qualitative)
If the diagram shows particles held close with many attractions, it supports lower tendency to escape into the gas phase (lower volatility).
If particles are farther apart with fewer/less effective attractions, it supports easier separation (higher volatility).
Mechanical behavior (qualitative)
A diagram suggesting a rigid, repeating arrangement with many simultaneous attractions supports greater rigidity.
A diagram suggesting limited attractions between discrete units supports softer or more easily disrupted behavior.
Mixtures: “multiple interacting species”
When more than one species is present, the representation should allow comparison of:
A–A, B–B, and A–B attractions (which are strongest and most frequent).
Whether particles are uniformly interspersed (consistent with homogeneous behavior) or clustered/segregated (consistent with limited compatibility).
Common expectations and pitfalls on AP-style items
What earns credit
Stating a macroscopic claim (e.g., “higher boiling point,” “less volatile,” “more rigid”) and tying it to a specific particulate feature (distance, number of contacts, orientation, charge pattern).
Using comparative language grounded in the model: “more/less packed,” “more/less aligned,” “greater/fewer attractions per particle.”
Frequent errors
Treating bigger particles as automatically “stronger” without showing why (more contacts, closer approach, stronger attraction regions).
Drawing multiple species but not indicating cross-interactions (A–B), which is often the main point of the representation.
Using particle spacing inconsistently (e.g., claiming strong attractions while drawing large separations).
FAQ
By combining cues: shorter distances, more interaction lines per particle, consistent alignment of attractive regions, and greater local coordination (more neighbours in contact).
Two distinguishable particle types plus evidence of interaction between them, such as proximity patterns, explicit A–B dashed lines, or orientation showing attractive regions facing.
Some attractions are directional. If orientation is shown (or not shown), it signals whether attractions depend on alignment or are largely non-directional in the model.
Point out a mismatch between the claim and the diagram feature (e.g., “strong attractions” but large separations), or missing evidence (species not distinguished; no A–B interactions shown).
Yes. For example, low volatility could be supported by closer packing, more frequent attractions, or stronger cross-interactions in a mixture—provided the diagram clearly encodes that evidence.
Practice Questions
Question 1 (3 marks) A student draws two particulate-level representations of pure substances X and Y. In X, particles are closely packed with many dashed lines between neighbours. In Y, particles are similarly sized but more widely spaced with few dashed lines. Using the representations, state which substance is more volatile and give one particulate-level reason.
Correct choice: Y is more volatile (1)
Reason links fewer/weaker attractions to easier particle separation/escape (1)
Uses a diagram feature explicitly (e.g., wider spacing or fewer dashed lines meaning fewer interactions per particle) (1)
Question 2 (6 marks) A mixture contains species A and B. A particulate diagram shows A and B uniformly interspersed. The diagram labels strong dashed lines between A–B contacts, but few dashed lines between A–A or B–B contacts. (a) Explain how the diagram communicates the presence of multiple interacting species. (2 marks) (b) Use the diagram to justify one macroscopic property consistent with this mixture. (4 marks)
Identifies A and B as different particle types in the diagram (1)
Notes uniform interspersion and/or explicit A–B contacts indicating interactions between different species (1) (b)
States a reasonable macroscopic property claim tied to interactions (e.g., components mix well/homogeneous behaviour; reduced tendency to separate) (1)
Justifies using strong A–B attractions as the key evidence (1)
Compares to weaker A–A and B–B attractions to support the claim (1)
Cites a specific diagram feature (e.g., many A–B dashed lines and uniform distribution) (1)

