AP Syllabus focus: ‘Vapor pressure and boiling point depend directly on the strength of intermolecular forces because interactions are completely overcome during vaporization; melting point also correlates with interaction strength.’
Intermolecular forces control how easily particles separate during phase changes.
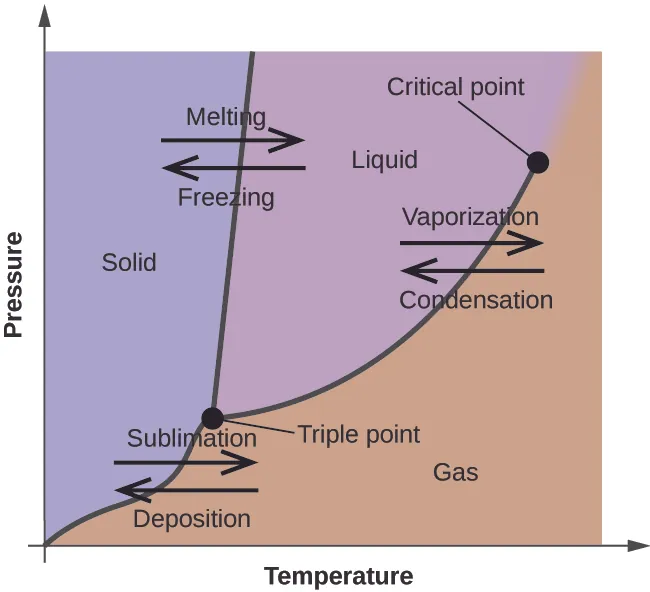
A typical phase diagram (pressure vs. temperature) maps where solid, liquid, and gas are stable and labels the phase boundaries for melting/freezing, vaporization/condensation, and sublimation/deposition. Because the liquid–gas boundary is the vapor-pressure curve, the diagram visually connects boiling to the condition where the liquid’s vapor pressure matches the external pressure. The triple point and critical point reinforce that phase behavior depends on both temperature and pressure, not temperature alone. Source
By connecting particle-level attractions to vapor pressure, boiling point, and melting point, you can predict and explain measurable trends across substances.
Core idea: phase change requires overcoming attractions
What changes during vaporization and melting
During vaporization (liquid → gas), particles must separate widely, so intermolecular forces (IMFs) must be completely overcome.
During melting (solid → liquid), particles remain close, but must gain enough freedom to move past one another, so attractions are weakened/disrupted rather than fully eliminated.
Strength of IMFs (qualitative)
Stronger IMFs mean particles attract more strongly, making it harder to separate them and requiring higher temperatures to change phase.
Vapor pressure and intermolecular forces
Vapor pressure reflects how readily particles escape from the liquid into the gas phase at a given temperature.
Vapor pressure: the pressure exerted by a vapor in equilibrium with its liquid (or solid) at a given temperature.
IMF strength vs vapor pressure (at the same temperature)
Stronger IMFs → lower vapor pressure
Fewer particles have enough energy to escape the liquid surface.
The vapor contains fewer particles, so equilibrium pressure is smaller.
Weaker IMFs → higher vapor pressure
More particles can separate from neighbors and enter the gas phase.
Boiling point and intermolecular forces
Why boiling point depends directly on IMFs
At the boiling point, the liquid’s vapor pressure equals the external pressure. Reaching that condition requires enough thermal energy for many particles to overcome liquid-phase attractions.
Stronger IMFs → higher boiling point
More energy (higher temperature) is required to fully overcome attractions during vaporization.
Weaker IMFs → lower boiling point
Less energy is needed for particles to escape into the gas phase.
Using boiling point and vapor pressure together
At a fixed temperature:
A substance with higher vapor pressure will typically have a lower boiling point (it reaches the needed vapor pressure at a lower temperature).
A substance with lower vapor pressure will typically have a higher boiling point.
Melting point and intermolecular forces
General correlation (with an important caution)
Melting point often increases as attractions between particles increase, because stronger attractions resist the rearrangement needed to transition from a rigid solid to a flowing liquid.
Stronger interparticle attractions → higher melting point (often)
However, melting point is also sensitive to how particles arrange in the solid, so the relationship is best treated as a correlation, not a perfect rule.
What “correlates” means here
Vapor pressure and boiling point depend directly on IMF strength because vaporization requires attractions to be completely overcome.
Melting point also correlates with interaction strength, but structural factors in solids can affect how strongly that trend appears.
FAQ
Boiling requires separating particles into a gas, so attractions must be fully overcome, giving a clearer trend.
Melting depends on how easily the solid structure can be disrupted; different solids can resist rearrangement differently even with similar attractions.
Branching usually reduces surface contact between molecules.
Less surface contact reduces the total attractive interactions between neighbouring molecules, often lowering the boiling point compared with a less-branched isomer.
Boiling occurs when vapour pressure equals external pressure.
Higher external pressure requires a higher temperature to reach that vapour pressure; lower external pressure reduces the boiling point.
Often, yes, because more molecules are able to enter the gas phase at equilibrium.
However, the observed rate can also depend on airflow, surface area, and whether the surrounding air is already saturated with vapour.
A larger enthalpy of vaporisation indicates more energy is required to separate particles into the gas phase.
Substances with larger enthalpies of vaporisation typically have lower vapour pressures at the same temperature and higher boiling points.
Practice Questions
Q1 (2 marks) Explain, in terms of intermolecular forces, why a liquid with stronger intermolecular forces has a lower vapour pressure at the same temperature.
Stronger intermolecular attractions hold particles in the liquid more strongly, so fewer particles escape into the vapour (1).
Fewer gas-phase particles at equilibrium gives a lower equilibrium pressure / vapour pressure (1).
Q2 (5 marks) Two pure liquids, A and B, are at the same external pressure. Liquid A has stronger intermolecular forces than liquid B. Compare A and B in terms of (i) boiling point and (ii) vapour pressure at the same temperature, and justify each comparison using particle-level reasoning.
A has a higher boiling point than B (1).
Justification: stronger forces in A require a higher temperature to overcome attractions during vaporisation / to reach vapour pressure equal to external pressure (1).
At the same temperature, A has a lower vapour pressure than B (1).
Justification: fewer A particles escape to the gas phase because attractions are stronger (1).
Links to equilibrium pressure depending on number of vapour particles / collision frequency (1).

