AP Syllabus focus: ‘A reaction mechanism is a sequence of elementary steps; it may include reactants, intermediates, products, and catalysts.’
Chemical equations show what changes overall, but not how it happens. Reaction mechanisms propose the step-by-step molecular events that convert reactants to products, explaining which species exist transiently during the reaction.
Core idea: the “pathway” of a reaction
Reaction mechanism: A proposed sequence of elementary steps that describes the molecular-level pathway by which reactants are converted to products.
A mechanism is a model for what particles do during a reaction (collide, rearrange bonds, form new species), written as multiple chemical steps rather than one overall equation.
Elementary steps (the building blocks)
Elementary step: A single molecular event that occurs in one “move,” representing a real microscopic process (not a summed overall reaction).
Each elementary step is written like a chemical equation, but it represents an actual event occurring at the particle level. In AP Chemistry, mechanisms are presented as a numbered list of steps that, taken together, describe the full transformation.
Species that may appear in a mechanism
The syllabus emphasises that mechanisms may include reactants, intermediates, products, and catalysts.
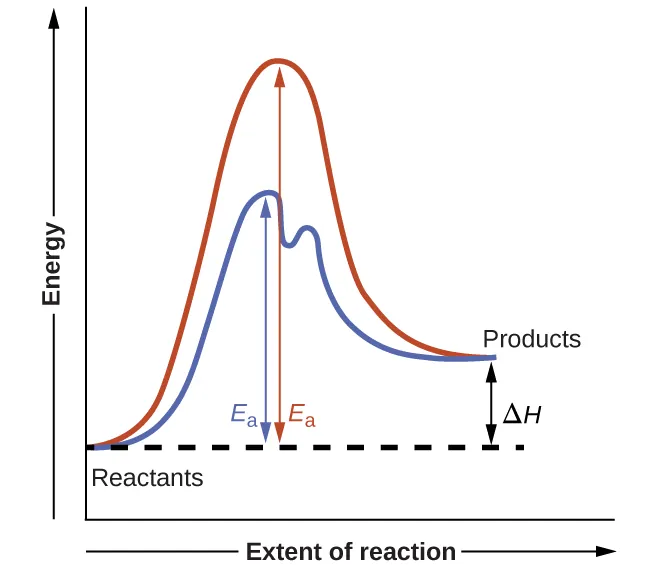
Potential-energy (reaction coordinate) diagram comparing an uncatalyzed one-step pathway to a catalyzed multi-step pathway. The catalyzed route has a lower rate-controlling barrier and introduces an intermediate (the energy “well” between two transition states), illustrating how catalysts change the mechanism without changing the overall reactant/product energy levels. Source
Each type plays a distinct role in the step sequence.
Reactants and products
Reactants are the starting substances for the overall reaction and are consumed through one or more steps.
Products are formed through one or more steps and persist after the reaction is complete.
Mechanisms often show products forming before the final step (for example, a product can be formed in an early step while other changes continue), but products are ultimately the stable outcome of the full sequence.
Intermediates (temporary species)
Intermediate: A species that is formed in one step and consumed in a later step, so it does not remain once the reaction is complete.
Intermediates are important because they represent “in-between” structures along the pathway. They can be atoms, ions, radicals, or molecules, and they may be present at low concentrations because they are quickly used up.
Key characteristics of intermediates in a mechanism:
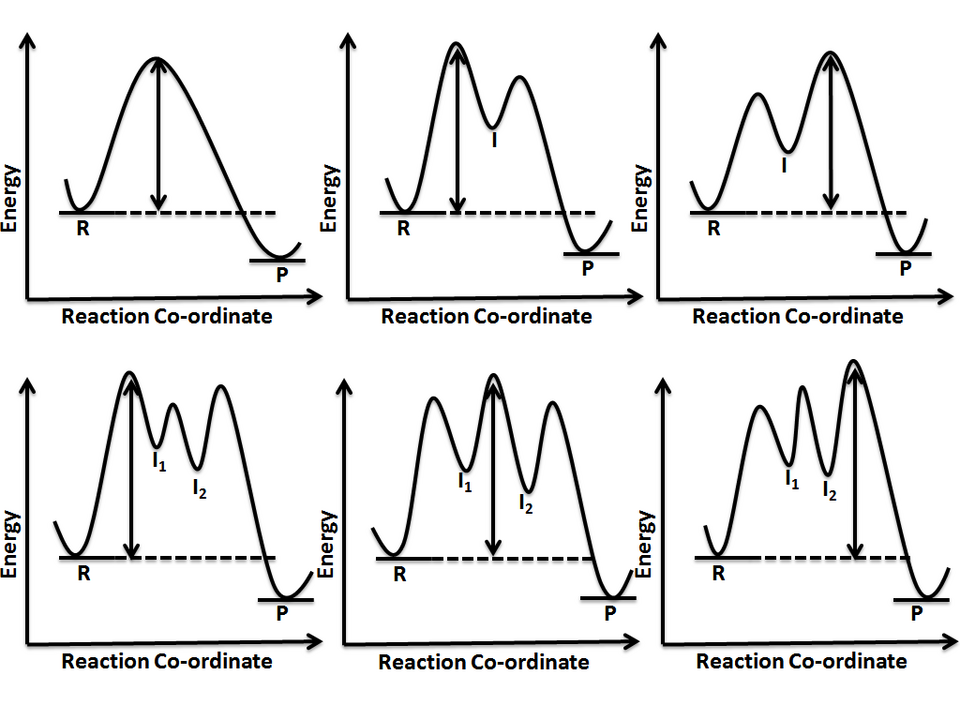
Reaction-coordinate diagrams showing reactions with 0, 1, and 2 intermediates. Each intermediate appears as a local minimum (valley) between transition-state maxima, making it visually clear how multi-step mechanisms differ from single-step processes and why intermediates do not appear in the overall equation. Source
They appear as a product of an earlier elementary step.
They appear as a reactant in a later elementary step.
They are not intended to be stable end substances of the overall process.
Catalysts (pathway-changers)
Catalyst: A substance that participates in the mechanism by providing an alternative pathway, and is regenerated during the sequence so it is not used up overall.
A catalyst can be involved directly in one or more elementary steps, even though it is not consumed overall. In mechanism notation, a catalyst often:
appears as a reactant in an early step (it is used to form a reactive species), and
appears as a product in a later step (it is regenerated).
How mechanisms are written and interpreted
Mechanisms are typically communicated as:
a list of elementary steps (Step 1, Step 2, …), sometimes with conditions noted
chemical formulas and phases/charges to show what species are present at each point
When reading a mechanism, focus on:
what species are present at each step
which species are created only briefly (intermediates)
whether any species is used and then re-formed (suggesting catalysis)
Because a mechanism is a proposed molecular story, different mechanisms can sometimes be suggested for the same overall reaction; the preferred one is the mechanism that best matches observations while remaining chemically reasonable.
FAQ
Yes. Multiple step-by-step pathways may be chemically possible.
Chemists compare predictions (such as which transient species should appear) against evidence to decide which proposal is most consistent.
Mechanisms describe microscopic events that are hard to observe directly.
They are accepted when they best fit experimental constraints and chemical reasoning, but may be revised with new evidence.
Not necessarily, but many are.
Some intermediates can accumulate if their formation is faster than their consumption, making them easier to detect in specialised experiments.
Yes, temporarily.
In a catalytic cycle, the catalyst may be converted into another form and later regenerated, so there is no net loss of catalyst at completion.
Common alternatives include:
arrow-pushing (organic chemistry) to show electron flow
reaction coordinate diagrams to show proposed energy changes along the pathway
catalytic cycles to track regeneration of catalyst forms
Practice Questions
(2 marks) State what is meant by a reaction mechanism and what is meant by an elementary step.
1 mark: Reaction mechanism is a sequence of elementary steps describing the pathway from reactants to products.
1 mark: Elementary step is a single molecular event occurring in one step (not a sum of steps).
(5 marks) A proposed mechanism includes reactants, products, intermediates, and a catalyst. Explain, in terms of how species appear across steps, what distinguishes an intermediate from a catalyst. Include two clear identifying features for each.
1 mark: Intermediate is formed in one step and consumed in a later step.
1 mark: Intermediate does not persist after completion (not present overall).
1 mark: Catalyst participates in steps but is regenerated later.
1 mark: Catalyst is not used up overall / net amount unchanged.
1 mark: Mechanism-level identification tied to appearance as reactant in one step and product in another (for catalyst), versus product then reactant (for intermediate).

