AP Syllabus focus: ‘An intermediate is formed in one step and consumed in another, so it exists only while the reaction is occurring.’
Reaction mechanisms often include species that do not appear in the overall balanced equation. Correctly identifying reaction intermediates and catalysts helps you interpret mechanisms, connect steps, and avoid common rate-law and stoichiometry errors.
What you are identifying in a mechanism
A reaction mechanism is written as a sequence of elementary steps. Each step lists reactants and products for that step, so you can track which species are produced and later used up.
Reaction intermediate
Intermediate: A species that is formed in one elementary step and consumed in a later step, so it is present only while the reaction is occurring and does not remain at the end.
Intermediates are “internal” to the mechanism: they are necessary for the pathway but cancel when you add the steps.
Catalyst
Catalyst: A species that participates in the mechanism but is regenerated, so it is not used up overall and its net amount is unchanged by the reaction.
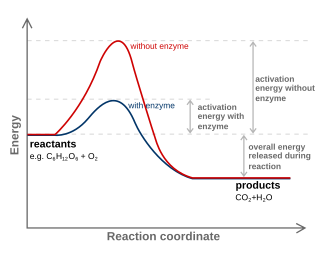
Energy profile showing how a catalyst (or enzyme) lowers the activation energy barrier compared with an uncatalyzed pathway. The diagram highlights that the vertical gap between reactants and products (the overall energy change) is the same in both cases, while the highest point (transition state) is lower for the catalyzed path. This supports the mechanistic idea that catalysts provide an alternate pathway without being consumed overall. Source
Catalysts also cancel when steps are added, but unlike intermediates they typically appear as a reactant early and are re-formed later.
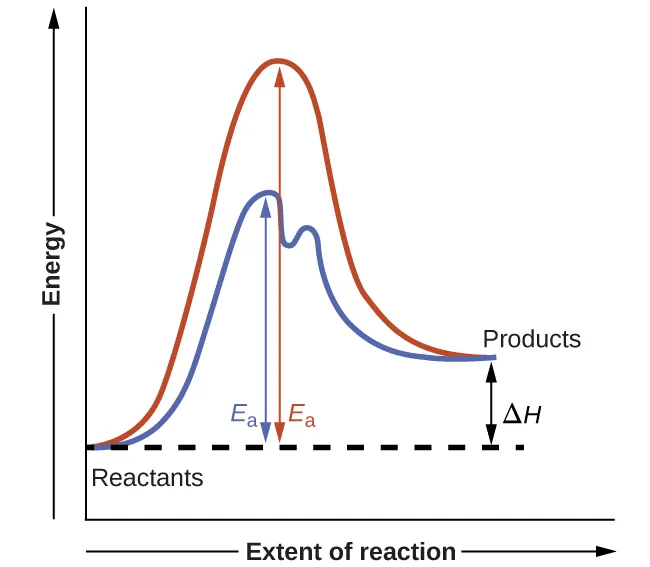
Reaction-coordinate diagram comparing uncatalyzed (single high barrier) and catalyzed (multi-step, lower barriers) pathways for the same overall reaction. The valley between the two peaks on the catalyzed curve represents an intermediate energy minimum, while the reduced peak height illustrates a lower activation energy . The starting and ending energy levels remain unchanged, emphasizing that a catalyst changes the pathway (mechanism) rather than the overall thermodynamics. Source
How to spot an intermediate from elementary steps
Use a bookkeeping approach across the steps.
Step-by-step identification checklist
Write down each species that appears anywhere in the mechanism.
For each species, note whether it appears on the reactant side and/or product side of each step.
Classify using patterns:
Intermediate pattern: produced in one step, then later consumed (often appears as a product in an early step and a reactant in a later step).
Not an intermediate: appears only as an initial reactant (never produced) or only as a final product (never consumed).
Confirm by “adding steps” conceptually:
When the elementary steps are summed, intermediates should cancel out (same coefficient on both sides across steps).
Common visual cues
Intermediates are often written in the middle of a mechanism and may be labelled (e.g., “intermediate”), but you should rely on the formed/consumed rule rather than formatting.
If a species is produced and consumed more than once, it can still be an intermediate; what matters is that it is not present in the net reaction.
How to distinguish intermediates from catalysts
Intermediates and catalysts can both cancel in the net equation, so focus on the net role and where they appear.
Catalyst vs intermediate patterns
Catalyst pattern: present as a reactant in an early step and appears again as a product in a later step (regenerated).
Intermediate pattern: present as a product in an early step and appears later as a reactant (used up).
A quick test:
If removing the species would prevent the first step from occurring, it may be acting as a catalyst/reactant in that step.
If the species is created by the mechanism itself, it is an intermediate.
Avoiding typical mistakes
Do not call a species a catalyst just because it cancels; it must be regenerated and not formed as a necessary first-time product of the mechanism.
Do not label spectators (e.g., ions present in solution but not written in steps) as catalysts or intermediates; only classify species explicitly participating in elementary steps.
Why correct identification matters (within mechanisms)
Intermediates help you understand how the reaction can proceed through multiple smaller collisions rather than an unlikely single event.
Identifying catalysts clarifies why a pathway can change without changing the overall reactants/products, since the catalyst is returned.
FAQ
Yes. List every species and mark each time it appears as a product or reactant across steps.
Any species with at least one “formed” and one later “consumed” occurrence is an intermediate.
No. Being an intermediate requires appearing in separate elementary steps: formed in one step and consumed in another. A same-step appearance is not meaningful for intermediate classification.
Often, but not automatically. A radical is an intermediate only if it is formed in one step and consumed in a later step, and it does not appear in the overall reaction.
It can still be an intermediate. The key is that it is internally cycled and cancels from the overall equation; repeated formation/consumption just indicates a more complex pathway.
It is typically acting like a facilitator within that step (sometimes written to show association/dissociation), but classification should be based on whether it is regenerated across the full set of steps, not within one line.
Practice Questions
Q1 (2 marks) A proposed mechanism has two steps: Step 1: Step 2: Identify the intermediate and state one reason.
identified as the intermediate (1)
Reason: it is formed in Step 1 and consumed in Step 2 / exists only during the reaction (1)
Q2 (5 marks) A mechanism is proposed: Step 1: Step 2: Step 3: (a) Identify the intermediate(s). (2 marks) (b) Identify the catalyst, if any. (2 marks) (c) Give one statement that justifies your catalyst choice. (1 mark)
is an intermediate (1)
is an intermediate (1) (b)
is the catalyst (2) (c)
is consumed in Steps 1/2 and regenerated in Step 3 / net amount unchanged (1)

