AP Syllabus focus: ‘Precipitation reactions often mix aqueous ions to form an insoluble (or sparingly soluble) ionic compound; sodium, potassium, ammonium, and nitrate salts are soluble.’
Precipitation reactions are a major class of aqueous reactions where dissolved ions recombine to form a solid. Mastery depends on recognising which ionic compounds stay dissolved and which form an insoluble product.
What a precipitation reaction is
A precipitation reaction typically occurs when two aqueous ionic solutions are mixed and at least one possible product is insoluble in water, so it forms a solid that separates from solution.
Precipitation reaction: An aqueous reaction in which dissolved ions form an insoluble (or sparingly soluble) ionic compound, producing a solid precipitate.
The key skill is to decide, from the ions present, whether any insoluble ionic compound can form.
Core idea: ions swap partners, then solubility decides
When two ionic solutions mix, the cations and anions can recombine in new pairings. If a new pairing corresponds to an insoluble ionic compound, solid forms; if all potential products are soluble, no precipitate forms.
Solubility terms you must use correctly
Solubility language in AP Chemistry is qualitative for this topic: “soluble” means it remains as separated ions in water; “insoluble” means it forms a solid under typical aqueous conditions.
Spectator ions: Ions present in solution that do not become part of the precipitate and therefore remain aqueous before and after mixing.
A precipitate is written with the state symbol (s), while dissolved ions/ionic compounds are written as (aq).

Photograph of the bright yellow precipitate lead(II) iodide, PbI(s), forming when solutions containing Pb(aq) and I(aq) are mixed. This is a vivid example of how an insoluble ionic product separates from solution and is represented with the state symbol (s). Source
Key solubilities you are expected to know
AP Chemistry emphasises a small set of “always soluble” ionic compounds for identifying precipitation reactions.
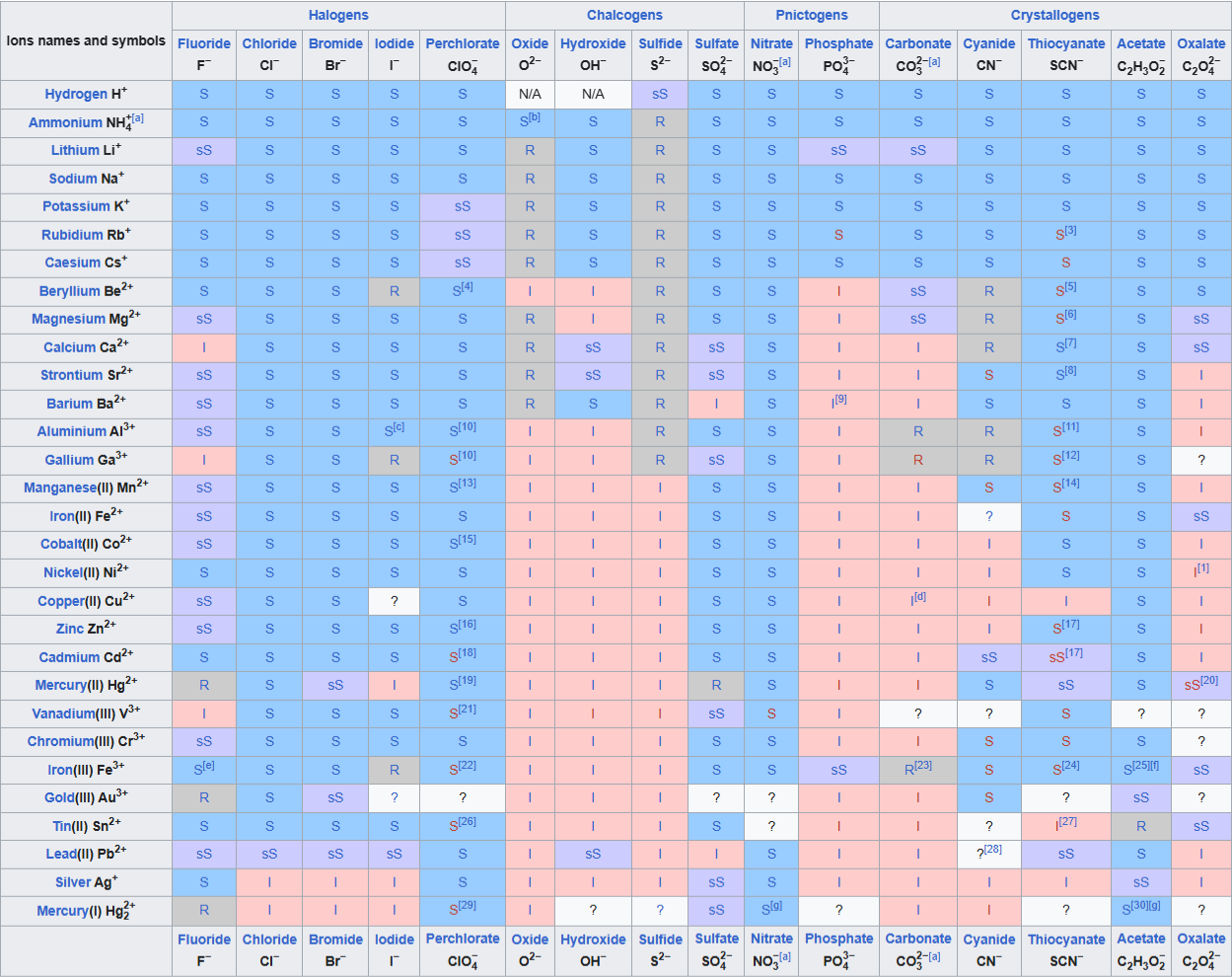
A solubility chart that summarizes which ionic compounds are soluble, slightly soluble, or insoluble based on cation–anion combinations. This visual helps you quickly predict whether a new ion pairing will remain aqueous or form a precipitate under typical conditions. Source
Always soluble (given by the syllabus emphasis)
Use these as quick filters: if an ionic compound contains any of the following, predict it is soluble in water (so it will not precipitate).
Group 1 cations: Na⁺ salts are soluble, K⁺ salts are soluble
Ammonium: NH₄⁺ salts are soluble
Nitrates: NO₃⁻ salts are soluble
This means:
If a potential product is a nitrate, it stays aqueous.
If a potential product contains Na⁺, K⁺, or NH₄⁺, it stays aqueous.
“Insoluble or sparingly soluble” as the driver for precipitation
Precipitation is predicted when an ion pairing forms an ionic compound that is insoluble (or sparingly soluble).
“Sparingly soluble” means a small amount may dissolve, but enough solid forms to be treated as a precipitate in typical lab mixing.
To stay aligned with the syllabus note, you are not expected to memorise a long solubility table here; instead, you use the always-soluble list to eliminate many possibilities, then recognise that a remaining ionic compound can be the precipitate.
How to identify whether a precipitate forms (process)
Use a consistent decision path when two solutions are mixed.
Step-by-step identification
List the ions present in each aqueous solution (separate the compound into its ions).
Consider the new cation–anion combinations that could form upon mixing.
Check each possible product against the always soluble list:
If it contains Na⁺, K⁺, NH₄⁺, or NO₃⁻, label it (aq).
If any remaining product is not flagged as always soluble, predict that it is the insoluble (or sparingly soluble) compound and will form a precipitate (s).
Identify spectator ions as those not in the solid product; they remain (aq).
Practical interpretation
In an actual mixture, the appearance of a solid indicates that the solution now contains an ion combination whose solubility has been exceeded; the solid is the precipitated ionic compound.
FAQ
“Insoluble” often means the dissolved concentration is extremely small. “Sparingly soluble” means some dissolves, but a visible solid still forms under typical mixing.
In quantitative terms, sparing solubility corresponds to a small $K_{sp}$ and a low equilibrium ion concentration.
Nitrates tend to form ions that are well-stabilised by water (strong hydration), making dissolution favourable.
AP-level usage: treat $NO_3^-$ as a reliable marker that an ionic compound remains $aq$.
Yes. Solubility can increase or decrease with temperature, so a compound that precipitates at one temperature might dissolve at another.
However, AP precipitation predictions typically assume standard laboratory temperatures unless told otherwise.
Yes, if conditions change (dilution, temperature) or if ions are removed from solution.
Another common cause is complex ion formation, where a dissolved ligand binds a metal ion and increases its solubility.
“No precipitate” means no solid forms; ions may still be present together in solution.
For precipitation identification, you are only deciding whether an insoluble ionic product forms, not whether ions have mixed or exchanged partners.
Practice Questions
Question 1 (1–3 marks) When aqueous solutions of and are mixed, does a precipitate form? State your reasoning using the key solubilities.
1 mark: States no precipitate forms.
1 mark: Uses nitrate solubility: all salts are soluble.
1 mark: Uses and/or solubility: their salts are soluble, so all possible products remain aqueous.
Question 2 (4–6 marks) Two colourless solutions are mixed in three separate beakers: A: with
B: with
C: with
For each beaker, state whether a precipitate forms and justify each decision using only the key solubilities listed in the syllabus focus.
1 mark each (3 marks): Correctly states no precipitate for A, B, and C.
1 mark: Justifies using nitrate solubility ( salts soluble) applied to at least one mixture.
1 mark: Justifies using Group 1 cation solubility (, salts soluble) applied to at least one mixture.
1 mark: Justifies using ammonium solubility ( salts soluble) applied to mixture B.

