AP Syllabus focus: ‘Detecting an intermediate experimentally can support one proposed mechanism over another; collecting intermediate‑detection data is not assessed on the AP Exam.’
Reaction mechanisms are models that explain how reactions proceed step-by-step. Experimental detection of short-lived intermediates provides powerful support for one mechanism over competitors, even when the overall reactants and products are identical.
Why detecting intermediates matters
Mechanisms often predict that certain species should exist briefly during the reaction. If that species is observed under reaction conditions, it can:
Support a proposed mechanism relative to alternatives that do not generate that species
Clarify the sequence of bond-making/bond-breaking events
Indicate whether multiple pathways occur (more than one intermediate may be detectable)
However, detection is usually supporting evidence, not absolute proof: different mechanisms can sometimes generate the same intermediate.
What “intermediate” means in evidence terms
Reaction intermediate: A species formed in one elementary step and consumed in a later step, so it does not appear in the overall balanced equation and typically has a transient, nonzero concentration during the reaction.
A key evidence distinction is between an intermediate and a transition state:
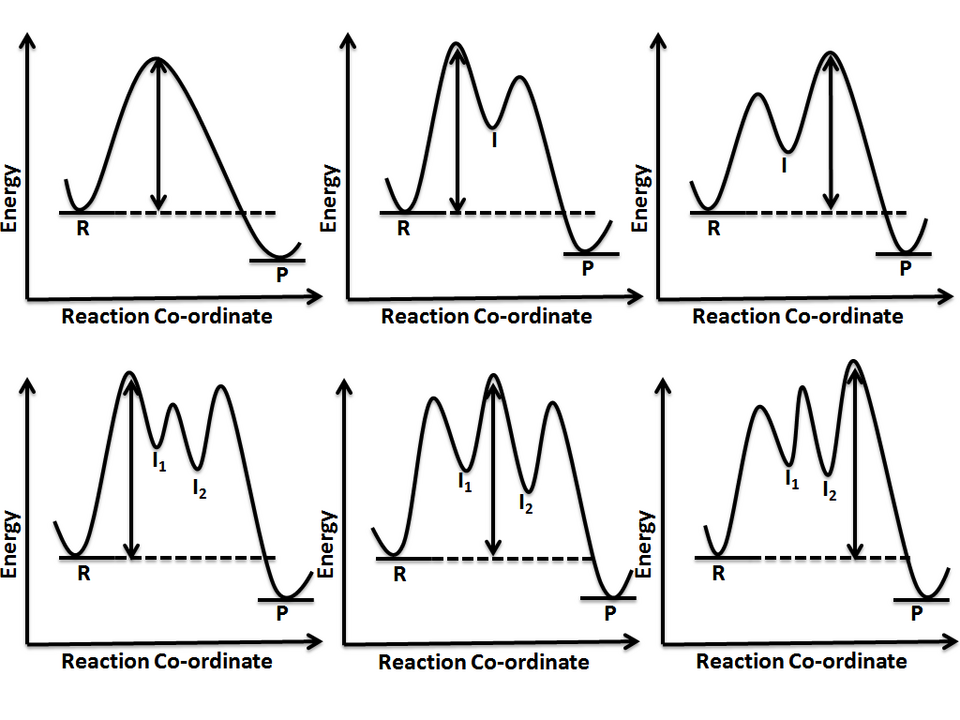
Reaction coordinate (energy) diagrams comparing reactions with no intermediate vs. one or two intermediates. Intermediates appear as local minima between transition-state peaks, emphasizing that they can have a transient, nonzero concentration during the reaction even though they do not appear in the overall equation. This visual helps connect mechanistic “steps” to energy barriers and detectable species. Source
Intermediates can sometimes be detected or even isolated (if sufficiently stable).
Transition states are not isolable species; they are energy maxima along the reaction coordinate and are inferred rather than directly collected.
What intermediate-detection evidence looks like
The syllabus emphasises that collecting intermediate-detection data is not assessed on the AP Exam, but you should understand how such evidence supports mechanisms and how it is interpreted.
Direct, time-resolved observation
Direct detection aims to “see” an intermediate while the reaction runs, often requiring fast techniques because intermediates can exist for milliseconds or less.
Spectroscopic signatures: an intermediate may have a distinctive absorption peak or signal
UV–Vis: appearance/disappearance of a new band over time
IR: transient new bond vibrations
NMR: new chemical environment signals (more feasible for longer-lived intermediates)
EPR: especially useful for radical intermediates
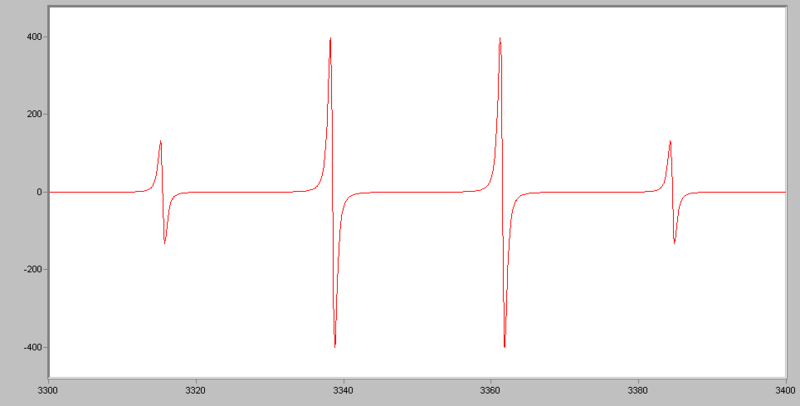
Example EPR spectra illustrating how radicals produce characteristic multi-line patterns due to hyperfine splitting. The displayed methyl-radical spectrum is a concrete example of a “spectroscopic signature” that can directly indicate the presence of a radical intermediate under reaction conditions. This supports the mechanistic idea that detecting the right signal strengthens (but does not alone prove) a proposed pathway. Source
Time dependence consistent with an intermediate:
Signal rises from ~0, reaches a maximum, then falls as it is consumed
The maximum often occurs when formation and consumption rates are comparable
Quenching and trapping approaches
If an intermediate is too short-lived to observe continuously, chemists may “freeze” or divert it.
Quenching: rapidly stopping the reaction (for example, by cooling or changing pH) and analysing what is present at that instant
Trapping: adding a reagent that reacts quickly and selectively with the intermediate to form a more stable, identifiable product
Strong evidence occurs when the trapped product forms only under conditions where the mechanism predicts intermediate formation
Indirect signatures consistent with intermediates
Even without directly observing the intermediate, some observations can be consistent with its presence:
Transient products that appear early and then diminish (suggesting further consumption)
Isotopic labelling patterns that match an intermediate’s rearrangement possibilities (used to discriminate between competing step sequences)
Dependence on conditions (solvent, acidity, inhibitors) that selectively stabilise or destabilise a proposed intermediate, changing its detectability
How intermediate evidence supports one mechanism over another
To use detection as mechanistic support, the observation should align with multiple predictions of the proposed pathway:
Identity match: the detected species fits the structure/charge/radical character predicted
Timing match: it appears after reactants mix and disappears as products accumulate
Consistency with stoichiometry: the intermediate’s formation/consumption fits the stepwise accounting of atoms and charge
Selectivity: alternative mechanisms would not reasonably generate the same species under the same conditions
Limitations and common pitfalls
Intermediate detection must be interpreted cautiously:
A detected species could be a side-product, impurity-derived species, or off-pathway complex
The act of trapping can perturb the mechanism (changing which pathway dominates)
Multiple intermediates may interconvert; observing one does not automatically establish the full step order
Failure to detect an intermediate does not disprove it; it may be below detection limits or too short-lived
FAQ
Intermediates can have a measurable lifetime and sometimes accumulate to detectable concentrations.
Transition states are not isolable species; they are inferred from trends rather than directly collected.
Convincing traps are fast and selective for the proposed intermediate.
They should not appreciably react with starting materials or products, and the trapped product should correlate with conditions that favour intermediate formation.
Solvents and pH can stabilise charge or radical character through solvation and acid–base interactions.
This changes the intermediate’s lifetime and concentration, affecting detectability.
Yes. Parallel pathways can operate, especially if conditions change.
Different intermediates may dominate at different temperatures, concentrations, or in the presence of inhibitors.
It suggests a species is produced and then consumed.
This time profile is characteristic of an intermediate whose concentration peaks when its formation and consumption rates are comparable.
Practice Questions
A proposed mechanism includes an intermediate . Explain how experimentally detecting during the reaction could support this mechanism over an alternative mechanism that does not include .
States that observing indicates it is formed under reaction conditions and is consistent with the proposed stepwise pathway (1)
Links this to discrimination: the alternative mechanism is less supported because it does not predict (1)
A student claims that because a transient species is detected, the entire proposed mechanism is proven correct. Evaluate this claim using mechanistic reasoning about intermediates and detection.
Explains that detecting provides supporting evidence but does not uniquely prove a full mechanism (1)
Notes that different mechanisms can sometimes generate the same intermediate (1)
Identifies that could be an off-pathway species/side-product and detection must establish relevance (1)
Mentions the importance of timing/appearance-then-disappearance behaviour being consistent with formation and consumption (1)
Mentions experimental limitations (e.g., trapping/quenching can perturb pathways or detection limits) (1)

