AP Syllabus focus: ‘Strong ion–ion interactions give ionic solids low vapor pressure and high melting and boiling points; they are brittle and conduct electricity only when ions are mobile (molten or dissolved).’
Ionic solids are held together by strong electrostatic attractions that strongly influence phase-change behavior and mechanical and electrical properties. Understanding these properties requires linking macroscopic observations to what ions can and cannot do inside a crystal lattice.
What makes an ionic solid “ionic”
Ionic solids form when cations and anions arrange into an extended, repeating structure that maximizes attractions and minimizes repulsions.
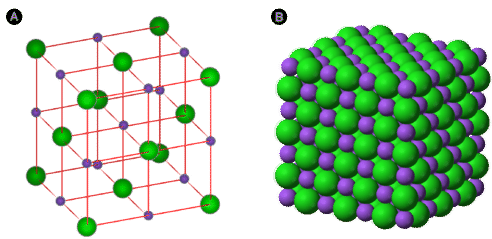
Two complementary models of the NaCl ionic lattice are shown: an expanded model that makes the alternating ion pattern and coordination easier to see, and a space-filling model that better represents how ions pack in a real crystal. The color-coding distinguishes cations from anions, reinforcing that the “particles” in an ionic solid are ions in a continuous 3D array rather than discrete molecules. Source
Ionic solid: A crystalline solid made of alternating cations and anions in a 3D lattice, held together primarily by Coulombic (electrostatic) attraction.
Because there are no discrete molecules to “separate,” many properties depend on disrupting the entire lattice rather than just weakening a few intermolecular attractions.
Strength of ion–ion attraction (what controls it)
The strength of attraction between ions increases with:
Greater ionic charge magnitude (e.g., with stronger than with )
Smaller ionic size (shorter ion–ion distance)
Lattice energy: The energy change associated with forming an ionic solid from gaseous ions (released on formation; required in magnitude to separate the solid into gaseous ions). Larger magnitude means a more strongly held lattice.
A larger-magnitude lattice energy generally corresponds to higher melting/boiling points and lower volatility.
= relative electrostatic potential energy (proportional to J)
= ionic charges (in units of the elementary charge or C)
= distance between ion centres (m)
This proportionality explains why ionic solids built from small, highly charged ions tend to be especially “high-melting.”
Melting point, boiling point, and vapor pressure
Why melting and boiling points are high
To melt an ionic solid, enough energy must be supplied to partially overcome the ion–ion attractions that lock ions into fixed lattice positions. To boil it, those attractions must be overcome to a much greater extent, separating ions into the gas phase (often requiring extremely high temperatures and sometimes accompanied by decomposition).
Key macroscopic results:
High melting points: significant energy needed before ions can move past one another
High boiling points (when boiling is feasible): separating ions completely is energetically costly
Trends track attraction strength: higher charges and smaller ions typically increase these temperatures
Why vapor pressure is low
Vapor pressure reflects how easily particles escape into the gas phase. In ionic solids, ions are strongly stabilized in the lattice, so very few particles have enough energy to escape under ordinary conditions.
Stronger ion–ion attractions ⟶ lower vapor pressure
Low vapor pressure aligns with the idea that ionic solids are generally nonvolatile
Brittleness (why ionic crystals shatter)
Ionic solids are typically brittle rather than malleable. Under stress (e.g., a hammer blow), lattice layers may be forced to shift slightly. If that shift brings like-charged ions closer together:
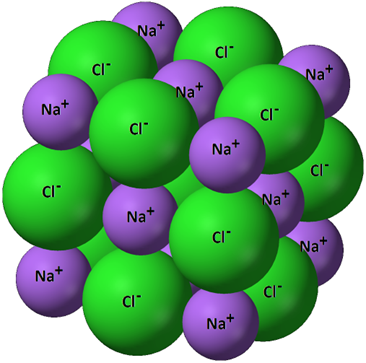
A schematic of the NaCl lattice highlights that small displacements can convert locally favorable cation–anion attractions into unfavorable like-charge neighbors. The diagram makes the brittleness mechanism concrete: once like charges align, electrostatic repulsion rises sharply and the crystal fractures rather than plastically deforming. Source
cation–cation and/or anion–anion repulsions increase sharply
the local repulsion can exceed the lattice’s ability to rearrange
the crystal cleaves along planes and fractures
This brittleness is a direct consequence of an ordered lattice of alternating charges: small displacements can create strongly unfavorable like-charge contacts.
Electrical conductivity (when ions are mobile)
Ionic substances conduct electricity only when they contain mobile charged particles.
Solid ionic compounds: do not conduct
In the solid state:
ions are fixed in lattice positions (they vibrate but do not translate)
charges cannot flow through the crystal
result: poor electrical conductivity
Molten or dissolved ionic compounds: conduct
When ionic solids are molten or dissolved in water, ions become free to move:
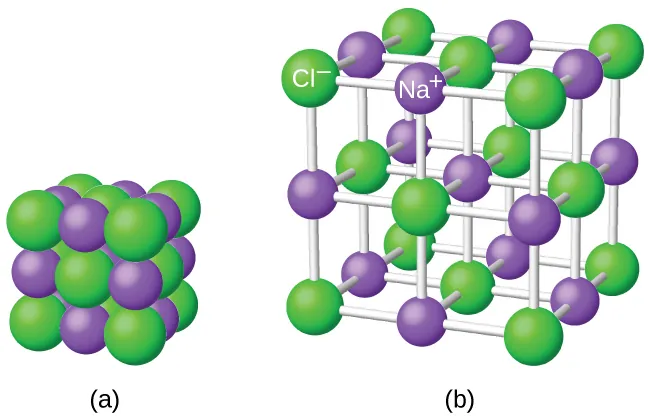
The NaCl lattice is depicted as a regular 3D arrangement of alternating ions, with an expanded view that makes the geometry and nearest-neighbor relationships more visible. This supports the conductivity idea by emphasizing that in the solid, ions occupy fixed lattice positions; electrical conduction requires disrupting that rigid arrangement so ions can migrate. Source
molten ionic liquid: ions move within the liquid, carrying charge
aqueous solution: ions are separated and stabilized by water, allowing migration toward electrodes
result: good electrical conductivity (an electrolyte)
Conductivity depends qualitatively on how many ions are present and how easily they move, but the core requirement is the same: ionic mobility.
FAQ
Yes, a few show measurable solid-state conductivity due to defects or unusual structures.
Defect ions (vacancies/interstitials) can hop between sites.
Some “fast ion conductors” allow one ion type to move through channels.
This is not typical for most classroom ionic solids.
At very high temperatures, ions (or polyatomic ions) may undergo chemical change before enough intact formula units can enter the gas phase.
This can involve bond breaking within polyatomic ions or reactions with the environment, so “boiling point” may not be practically observable.
Imperfections such as grain boundaries and microcracks concentrate stress.
Smaller crystals can sometimes appear less prone to catastrophic cracking because cracks have shorter paths, but the underlying brittleness mechanism (like-charge repulsion after lattice shift) still applies.
Water molecules stabilise separated ions via strong ion–dipole interactions.
This hydration lowers the energetic penalty for separating ions and prevents immediate recombination, producing dispersed, mobile charge carriers throughout the solution.
Yes. Lower melting can occur with:
larger ions (greater $r$)
lower charges
lattices that pack less efficiently
The bonding is still predominantly ionic; the lattice is simply easier to disrupt.
Practice Questions
(2 marks) Explain why solid sodium chloride does not conduct electricity but molten sodium chloride does.
Solid: ions fixed in lattice / not mobile (1)
Molten: ions free to move and carry charge (1)
(5 marks) An ionic solid X has a much higher melting point than ionic solid Y. Suggest two particle-level reasons for this difference, and explain why both X and Y are brittle and have low vapour pressure.
Reason 1 linked to stronger attraction: higher ionic charges in X (1) with explanation of stronger Coulombic attraction / larger lattice energy (1)
Reason 2 linked to stronger attraction: smaller ions/shorter interionic distance in X (1) with explanation of stronger attraction / larger lattice energy (1)
Brittleness: stress shifts layers bringing like charges adjacent causing repulsion and fracture (1)
Low vapour pressure: ions strongly held; few particles escape to gas (1)
(Max 5: award any 5 correct marking points)

