AP Syllabus focus: ‘Redox reactions involve electron transfer shown by oxidation-number changes; combustion is a redox subclass where a species reacts with O₂ (hydrocarbons form CO₂ and H₂O).’
Redox and combustion reactions appear across many AP Chemistry contexts. Being able to quickly identify them from reactants and products helps you predict electron flow, common products, and reaction patterns.
What makes a reaction “redox”?
A reaction is oxidation–reduction (redox) if it involves electron transfer. In many chemical equations, electron transfer is tracked indirectly by changes in oxidation numbers.
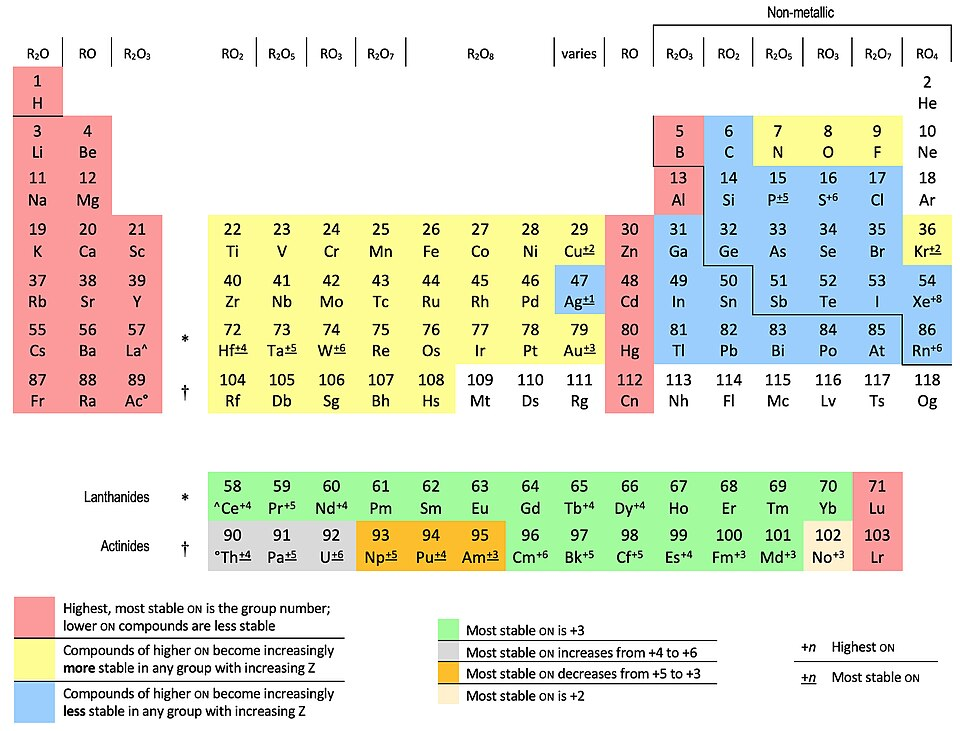
Periodic-table overview of common oxidation-number trends by element. This helps you quickly anticipate which elements commonly appear in multiple oxidation states (especially transition metals), making oxidation-number changes—and therefore redox behavior—easier to spot. Source
Redox reaction: A chemical reaction that involves electron transfer between species, evidenced by changes in oxidation numbers from reactants to products.
In a redox process:
At least one element’s oxidation number increases (loss of electron density).
At least one element’s oxidation number decreases (gain of electron density).
Both changes must occur in the same overall reaction (electrons are conserved overall).
Using oxidation-number changes to identify redox
To decide whether a reaction is redox, compare oxidation numbers of atoms in reactants vs products.
If no oxidation numbers change, it is not a redox reaction.
If any oxidation numbers change, it is a redox reaction (because oxidation-number changes are used as evidence of electron transfer).
Oxidation number: A bookkeeping charge assigned to an atom in a substance that helps track electron transfer; redox is identified when these assigned values change during a reaction.
What makes a reaction “combustion”?
Combustion is a specific and very common subclass of redox.

Photograph of a match ignition used to illustrate combustion as an energy-releasing chemical reaction. This provides a concrete visual anchor for combustion as a redox process driven by reaction with , typically producing heat and light. Source
A combustion reaction occurs when a species reacts with and is oxidised by it.
Key identification features:
is a reactant.
The reaction is redox, with oxygen typically being reduced (oxidation number goes from 0 in to −2 in many products).
For hydrocarbons (compounds containing only C and H), the characteristic products of complete combustion are and .
Hydrocarbon combustion pattern (complete combustion)
The most recognisable combustion equations are hydrocarbons reacting with oxygen to form carbon dioxide and water.
= number of carbon atoms in the hydrocarbon formula (unitless)
= number of hydrogen atoms in the hydrocarbon formula (unitless)
This pattern is especially useful for identifying combustion even before balancing:
If reactants include hydrocarbon +
And products include and
Then the reaction is combustion and therefore redox.
Quick identification checklist (high-utility)
Spotting redox quickly
Look for elements in their free form (oxidation number 0) turning into compounds (oxidation number becomes nonzero), or the reverse.
Look for , halogens (like ), or metals forming ions—these often indicate redox.
Confirm by checking whether an element’s oxidation number changes between sides.
Spotting combustion quickly
Reactant side includes .
If fuel is a hydrocarbon, products are and (for complete combustion).
Because oxygen’s oxidation number changes from 0 in to typically −2 in products, combustion fits the criterion “electron transfer shown by oxidation-number changes.”
FAQ
Look for $O_2$ as a reactant and an oxide as a product (e.g., $SO_2$, $MgO$).
These are still combustion reactions in a broad sense, and they are still redox because oxygen is reduced from 0 to typically $-2$.
Oxidation numbers track electron density shifts using consistent rules.
When they change, it indicates electrons have been effectively transferred between atoms, matching the operational definition of redox used in chemistry.
No. $O_2$ may appear in reactions that are not typically classified as combustion (for example, some slow oxidation processes).
Combustion generally refers to a direct reaction with $O_2$ forming oxides, often with significant energy release.
Yes. Any reaction with oxidation-number changes is redox, even without $O_2$.
Common examples include metal displacement reactions and reactions involving halogens such as $Cl_2$.
Check the identity of the reactant: if it is not a hydrocarbon, the products will not necessarily be $CO_2$ and $H_2O$.
Classify by the key test: does a species react with $O_2$ to form oxides, with oxidation-number changes?
Practice Questions
Q1 (2 marks) Consider the reaction: .
State whether this is a redox reaction and justify your answer using oxidation numbers.
1 mark: Identifies reaction as redox.
1 mark: Justification: oxidation number changes and changes (oxidation numbers change/electron transfer implied).
Q2 (5 marks) Propane combusts in oxygen to form carbon dioxide and water.
(a) Write the chemical formula of propane. (1 mark)
(b) Identify this reaction as combustion and explain why combustion is a subclass of redox. (2 marks)
(c) State the oxidation number of oxygen in and in , and use this to support that the reaction is redox. (2 marks)
(a) 1 mark: .
(b) 1 mark: Combustion because a species reacts with and (for a hydrocarbon) forms and .
(b) 1 mark: It is redox because electron transfer is shown by oxidation-number changes.
(c) 1 mark: Oxygen is 0 in .
(c) 1 mark: Oxygen is in ; change indicates reduction of oxygen/oxidation-number change supports redox.

