AP Syllabus focus: ‘In concentration cells, determine spontaneous electron flow by identifying the direction that moves the system toward equilibrium.’
Concentration cells are electrochemical cells where the driving force is a concentration difference, not different substances. To predict electron flow, focus on how the redox process reduces that concentration difference toward equilibrium.
What a Concentration Cell Is
A concentration cell uses the same redox couple in both half-cells, but at different concentrations (or, more generally, different activities/partial pressures). Because the chemical identities match, the cell’s tendency to run comes entirely from “wanting” to remove the gradient.
Concentration cell: A galvanic cell in which both half-cells contain the same species undergoing oxidation/reduction, but at different concentrations (or activities), producing a voltage that drives the system toward equal conditions.
The key conceptual test is: which direction of electron flow makes the two sides more similar?
The Central Rule: Electron Flow Reduces the Gradient
In any spontaneous concentration cell:
The cell operates in the direction that moves the system toward equilibrium by reducing the concentration difference.
The half-cell that is effectively “short” of a species will tend to produce it; the half-cell that has “extra” will tend to consume it.
Electrons move through the wire from oxidation to reduction, and the coupled ion changes occur to lessen the disparity.
Metal–Metal Ion Concentration Cells (Most Common AP Focus)
Consider on both sides, but with different .
To move toward equilibrium, the cell should:
Increase the lower ion concentration
Decrease the higher ion concentration
That happens if:
The dilute side is the anode (oxidation):
This adds to the dilute solution, raising its concentration.
The metal electrode on this side tends to lose mass.
The concentrated side is the cathode (reduction):
This removes from the concentrated solution, lowering its concentration.
The metal electrode on this side tends to gain mass.
So, for metal/ion concentration cells:
Electrons flow from the dilute-ion half-cell to the concentrated-ion half-cell.
Why This Direction Is “Spontaneous”
If is higher on one side, reducing there is favoured because it relieves the “excess” of ions; oxidising the metal on the dilute side supplies ions where they are “needed.” The overall effect is concentration equalisation.
How to Decide Direction Without Memorising
When given a concentration cell, determine which species differs (often an aqueous ion), then apply a “fix the imbalance” lens:
If a side has lower concentration of a reactant needed for reduction (like ), reduction there would make it even lower, which is the wrong direction for reaching equilibrium; that side should instead run oxidation to generate more of that species.
If a side has higher concentration of that species, reduction there consumes it and pushes the system toward equality.
A quick reasoning checklist:
Identify the species with different concentration.
Ask: “Which half-reaction would increase the low concentration and decrease the high concentration?”
Assign oxidation/reduction accordingly, then infer electron flow (oxidation side reduction side).
Direction of Ion Movement (Qualitative)
Because charge must balance as electrons move:
The solution at the anode tends to become more positive (often producing cations such as ).
The solution at the cathode tends to become less positive (often consuming cations).
Ions in the salt bridge (if present) migrate to maintain electroneutrality, enabling continued current.
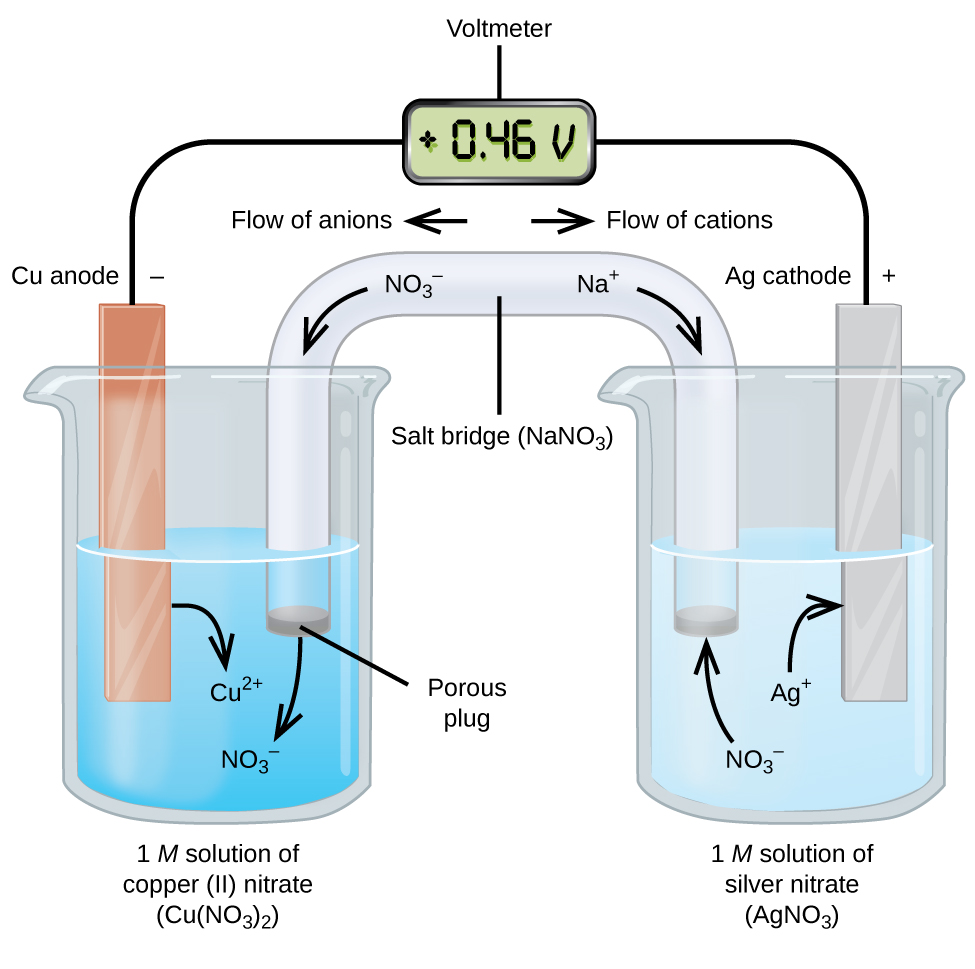
Galvanic cell diagram showing electron flow through the external circuit and counter-ion migration through the salt bridge. The figure highlights that anions migrate toward the anode and cations migrate toward the cathode to prevent charge buildup, which is essential for sustained current. Source
When the Cell Stops
As the cell runs:
The concentration difference shrinks.
The driving force decreases.
When the concentrations become effectively equal, there is no remaining “push” toward equilibrium, so the cell potential goes to zero and net electron flow stops.
FAQ
No. They can use the same metal and the same ion; the voltage arises from a concentration (activity) difference that the cell reduces over time.
Focus on which half-reaction would consume the excess and produce the deficiency for the species whose concentration differs, then assign oxidation/reduction accordingly.
The same “reduce the gradient” logic applies, but electrode mass may not change; instead, solution species interconvert near each electrode.
Yes. Treat different partial pressures like different concentrations: the spontaneous direction reduces the pressure difference, shifting the electrode reactions accordingly.
Non-ideal behaviour can matter: activity effects, liquid junction potentials, slow electrode kinetics, or mixing can reduce or obscure the expected driving force.
Practice Questions
(2 marks) In a concentration cell, one half-cell has and the other has . State the direction of spontaneous electron flow.
Electrons flow from the 0.10 M half-cell to the 1.0 M half-cell. (1)
Justification linked to moving toward equilibrium: dilute side oxidises to increase and concentrated side reduces to decrease . (1)
(5 marks) A concentration cell is constructed with in both half-cells. The left has lower than the right. Without calculations, determine (i) which side is the anode, (ii) which side gains electrode mass, and (iii) how the concentration difference changes as the cell operates.
Left (lower ) is the anode because oxidation produces . (2)
Right (higher ) is the cathode because reduction consumes . (1)
The cathode electrode gains mass due to plating of . (1)
The concentration difference decreases (left increases; right decreases), moving toward equilibrium. (1)

