AP Syllabus focus: ‘Molecular solids consist of discrete covalently bonded molecules held together by relatively weak intermolecular forces; they typically have low melting points and do not conduct electricity.’
Molecular solids are common in everyday materials, from dry ice to sugar crystals. Their macroscopic properties follow directly from how intact molecules pack together and how weakly those molecules attract one another.
What a molecular solid is
Molecular solids are built from individual neutral molecules.
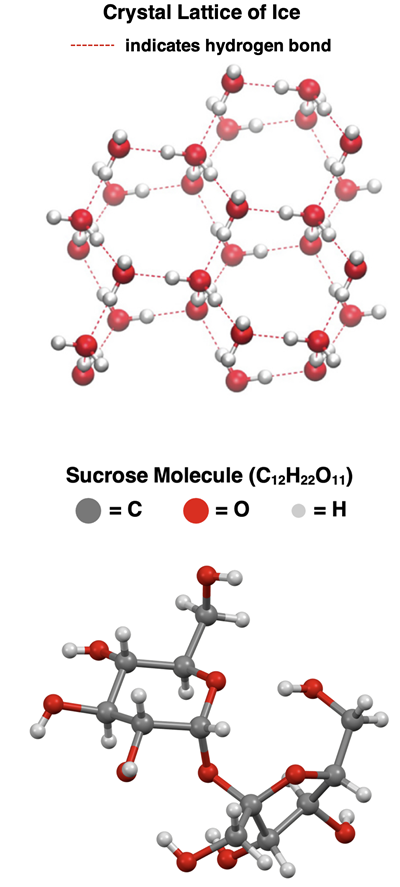
Crystal lattice model of ice showing discrete molecules connected by a network of hydrogen bonds (dashed lines). This visual reinforces that the crystal is maintained by intermolecular attractions between whole molecules rather than by ionic bonding or a covalent network extending through the solid. Source
Within each molecule, atoms are held together by covalent bonds, but different molecules are held together only by intermolecular forces (IMFs).
Molecular solid: a solid composed of discrete, covalently bonded molecules whose crystal lattice is maintained by intermolecular attractions rather than covalent or ionic bonds between particles.
Because the lattice is supported by relatively weak attractions, the solid’s bulk properties are dominated by how easily molecules can be separated from one another compared with breaking covalent bonds inside a molecule.
Intermolecular attractions in molecular solids
Types of intermolecular forces present
The “glue” between molecules can include:
London dispersion forces (LDF): present between all molecules; often the dominant attraction in nonpolar molecular solids.
Dipole–dipole attractions: added attraction in polar molecular solids.
Hydrogen bonding: a particularly strong dipole-based attraction when H is bonded to N, O, or F and interacts with a lone pair on N, O, or F in a neighboring molecule.
The key idea for solids is that these forces act between whole molecules, so changing the molecule’s size, shape, and polarity changes packing efficiency and the strength/number of contacts.
How packing affects strength
Even with the same kinds of IMFs, lattice stability depends on how well molecules pack:
More surface contact between molecules generally increases LDF and stabilises the solid.
Compact, symmetrical molecules often pack efficiently and can have higher melting points than similarly sized, less symmetrical molecules.
Flexible or irregular shapes may pack less efficiently, reducing lattice stability and lowering melting point.
Characteristic properties of molecular solids
Melting point (typically low)
The syllabus emphasis is that molecular solids are held together by relatively weak intermolecular forces, so they typically have low melting points. Melting requires disrupting intermolecular attractions while leaving covalent bonds within each molecule intact. As a result:
Many molecular solids melt at comparatively low temperatures (some even sublime readily).
Stronger IMFs (especially hydrogen bonding) and better packing can raise melting points, but they still reflect IMF strength, not covalent bond breaking.
Electrical conductivity (do not conduct)
Molecular solids do not conduct electricity because they lack mobile charge carriers:
Molecules in the lattice are neutral overall.
There are no free ions and no delocalised electrons able to move through the solid.
Even if a molecule contains polar bonds, the charges are partial and are not free to migrate across the crystal.
This is why molecular solids are generally classified as insulators in the solid state.
Mechanical behavior and volatility
Because intermolecular attractions are weaker than bonding in many other solid types, molecular solids are often:
Soft and easily deformed (molecules can shift when forces between them are overcome).
More likely to be brittle rather than malleable (slippage can disrupt the lattice without a mechanism for sustained deformation).
More volatile (higher tendency to evaporate or sublime) when IMFs are especially weak.
Examples and what to notice
Typical molecular solids include:
Dry ice, CO₂(s): nonpolar molecules; lattice dominated by LDF, leading to ready sublimation.
Iodine, I₂(s): nonpolar but large and highly polarisable; stronger LDF makes it a solid at room temperature.
Sucrose (table sugar): large polar molecule; multiple IMFs contribute, giving a crystalline solid that still remains nonconducting.
In all cases, the solid consists of intact molecules arranged in a repeating pattern; macroscopic differences trace back to IMF type/strength and packing efficiency.
FAQ
Many are crystalline because repeated packing minimises energy, but some can be poorly crystalline or glassy if molecules are irregular or the solid forms rapidly.
Disorder reduces the sharpness of melting behaviour and can broaden the melting range.
Even with only LDF, melting points can differ because LDF strength scales with polarisability and contact area.
Larger electron clouds → stronger instantaneous dipoles
Better packing/surface contact → stronger net attractions
Softness reflects how easily molecules can be displaced when stress is applied.
If intermolecular attractions are weak and directional locking is minimal, layers or groups of molecules can shift without breaking covalent bonds.
In general, no, but special cases exist if the solid contains a significant population of mobile charged species created internally (for example, through partial ionisation) or if it is heavily doped with ionic impurities.
These are exceptions rather than typical AP-level expectations.
Sublimation is favoured when the solid-to-gas transition does not require breaking strong attractions.
Molecular solids with weak IMFs can go directly to gas because separating molecules completely is not energetically prohibitive compared with substances held by much stronger particle-level attractions.
Practice Questions
Q1 (2 marks) A student claims that molecular solids conduct electricity when melted because the molecules can move. State whether the claim is correct and give one reason.
Claim is incorrect (1)
Reason: even when melted, molecular substances have neutral molecules and no mobile ions/delocalised electrons to carry charge (1)
Q2 (5 marks) A molecular solid X has a much lower melting point than molecular solid Y. Both solids are made of discrete covalently bonded molecules. (a) Explain, in terms of particle-level attractions, why X can have a lower melting point than Y. (3 marks) (b) State one reason molecular solids typically do not conduct electricity in the solid state. (2 marks)
Melting requires overcoming intermolecular forces between molecules, not breaking covalent bonds within molecules (1)
X has weaker/more easily disrupted intermolecular attractions than Y (1)
Acceptable elaboration: fewer/less effective intermolecular contacts due to smaller size, lower polarisability, lower polarity, lack of hydrogen bonding, or poorer packing efficiency (1) (b)
Particles are neutral molecules overall (1)
No mobile charge carriers (no free ions and no delocalised electrons) in the solid lattice (1)

