AP Syllabus focus: ‘You are not assessed on the meanings of “oxidizing agent/reducing agent” or on solubility rules beyond those implied by common soluble salts.’
These notes clarify what the AP Chemistry exam does not require you to memorise in reaction classification. Knowing these boundaries helps you study efficiently and avoid spending time on low-yield memorisation.
What the exam explicitly does not require
Non-memorised redox labels
The AP exam does not assess you on memorising the meanings of the terms “oxidizing agent” and “reducing agent.” In practice, this means:
You should not devote study time to learning formal label-based definitions as standalone recall items.
If those terms appear in a prompt, they are typically used as descriptive language; scoring is tied to chemically supported reasoning (for example, identifying which species changes in a way consistent with electron transfer), not to reciting definitions.
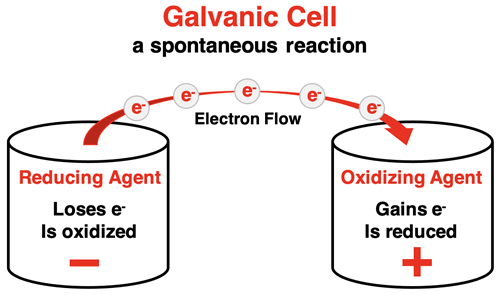
Schematic of a galvanic (voltaic) cell showing spatial separation of oxidation and reduction half-reactions. The labels indicate which electrode is the anode vs. cathode, the direction of electron flow through the wire, and compensating ion migration through the salt bridge to maintain charge balance. This supports evidence-based redox reasoning by focusing on what changes (electron transfer and species transformation) rather than vocabulary recall. Source
Practical implication for writing responses:
Prefer describing what happens (for example, which substance causes another to change) rather than relying on memorised labels.
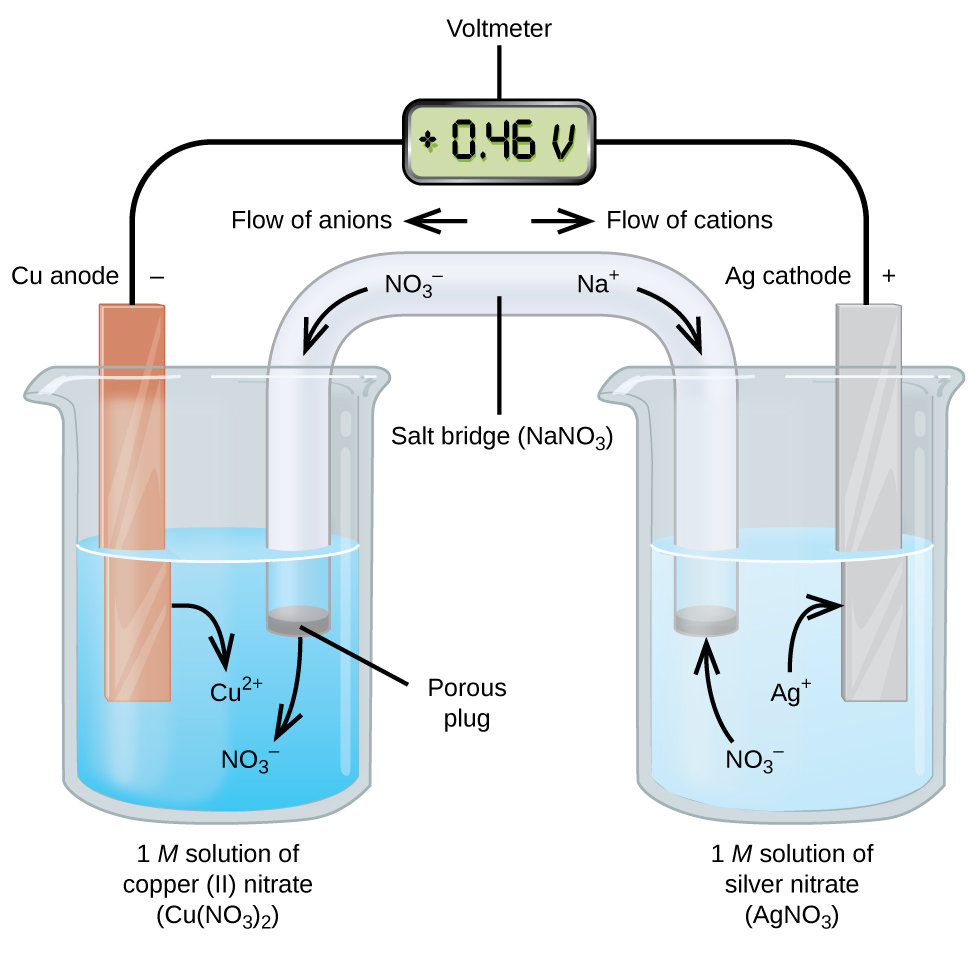
Diagram of a standard galvanic cell layout in which oxidation and reduction occur in separate half-cells connected by an external wire and an ion-conducting salt bridge. The figure highlights how electron flow in the circuit is coupled to ion movement in solution to preserve electrical neutrality. It provides a concrete visual reference for writing explanations that track matter and charge changes rather than relying on labels alone. Source
If you choose to use the terms, make sure the rest of your explanation is consistent with the reaction as written; the label alone is not the goal.
Limited solubility rules you’re expected to know
You are also not assessed on solubility rules beyond those implied by common soluble salts. The course expectation is limited to a small, high-frequency set:
Sodium salts () are soluble
Potassium salts () are soluble
Ammonium salts () are soluble
Nitrate salts () are soluble
Practical implication for precipitation questions:
You should be comfortable assuming compounds containing these ions will be aqueous in typical AP contexts.
You do not need to memorise an extended solubility chart (for example, detailed exceptions for halides or sulfates) unless your teacher or lab work uses one for local assessments.
How to apply this exam note to studying and test-taking
Study prioritisation (what to focus on instead of memorising extras)
To align with the exam note, keep your preparation targeted:
Learn to recognise the “always soluble” ions above quickly and confidently.
When a question requires solubility beyond that short list, expect one of the following:
The necessary information is provided (for example, via a table, a list of observations, or given phases)
The reaction is written in a way that makes the outcome evident (for example, a solid product is explicitly shown or implied by an observation)
Common exam-style cues that replace memorisation
AP questions often signal what you need without requiring a full rule set:
States of matter included in an equation (such as (aq) vs (s))
Observations (formation of a solid, cloudiness, or another stated result)
Given lists of soluble/insoluble compounds in the problem stem
Communication tips (earning credit without “forbidden memorisation”)
Use evidence-based wording: refer to what is shown (states, observations, or provided data).
Avoid overclaiming: don’t invent solubility “rules” you are unsure of; stick to the guaranteed soluble ions or information supplied in the question.
Treat oxidizing agent/reducing agent terminology as optional vocabulary, not required memorisation.
FAQ
It’s widely used scientific vocabulary, so textbooks include it for completeness and to support later chemistry courses.
AP items may still use the phrase, but marks are awarded for chemically consistent reasoning rather than reciting a definition.
It refers to ions that virtually always form soluble ionic compounds in typical aqueous contexts:
$\mathrm{Na^+}$, $\mathrm{K^+}$, $\mathrm{NH_4^+}$, $\mathrm{NO_3^-}$
You’re expected to treat these as reliable defaults unless a question explicitly indicates otherwise.
Look for information the question gives you (states, observations, or a provided list/table).
If none is provided, the item is usually designed so the “always soluble” ions are enough to decide, or solubility is not essential to earning full credit.
It can help for speed in classroom quizzes or labs, and for chemistry beyond AP.
For AP scoring, however, it’s typically low-yield compared with mastering how to use information presented in the prompt.
Not for using the words themselves, but you can lose marks if the label contradicts your stated chemical evidence.
If you choose to use them, ensure the rest of your explanation clearly supports the claim.
Practice Questions
(1–3 marks) The exam note states that you are not assessed on solubility rules beyond those implied by common soluble salts. Name two ions whose salts are expected to be soluble for AP Chemistry purposes. (2 marks)
1 mark for any correct ion: , , ,
1 mark for a second correct ion from the list
(4–6 marks) A student revises by memorising (i) detailed solubility exceptions and (ii) formal definitions of “oxidising agent” and “reducing agent”. Using the AP exam note, explain why this is inefficient and identify the limited solubility knowledge that is still expected. (5 marks)
1 mark: states that meanings of “oxidising agent/reducing agent” are not assessed as memorisation
1 mark: states that extended solubility rules/exceptions are not assessed as memorisation
1 mark: identifies that salts of , , and/or are expected soluble
1 mark: identifies that nitrates () are expected soluble
1 mark: explains that exam questions will typically provide needed information (e.g., states/observations/data) when solubility beyond this set is required

