AP Syllabus focus: ‘When added together, the elementary steps in a mechanism should reproduce the overall balanced chemical equation.’
Checking a proposed reaction mechanism against the overall equation is a bookkeeping test: the steps must add up exactly to the balanced net reaction. This ensures the mechanism is chemically plausible at the most basic level.
What it means to “check” a mechanism
A reaction mechanism is a sequence of elementary steps that, when combined, should yield the experimentally known overall reaction.
Overall (net) chemical equation: The balanced reaction obtained after summing all steps and cancelling species that do not appear in the net change.
A mechanism “passes” this check only if the net equation matches the overall equation in stoichiometry, atom counts, and net charge.
How to add elementary steps correctly
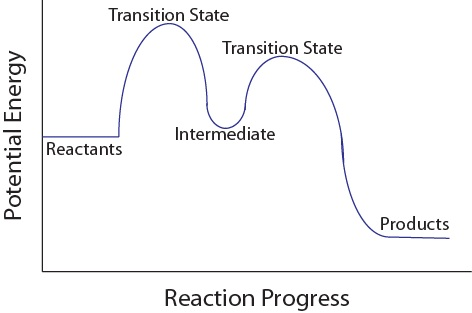
A multistep reaction-coordinate diagram highlighting two transition states separated by a local minimum labeled “intermediate.” This visual supports the idea that intermediates are formed and consumed within the mechanism pathway, so they cancel when elementary steps are summed to reproduce the overall reaction. Source
Mechanism checking is done by algebraically adding chemical equations.
= “sum of,” meaning add left sides together and add right sides together
Use a systematic workflow.
Step 1: Write each step clearly and consistently
Copy each elementary step with correct formulas (including charges) and physical states if provided.
If a step is written as reversible, only add it in the direction it is used in the proposed mechanism.
If needed, multiply an elementary step by an integer so that shared species will cancel properly (you must multiply all coefficients in that step).
Step 2: Add left-hand sides and right-hand sides
Combine all reactants from every step into one “total reactant” side.
Combine all products from every step into one “total product” side.
Keep coefficients explicit; avoid cancelling too early.
Step 3: Cancel species that appear on both sides
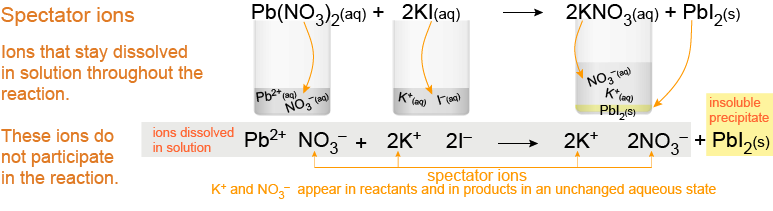
Diagram of a complete ionic equation being simplified to a net ionic equation by canceling spectator ions that appear unchanged on both sides. The visual parallels mechanism checking: species present on both sides are not part of the net change, so careful coefficient-based cancellation produces the final net equation. Source
Species that appear as both a reactant and a product are not part of the net change and must cancel.
Intermediates cancel completely because they are produced in one step and consumed in a later step.
A catalyst (if present) also cancels in the net equation because it is regenerated.
During cancellation:
Only cancel identical species (same formula, charge, and phase).
Cancel coefficients carefully (e.g., if 2 molecules appear on one side and 1 on the other, 1 remains).
Required matching checks
After cancellation, compare your net result to the given overall balanced equation.
Stoichiometry match
The remaining coefficients must match the overall equation (or be reducible to the same ratio).
If your net equation is a multiple of the overall equation (e.g., everything doubled), reduce to simplest whole-number coefficients before deciding it is incorrect.
Conservation of atoms and charge
Even if the equation “looks close,” it must satisfy conservation laws:
Atoms: each element count must match between reactants and products.
Charge: total charge on the reactant side must equal total charge on the product side (especially important for ionic mechanisms).
No “extra” leftover species
If any species remain that are not present in the overall equation, something is wrong:
A step may be missing.
A step may be written in the wrong direction.
Coefficients may require scaling.
The proposed mechanism may simply be incompatible with the overall reaction.
What this check can and cannot tell you
Passing the sum-to-overall check means the mechanism is consistent with the net reaction, but it does not prove the mechanism is correct. Multiple distinct mechanisms can reproduce the same overall equation, so this is a necessary (not sufficient) test.
FAQ
Because adding steps is algebraic: multiplying a step scales how many times that event occurs in the summed pathway.
It does not change the chemistry of that step; it changes how many times it is used overall.
Use the step in the direction required by the proposed pathway.
If the reverse direction is needed, rewrite it reversed before adding so the correct species cancel.
Try reducing the net equation to simplest whole-number coefficients.
If it still disagrees, the mechanism is inconsistent (often a missing step or an incorrect coefficient).
Electrons are treated like any other species: they must cancel when half-reactions are added.
Any leftover $e^-$ indicates the net equation is not a valid overall reaction.
Yes, if they are explicitly included in the elementary steps, they may cancel.
However, if they remain in the net equation but are absent from the stated overall equation, the mechanism and overall equation are not written at the same level of detail.
Practice Questions
Q1 (1–3 marks) A proposed mechanism for is: Step 1: Step 2: Show that the steps add to the overall equation and identify the intermediate.
Adds steps and cancels to obtain (2 marks)
Identifies as the intermediate (1 mark)
Q2 (4–6 marks) Overall reaction: Proposed mechanism: Step 1: Step 2: (a) Add the steps to obtain the net equation. (b) Identify any intermediate and justify using cancellation. (c) Verify charge conservation in the net equation.
Correctly sums steps and cancels species to obtain the given overall equation (3 marks)
Identifies as an intermediate and justifies because it is produced then consumed and cancels (1–2 marks)
Checks net charge reactants equals products in the overall equation (e.g., left: ; right: ) (1 mark)

