AP Syllabus focus: ‘Reaction order can be inferred from how a reactant concentration changes with time, using concentration–time graphs.’
Reaction order is not read from the balanced equation; it is inferred from how concentration changes during a run. Concentration–time graphs let you diagnose order by recognising characteristic curve shapes and linearised plotting choices.
Using concentration–time data to infer reaction order
Core idea: pattern over time
A single experiment can suggest the likely reaction order by how a reactant’s concentration decreases as time passes. This approach uses the shape of a concentration–time curve and, more reliably, whether a transformed plot becomes a straight line.
Integrated rate law: A relationship between reactant concentration and time whose form depends on the reaction order; it is used to test which order best matches concentration–time data.
Always choose one reactant species (often labelled A) and track its concentration at multiple times under constant conditions (especially constant temperature).
What the raw [A] vs. time curve can tell you
When plotting vs. time, different orders show different visual behaviours:
Zero-order behaviour (diagnostic shape):
decreases by roughly equal amounts in equal time intervals
the curve looks close to a straight line (constant slope)
First-order behaviour (diagnostic shape):
decreases quickly at first, then more slowly
the curve is smoothly curved (slope magnitude shrinks over time)
Second-order behaviour (diagnostic shape):
drops very rapidly at early times when concentration is high
curvature is stronger than first order, with an especially pronounced early-time change
Because real data include noise and rounding, visual inspection of vs. time is best treated as a preliminary clue, not final proof.
Linearised plots: the standard graph-based test
The three “order tests”
To infer order more confidently, test which transformation gives the most linear graph (best straight line) against time:
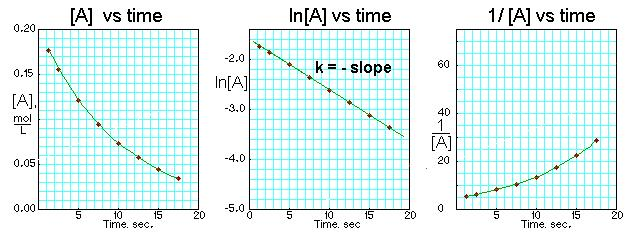
Three side-by-side kinetic plots for the same dataset illustrate the integrated-rate-law test for reaction order. The vs. time plot is linear (with slope ), indicating first-order behavior in , while vs. time and vs. time remain curved. This visual comparison captures the core idea that the “most linear” transformation identifies the order. Source
Plot vs. time
Plot vs. time
Plot vs. time
The order is inferred from which plot is most linear over the greatest time range. In practice, “most linear” means points fall close to a straight line with minimal systematic curvature (not a line that fits only the first few points).
What “most linear” means (without overrelying on statistics)
When judging linearity, look for:
Random scatter around a straight trend rather than consistent bending
Similar quality of fit across early and late times (not just one region)
Reasonable behaviour near (e.g., the plotted value should be consistent with the initial concentration after transformation)
If using a graphing tool that provides , it can support your judgement, but AP-style reasoning should emphasise the shape and linearity rather than treating as the only criterion.
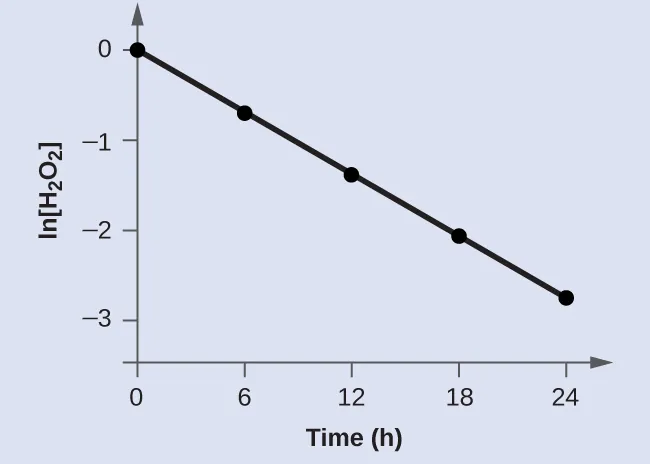
A worked example plot of versus time shows an approximately straight line, consistent with first-order kinetics for hydrogen peroxide decomposition. The linear trend illustrates the integrated rate-law criterion: first order is supported when vs. is linear over the measured time range. This type of figure is useful for explaining conclusions from experimental data rather than from the balanced equation. Source
Practical details that affect your inference
Choosing the concentration data
Use data where:
Temperature and volume are effectively constant
Concentrations are measured or derived consistently (same units throughout)
Time intervals capture both early and late behaviour (enough points to see curvature)
If multiple reactants are present, concentration–time analysis typically follows one reactant while the others are in known relationships or held in large excess; otherwise, changing concentrations of multiple reactants can complicate interpretation of the apparent order.
Common pitfalls
Using product data directly: product formation curves can be used, but only if you correctly relate them to reactant consumption and keep sign conventions consistent.
Including points after the reaction stops being well-behaved: mixing delays, gas loss, side reactions, or instrument saturation can introduce curvature unrelated to order.
Confusing “straight line” with “constant rate”: a straight line in vs. time suggests zero-order-like behaviour, but you still need to check that conditions truly stayed constant and that the linearity holds broadly.
What this method can and cannot establish
It can identify the most consistent order model (0, 1, or 2) for a reactant from concentration–time behaviour.
It cannot prove a mechanism; different mechanisms can produce the same overall order.
It does not require knowing the rate constant in advance; the focus is order from graph behaviour.
FAQ
In practice, aim for at least 6–10 well-spaced time points.
More points are especially helpful early in the reaction where curvature differences between orders are most visible.
Because first-order decay is exponential in $[A]$.
The natural log transforms an exponential relationship into an approximately linear one in time.
It becomes difficult because all three plots may look nearly linear over a short range.
You generally need a meaningful fractional change in $[A]$ to see distinguishing curvature.
Check for experimental issues (mixing delay, temperature drift, measurement saturation).
Also consider that the reaction may not follow simple 0/1/2 order in the measured interval.
Linearity-based order inference does not depend on the specific concentration unit, as long as you are consistent.
However, transformations like $1/[A]$ require a unit that is truly proportional to amount per volume.
Practice Questions
(2 marks) A student plots three graphs using the same concentration–time dataset for reactant A: vs , vs , and vs . Only the vs plot is linear. What is the order with respect to A, and what is the key graphical evidence?
Identifies first order with respect to A (1)
States that vs time being a straight line (most linear) indicates first-order behaviour (1)
(5 marks) Describe a graph-based procedure to determine the reaction order with respect to a reactant A using concentration–time measurements from a single run. Your answer should state which plots you would make and how you would decide the order.
States that concentration of A is measured at multiple times under constant conditions (1)
States to plot vs , vs , and vs (1)
Explains that the plot that is most linear identifies the order (1)
Correctly links linear plots to orders: vs (zero), vs (first), vs (second) (2)

