AP Syllabus focus: ‘The free energy change for dissolution reflects multiple contributions: breaking interactions in the solid, reorganizing solvent, and forming solute–solvent interactions.’
Dissolution is not a single-step event; it is a thermodynamic “accounting” of several microscopic changes. ΔG° of dissolution captures the net effect of these competing contributions under defined standard conditions.
What ΔG° of Dissolution Represents
The idea behind “free energy change”
Dissolution describes a solute becoming dispersed throughout a solvent as particles separate from the original solute phase and become surrounded by solvent molecules/ions. The standard Gibbs free energy change of dissolution is the overall driving force for that transformation when all species are in their standard states.
Standard Gibbs free energy change of dissolution, ΔG°diss: the free energy change when a solute dissolves to form dissolved particles in their standard state (commonly idealised as 1.0 M for solutes) at a specified temperature.
A key point for AP Chemistry is that ΔG°diss is a net quantity: it does not “belong” to just the solute or just the solvent, but to the whole system undergoing dissolution.
Why multiple contributions matter
The specification emphasises that ΔG°diss reflects multiple contributions, meaning dissolution should be viewed as a sum of underlying physical changes. Conceptually, the overall change can be organised into three interacting pieces:
Breaking interactions in the solid (or solute phase)
Solute particles (ions, molecules, or atoms) are held together by attractions (ionic attractions, metallic bonding, intermolecular forces).
Separating these particles requires overcoming those attractions, which tends to oppose dissolution.
Reorganising the solvent
Solvent molecules are already interacting with each other (for water, hydrogen bonding is especially important).
Making space for solute particles typically requires disrupting some solvent–solvent attractions and arranging solvent around new particles, which can either oppose or assist dissolution depending on the system.
Forming solute–solvent interactions
New attractions form between solute and solvent particles (for aqueous solutions, often described as hydration).
Stronger solute–solvent attractions generally promote dissolution by stabilising the dissolved state.
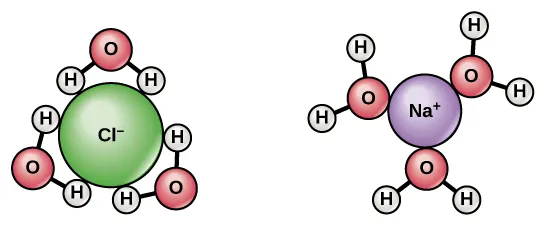
This diagram shows water molecules orienting around ions to form hydration shells: the partially negative oxygen ends point toward cations, while the partially positive hydrogen ends point toward anions. The figure makes the microscopic basis of ion–dipole attraction concrete, showing how new solute–solvent interactions stabilize dissolved ions. This is a key part of why hydration can strongly influence the net . Source
Interpreting ΔG°diss as a “competition”
Net balance, not a single cause
Because ΔG°diss is the result of competing effects, you should avoid single-factor reasoning such as “high lattice energy means insoluble” without considering solvent and solute–solvent effects. Two substances can both require significant separation of solute particles, yet differ greatly in ΔG°diss because one forms much more stabilising solute–solvent interactions (or requires less disruptive solvent reorganisation).
What ΔG°diss does (and does not) tell you
ΔG°diss is best interpreted as describing the thermodynamic tendency for dissolution under standard-state definitions.
If ΔG°diss is more negative, dissolution is more thermodynamically favoured under those standard conditions.
If ΔG°diss is more positive, the dissolved state is less favoured under those standard conditions.
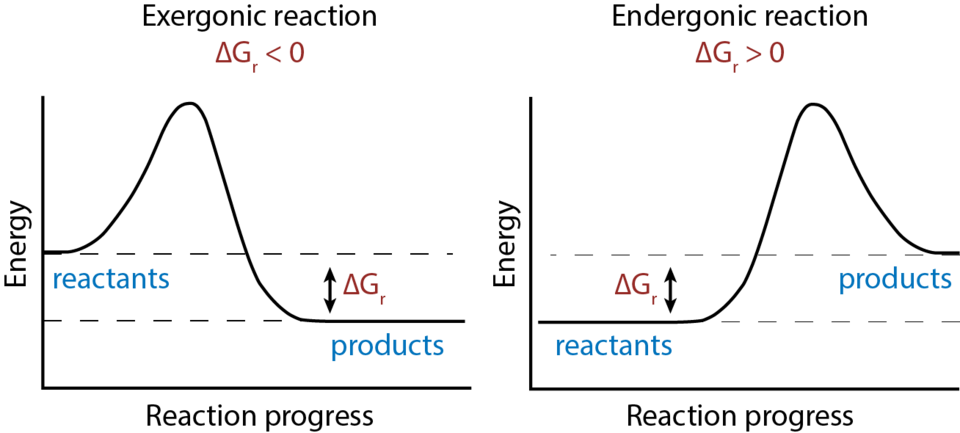
Energy profile diagrams compare exergonic and endergonic processes by plotting Gibbs free energy versus reaction progress. The vertical drop (or rise) between reactants and products represents the sign of and therefore whether the process is thermodynamically favorable under the stated conditions. This reinforces that dissolution outcomes depend on the net free-energy balance, not a single microscopic step. Source
However, ΔG°diss does not identify which microscopic contribution dominates unless additional information is provided (for instance, separate enthalpic/entropic discussion or molecular-level evidence). It also does not directly describe how fast dissolution occurs, since rate depends on kinetic factors such as surface area and stirring rather than the free-energy accounting.
Connecting the specification statement to real systems
Solids vs liquids, ionic vs molecular solutes
The three contributions appear in all dissolution processes, but their relative importance varies:
Ionic solids in water: breaking ionic attractions can be substantial, and the extent to which water stabilises ions through ion–dipole interactions strongly affects ΔG°diss.
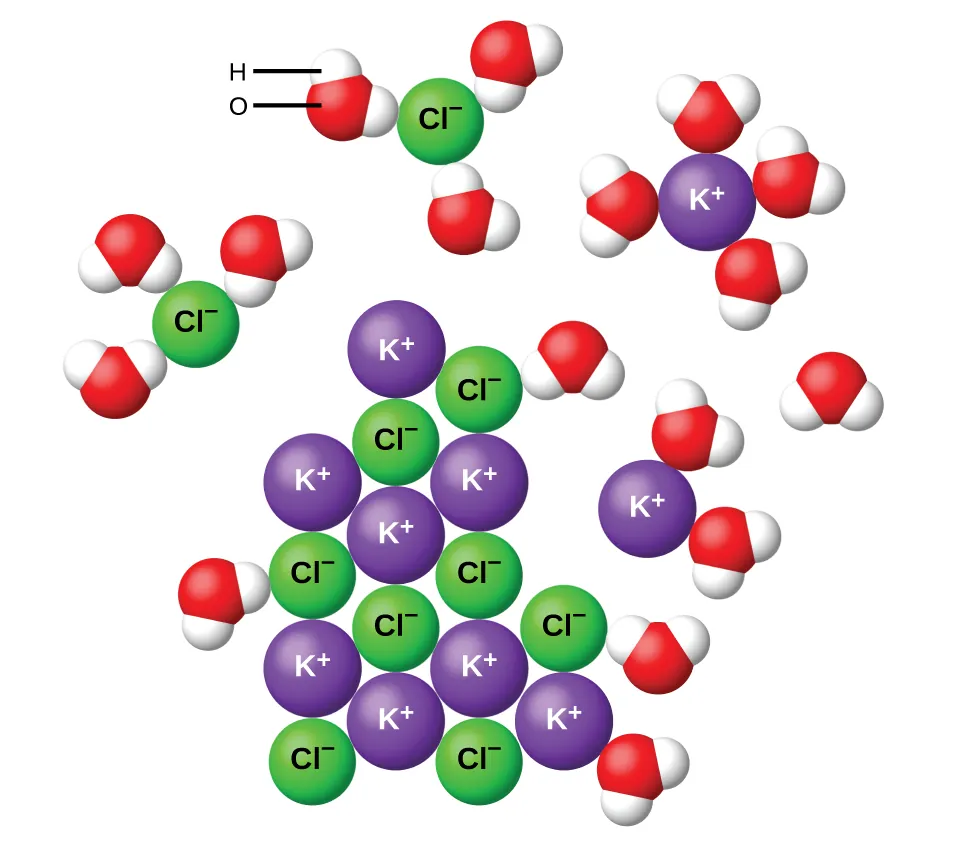
The diagram depicts an ionic solid (KCl) separating into ions while water molecules surround each ion, illustrating hydration during dissolution. It emphasizes that dissolving an ionic solid requires overcoming ion–ion attractions while simultaneously forming stabilizing ion–dipole interactions with the solvent. This visualization supports the idea that reflects competing contributions rather than a single cause. Source
Molecular solids in water: disrupting crystal packing competes with whether the molecule can make favourable interactions with water (polarity and hydrogen bonding capability often matter).
Nonpolar solutes in water: solvent reorganisation can be a major factor because water may need to adopt more ordered local structures around nonpolar particles, changing the net free energy.
The required AP-level takeaway is that ΔG°diss is a single reported value that bundles these effects into one thermodynamic result, consistent with the syllabus wording about “multiple contributions.”
FAQ
It treats the solution as behaving ideally at a defined reference concentration.
In real solutions, particle interactions cause deviations, so chemists use activities; the 1.0 M standard state is a convenient reference, not a guarantee of ideal behaviour.
The solvent may need to lose favourable solvent–solvent interactions or adopt a more constrained arrangement around the solute.
That structural “cost” can outweigh the stabilisation from new solute–solvent attractions.
Equilibrium measurements tied to how much solute exists in the dissolved state at a defined temperature.
From such data, ΔG° can be obtained via thermodynamic relationships, but the measurement is fundamentally about equilibrium, not rate.
ΔG°diss is a state function for the chemical change and does not depend on surface area.
Surface area affects how quickly equilibrium is reached, not the thermodynamic value itself (assuming the same phases and conditions).
If dissolved particles react further (for example, forming complexes), the dissolved species are stabilised.
That shifts the overall free-energy balance for the process as observed, because “dissolution” effectively becomes coupled to additional stabilising interactions in solution.
Practice Questions
(5 marks) A student claims: “If a solute has strong attractions between its particles, its ΔG° of dissolution must be positive.” Evaluate this claim using the meaning of ΔG° of dissolution and the contributions named in the specification.
Describes ΔG°diss as the net free energy change for dissolution under standard states (1)
Explains that breaking solute–solute interactions can oppose dissolution (1)
Explains that solvent reorganisation contributes and can oppose or favour depending on the case (1)
Explains that forming solute–solvent interactions can favour dissolution and offset strong solute attractions (1)
Concludes the claim is not necessarily true because ΔG°diss depends on the balance of all contributions (1)
(2 marks) Explain, in terms of the components of ΔG° of dissolution, why dissolving a solid involves more than simply “breaking the solid apart”.
States that ΔG°diss includes multiple contributions rather than one step (1)
Identifies at least two of: breaking solute interactions, solvent reorganisation, forming solute–solvent interactions (1)

