AP Syllabus focus: ‘Equilibrium arguments like Le Châtelier’s principle do not apply to electrochemical systems because these systems are not at equilibrium.’
Electrochemical cells change reactant and product amounts by moving electrons through a circuit, so their behaviour is governed by thermodynamic driving forces rather than equilibrium “stress-response” reasoning.
Why Le Châtelier’s Principle Fails for Electrochemical Cells
What Le Châtelier’s Principle Assumes
Le Châtelier’s principle: If a system at equilibrium is disturbed (by concentration, pressure, or temperature), it shifts in the direction that partially counteracts the disturbance.
That definition contains the key limitation: it describes how a system responds when it is already at equilibrium (forward and reverse processes occurring at equal rates).
Electrochemical Cells Are Typically Not at Equilibrium
A working galvanic or electrolytic cell is usually in a nonequilibrium state because:
There is a measurable cell potential (a driving force), which indicates a tendency for net reaction in one direction.
Electrons are being pushed/pulled through an external circuit, continuously changing the reaction mixture.
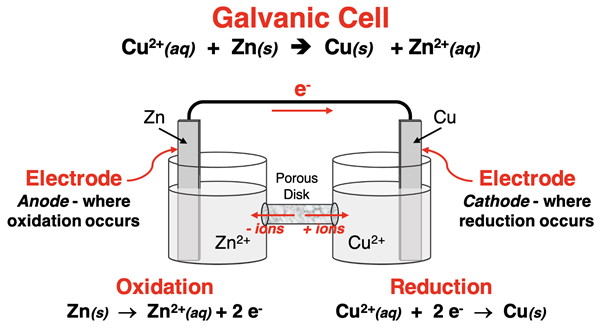
Labeled schematic of a Zn/Cu galvanic cell showing anode (oxidation) and cathode (reduction), with electron flow through the wire and ion migration through the separator. This helps link changing amounts of reactants/products to sustained current and a nonzero cell potential rather than to an equilibrium shift. Source
The system is evolving toward equilibrium, but it is not “sitting at” equilibrium while it operates.
Only when the cell reaches equilibrium does the driving force disappear. In practice, that corresponds to zero voltage, so the cell can no longer do electrical work spontaneously.
Voltage, Not “Shift,” Is the Meaningful Response Variable
In equilibrium problems, adding reactant or removing product is often described as causing a “shift.” In cell chemistry, the direct observable response to changing conditions is a change in cell potential, because concentrations alter the thermodynamic tendency for electrons to flow.
If conditions favour products more strongly, the magnitude of the cell potential increases for a galvanic cell.
As the reaction proceeds and the system approaches equilibrium, the cell potential decreases until it reaches zero.
Why “Adding Reactant Shifts Right” Is an Incomplete (and Often Wrong) Cell Argument
Applying Le Châtelier’s language to a cell can mislead because it suggests:
The system is already at equilibrium (it is not, while voltage is nonzero).
The response is a “shift” rather than a change in electrical driving force.
You can predict direction without tracking electron flow and the reaction quotient.
Instead, you should reason with thermodynamic favourability under current conditions: the cell reaction proceeds in the direction that reduces the driving force until equilibrium is reached.
The Correct Framework: Reaction Quotient and Driving Force
= cell potential under nonstandard conditions (V)
= standard cell potential (V)
= reaction quotient (unitless, constructed from activities/concentrations/pressures)
= moles of electrons transferred per mole of reaction (mol e−)
= gas constant (J mol−1 K−1)
= temperature (K)
= Faraday constant (C mol−1 e−)
This relationship captures what concentration changes actually do in a cell: they change Q, which changes E.
A system “moving toward equilibrium” in cell terms means Q is changing toward its equilibrium value, while E trends toward zero.
When Le Châtelier Becomes Applicable (and Why It’s Still Limited)
Le Châtelier’s principle can describe how an equilibrium mixture would respond to a disturbance, but an operating cell is designed to prevent equilibrium by:
separating half-reactions,
allowing electron transfer through a wire,
maintaining net current and net chemical change.
You can discuss equilibrium-style shifts only in the special case where the cell has effectively stopped (E = 0), because only then is the overall system at equilibrium and the usual equilibrium assumptions hold.
Practical Implication for AP Chemistry Reasoning
When asked about changing concentration or pressure in a cell, avoid “shift left/right” language as the core justification. Use statements tied to cell behaviour:
how the change affects Q,
how that changes the cell potential,
how the altered potential changes the tendency for net electron flow until equilibrium is approached.
FAQ
$K$ describes the equilibrium position the system would reach if allowed to run until no driving force remains.
It is a reference point for where $Q$ is heading, not a description of a working cell’s current state.
Only if you are explicitly discussing the hypothetical equilibrium mixture (or the cell after it has stopped with $E=0$).
For operating cells, focus on how $Q$ changes and how that changes $E$.
The overall redox reaction across both half-cells is the relevant system.
Because the half-reactions are physically separated and coupled by a circuit, the setup is designed to sustain non-equilibrium conditions while operating.
Not always; it depends on whether the change makes $Q$ smaller or larger relative to the products/reactants expression.
The sign of $\ln Q$ in the Nernst relationship determines whether $E$ rises or falls.
At equilibrium there is no net thermodynamic driving force for the redox reaction, so no net electron flow can be sustained.
That absence of driving force is reflected by zero cell potential, $E=0$.
Practice Questions
A student says: “If you add more reactant to a galvanic cell, Le Châtelier’s principle says the reaction shifts right, so the voltage must increase.” State why Le Châtelier’s principle is not the correct justification for predicting voltage changes in an operating cell.
States that Le Châtelier’s principle applies to systems at equilibrium, but an operating cell is not at equilibrium (1)
Links non-equilibrium operation to nonzero cell potential / net electron flow (1)
States correct approach: concentration changes alter and therefore (not an equilibrium “shift”) (1)
Explain, using thermodynamic reasoning, why equilibrium arguments like Le Châtelier’s principle do not apply to electrochemical cells while they are producing a current. In your answer, refer to the meaning of , the approach to equilibrium, and the condition at equilibrium.
Identifies that Le Châtelier requires an equilibrium system (1)
States that a cell producing current has net reaction and is therefore not at equilibrium (1)
Explains that a nonzero indicates a driving force for spontaneous net change (1)
Describes that as the cell runs, concentrations change so changes and changes accordingly (1)
States that the system approaches equilibrium as the driving force diminishes (1)
States that at equilibrium, (and no net current can be produced) (1)

