AP Syllabus focus: ‘Acid–base reactions involve transfer of one or more protons (H⁺) between chemical species.’
Acid–base reactions are identified by tracking where a proton goes during a reaction. In AP Chemistry, the key skill is recognising proton transfer in symbolic equations, names, and particulate-level descriptions.
What makes a reaction “acid–base” in AP Chemistry?
Acid–base reaction: A chemical reaction in which one species transfers one or more protons (H+) to another species.
To identify an acid–base reaction, look for evidence that an H+ has moved from one reactant to another.
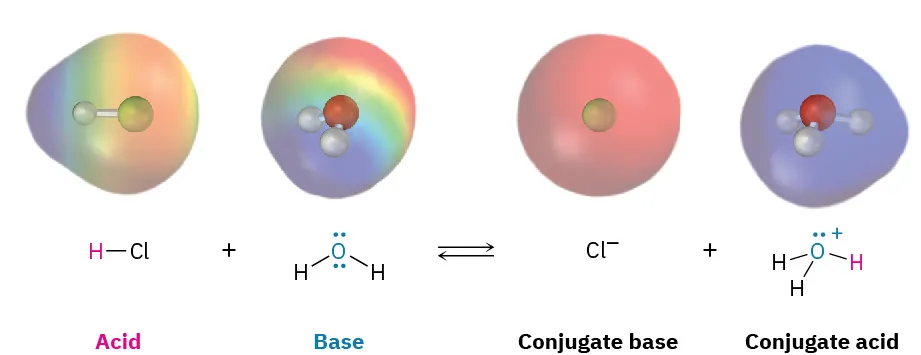
Molecular-level depiction of proton transfer when hydrochloric acid reacts with water, producing chloride and hydronium. By labeling the base and conjugate species, the image connects the symbolic idea of transfer to particle-level changes in bonding and charge. Source
This is a Brønsted–Lowry view of acid–base chemistry, emphasised for aqueous reactions.
Brønsted–Lowry species in proton transfer
Brønsted–Lowry acid: A species that donates a proton (H+).
Brønsted–Lowry base: A species that accepts a proton (H+).
A proton donor becomes “less protonated” after the reaction, and a proton acceptor becomes “more protonated.” You do not need to label conjugate pairs on this page; focus on spotting the transfer.
Visual and symbolic cues for proton transfer
Common patterns that signal an acid–base reaction
Appearance of H+ on one side only (often in net ionic form).
Formation of water from H+ and OH− (neutralisation).
A reactant with H (often written as an acid) produces a product missing that H.
A reactant gains an H in the products (often written as a base becoming protonated).
Recognising proton donors and acceptors by formulas
Many acids are written as molecules beginning with H (for example, formulas of the form HX or H2X), but identification should be based on H+ transfer, not memorisation.
Many bases are anions or molecules with lone pairs that can bond to H+; in aqueous chemistry, OH− is a very common base.
The generic model for identification
= acid (proton donor)
= base (proton acceptor)
= species after losing H+ (deprotonated form)
= species after gaining H+ (protonated form)
When you compare both sides, the key check is that the number of H atoms changes in the two reacting species in a way consistent with transfer, while other atoms remain associated with their original frameworks.
Distinguishing acid–base from non-acid–base reactions
What is not enough by itself
Any reaction in water is not automatically acid–base.
Dissolution (even if ions form) is not necessarily acid–base unless H+ is transferred.
Precipitation and many redox reactions do not involve H+ transfer, even if charges change or new solids form.
Practical checklist (equation-based)
Identify the species most plausibly containing a transferable H (often written with H in its formula).
Identify a plausible proton acceptor (often an anion, OH−, or a neutral species that can become positively charged).
Confirm that one product has one more H (or more, for multiple transfers) and the other has one fewer H, consistent with conservation of atoms and charge.
“One or more protons”: multiple transfers
Some reactions transfer more than one proton overall:
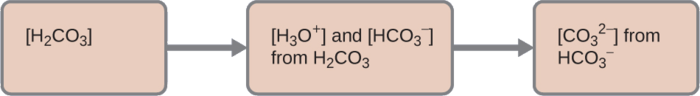
Stepwise ionization diagram for a diprotic acid system (carbonic acid), showing sequential formation of new conjugate pairs after each proton transfer. The figure highlights how the acid becomes progressively less protonated while increases with each step, illustrating “multiple transfers” as repeated single-proton events. Source
Polyprotic acids can donate H+ in steps.
A base may accept more than one proton if multiple basic sites exist. For identification, it is sufficient to show that proton transfer occurs at least once; multiple H+ transfers still classify the reaction as acid–base.
FAQ
Look for one species that loses an H atom and another that gains an H atom between reactants and products.
Check that the changes match conservation of atoms and charge.
Often, but not automatically.
Water formation suggests H+ and OH− combined, yet you should still verify a proton transfer pathway in the reaction as written.
Common acceptors include anions (negative charge) and neutral molecules with lone pairs.
In practice, confirm acceptance by seeing a product that is the same species with an added H and typically a higher positive charge.
Yes. Such species can either donate or accept H+ depending on the reaction partner.
Bicarbonate, HCO3−, is a common example in aqueous chemistry.
Charges can change due to electron transfer (redox) or ion pairing without proton movement.
Acid–base identification specifically requires tracking the movement of H+ between reactants and products.
Practice Questions
(2 marks) Consider the reaction: NH3(aq) + H2O(l) → NH4+(aq) + OH−(aq).
Explain why this reaction is an acid–base reaction.
States that a proton (H+) is transferred between species. (1)
Identifies H2O as proton donor and/or NH3 as proton acceptor, consistent with products. (1)
(5 marks) For the reaction: HCO3−(aq) + H2O(l) → CO3^2−(aq) + H3O+(aq)
(a) Identify the Brønsted–Lowry acid and Brønsted–Lowry base. (2)
(b) Justify your choices by describing the proton transfer and how formulas change. (3)
(a) HCO3− identified as the acid (donates H+). (1)
(a) H2O identified as the base (accepts H+). (1)
(b) Describes HCO3− losing H+ to become CO3^2−. (1)
(b) Describes H2O gaining H+ to become H3O+. (1)
(b) Links justification explicitly to proton transfer (not merely charge change). (1)

