AP Syllabus focus: ‘Using the Nernst equation conceptually, E = E° − (RT/nF) ln Q, helps explain concentration effects; algorithmic calculations alone do not show understanding.’
Cell potentials change when concentrations change because the driving force depends on how “far” the system is from its preferred product/reactant ratio. The Nernst equation links voltage to reaction composition in a concept-first way.
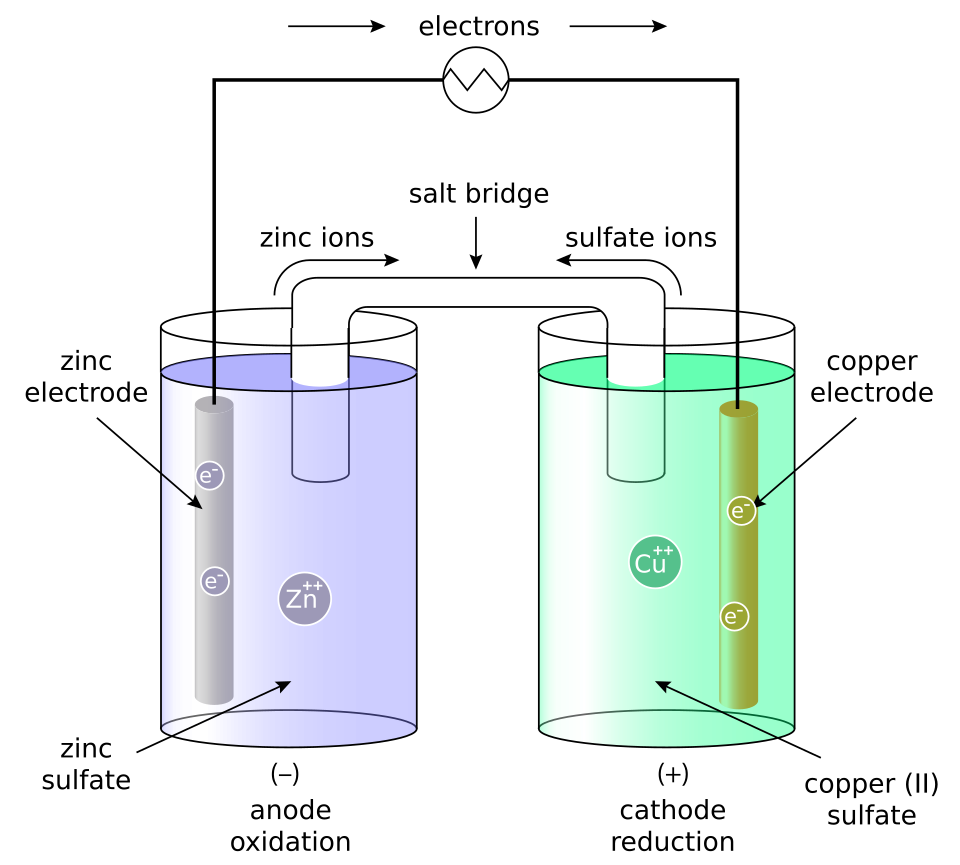
Labeled Daniell (Zn/Cu) galvanic cell diagram showing the anode/cathode, direction of electron flow through the wire, and ion migration through the salt bridge to maintain charge balance. This picture helps connect the abstract symbol to the physical cell whose voltage responds when solution compositions (and therefore ) change. Source
The Nernst equation as a meaning-making tool
The key idea is that nonstandard conditions shift the measured cell potential away from the standard potential because the relative amounts of reactants and products change the tendency for electron transfer.
Reaction quotient (Q): A ratio of product “amounts” to reactant “amounts,” each raised to their stoichiometric coefficients, using current (not necessarily equilibrium) conditions.
encodes composition:
If products are relatively abundant compared with reactants, is larger.
If reactants are relatively abundant, is smaller.
Pure solids and liquids are omitted from (treated as constant).
What the equation says (and how to read it)
The Nernst equation is most useful when you interpret how changing , , or must change .
= cell potential under the current conditions (V)
= standard cell potential (V)
= gas constant ()
= absolute temperature (K)
= moles of electrons transferred as written in the balanced redox reaction (mol e)
= Faraday constant ()
= reaction quotient (unitless, using activities/approximated by concentrations or partial pressures)
The minus sign is the most important “concept handle”:
As increases, increases, so decreases.
As decreases (below 1), is negative, so increases above .
Interpreting concentration changes without plugging in numbers
Think in terms of how a stress changes , then how changes, then how responds.
If you add reactant or remove product
decreases (denominator bigger and/or numerator smaller)
becomes more negative
becomes more positive
increases (greater driving force)
If you add product or remove reactant
increases
becomes more positive
the subtraction term becomes more negative
decreases (reduced driving force)
If only a gas pressure changes
For gases, partial pressures appear in similarly to concentrations:
Increasing a reactant gas pressure tends to lower → raises
Increasing a product gas pressure tends to raise → lowers
How and shape sensitivity (still qualitative)
The factor controls how strongly composition affects voltage.
Larger makes larger, so becomes more sensitive to changes in (composition matters more at higher temperature).
Larger makes smaller, so is less sensitive to the same multiplicative change in (more electrons “dilute” the effect per electron).
Common qualitative reasoning checkpoints
Don’t change when concentrations change; only changes because changes.
Use the balanced overall redox equation to build (coefficients become exponents).
If a species is not in (pure solid/liquid), changing its amount does not directly change , so it does not directly shift via the Nernst term.
“Concept over plug-and-chug” means you can justify direction and relative change in by tracking , not just computing.
FAQ
Their activity is effectively constant (defined as 1) in the standard-state framework.
Changing the mass of a pure solid/liquid doesn’t change its activity, so it doesn’t appear in $Q$.
The multiplier $\dfrac{RT}{nF}$ increases with $T$, so the magnitude of the $\ln Q$ correction typically grows at higher temperature.
This does not automatically mean $E$ increases; it means composition has a larger effect.
It comes from $\ln Q = 2.303\log Q$.
At $298\ \text{K}$, many courses use $E = E^\circ - \dfrac{0.0592}{n}\log Q$ as a convenient approximation, but the conceptual dependence is the same.
Because electrochemical free energy depends logarithmically on composition: $\Delta G = \Delta G^\circ + RT\ln Q$.
Voltage is linked to free energy per charge, so the logarithm carries over.
In dilute solutions and near-ideal gases, activities are well-approximated by:
activity $\approx$ concentration for solutes
activity $\approx$ partial pressure (relative to standard state) for gases
At higher ionic strength, deviations can matter.
Practice Questions
(2 marks) For a galvanic cell whose overall reaction has products in the numerator of , the product concentrations are increased while temperature is constant. State and explain what happens to .
1 mark: States decreases.
1 mark: Explains that increasing product concentration increases , so increases and the term becomes more negative, lowering .
(5 marks) Consider the reaction at constant . Using , explain qualitatively how changes when: (i) A is added, (ii) D is removed, and (iii) the reaction is written with a larger . No calculations required.
1 mark: Identifies that adding A decreases (reactant in denominator) and therefore increases .
1 mark: Links decreased to smaller/more negative and thus larger .
1 mark: Identifies that removing D decreases (product in numerator) and therefore increases .
1 mark: Links decreased to change and the negative sign leading to increased .
1 mark: Explains that larger reduces , so is less sensitive to changes in .

