AP Syllabus focus: ‘Predicting total ΔG° of dissolution can be challenging because the free-energy contributions from different factors can partially cancel.’
Solubility is controlled by a balance of energetic and entropic changes that often oppose each other. Because these competing contributions can be similar in magnitude, small differences in structure, temperature, or solvent can flip the predicted outcome.
Why “predicting solubility” is thermodynamically subtle
Solubility under a given set of conditions reflects whether dissolving is thermodynamically favourable overall. The difficulty is that dissolution is rarely driven by a single effect; instead, multiple free-energy contributions add together, and their sum may be small.
Free-energy cancellation: when large positive and negative contributions to a process’s Gibbs free energy offset, making the net small and highly sensitive to small changes.
A small net means the system is “on the fence,” so predictions based on one dominant factor (e.g., “ionic = soluble”) can fail.
Competing terms that can offset during dissolution
Enthalpy–entropy competition
Dissolution commonly features opposing signs in and , so the net free energy depends on which term wins at the temperature of interest.
= standard Gibbs free energy change for dissolution, J mol
= standard enthalpy change for dissolution, J mol
= absolute temperature, K
= standard entropy change for dissolution, J mol K
Even when you can qualitatively guess the signs, the magnitudes of and may be close enough to partially cancel.
Enthalpic “give-and-take” can nearly balance
In many dissolutions, enthalpy includes both energy costs and energy releases, such as:
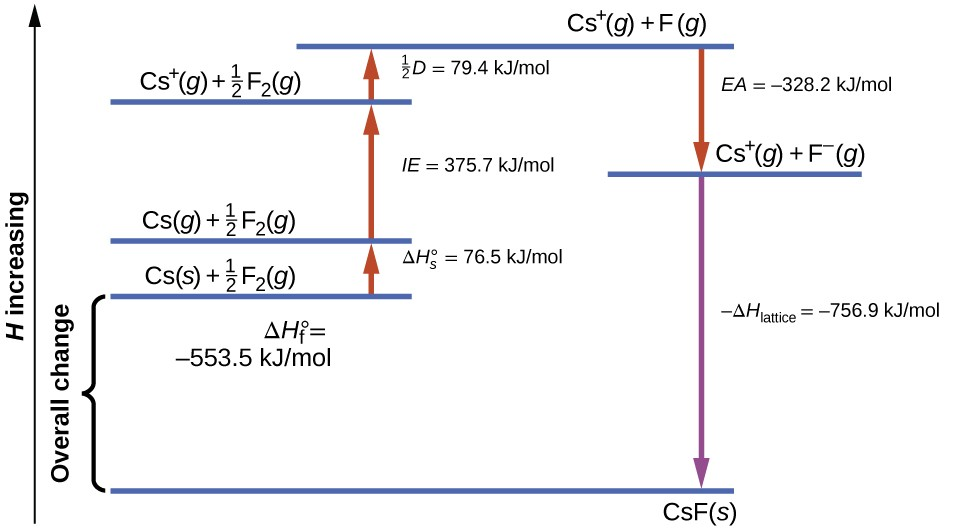
Born–Haber-type thermochemical cycle diagram showing enthalpy changes as a sequence of steps with arrows (energy increasing upward). This kind of stepwise energy accounting is the same logic used in dissolution: large positive steps (separating species) can be offset by large negative steps (forming stabilizing interactions), so the net can end up small. Source
Costs from separating solute particles (breaking an extended solid structure)
Releases from forming new solute–solvent attractions
If these are similar in magnitude, ends up near zero, and the outcome becomes dominated by entropy (or vice versa). Because these interactions depend on ion size, charge density, and molecular polarity, small chemical changes can shift the balance.
Entropy is not “always increasing” in a simple way
A frequent pitfall is assuming “mixing increases disorder, so .” In reality, for dissolution is a sum of multiple, sometimes opposing, contributions:
Dispersal of solute particles into solution (often increases entropy)
Solvent organisation around solute particles (can decrease entropy)
Changes in the number and freedom of independently moving species (context-dependent)
Because these can offset, can be small or even negative, especially when dissolving species that strongly constrain nearby solvent motion.
Why cancellations make predictions fragile
Small net amplifies “minor” effects
When cancellations occur, the net free energy can be comparable to typical uncertainties in estimation.
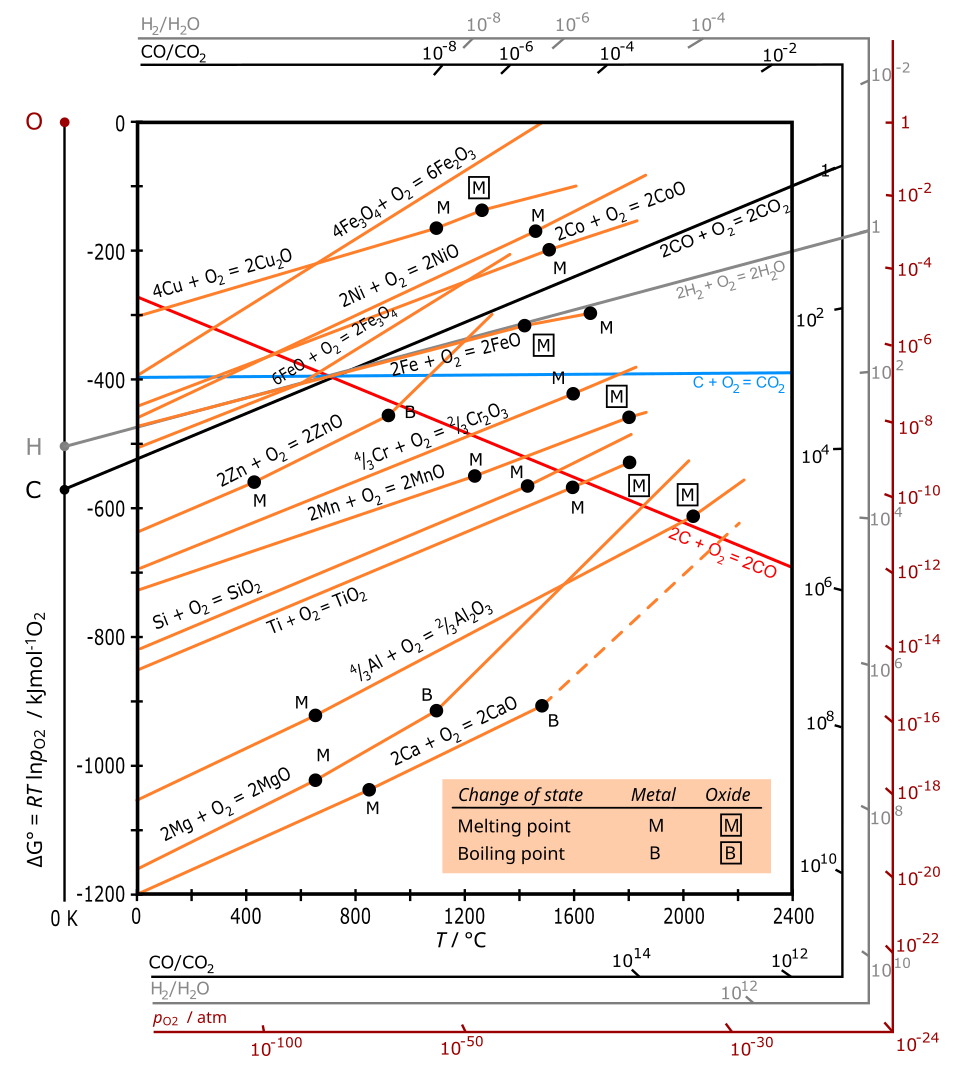
Ellingham diagram: a set of straight-line plots of versus temperature, where the slope relates to and the intercept relates to . The key study takeaway is that modest temperature changes can move a borderline process across , illustrating why ‘free-energy cancellation’ makes predictions fragile. Source
Then, seemingly minor factors become decisive:
Temperature: changing rescales the entropy term , so a borderline process can switch from unfavourable to favourable (or the reverse).
Different solid forms: changes in crystal packing (polymorphs) alter the balance of terms.
Solvent identity: a solvent that stabilises solute particles slightly better can change the net sign of .
Qualitative rules can contradict each other
Heuristics like “higher charge = stronger solute–solvent attractions” compete with “higher charge = harder to separate the solid.” Both can be true simultaneously, so the net effect is not obvious without knowing which term dominates for that specific solute–solvent pair.
The net result can be close to zero
If is small because of cancellation, the system may show:
Only modest solubility (neither “insoluble” nor “highly soluble”)
Strong sensitivity to conditions (temperature changes or slight formulation changes)
In this regime, predicting solubility is hard precisely because the process is governed by a difference of large numbers, not a single large driving force.
FAQ
Different crystal packing changes the energetic cost of separating particles and can also alter how a solvent organises around released species. If terms nearly cancel, small structural differences can dominate the net $\Delta G^\circ$.
When $\Delta H^\circ$ and $T\Delta S^\circ$ nearly cancel, changing $T$ noticeably shifts the $T\Delta S^\circ$ term.
A borderline $\Delta G^\circ$ can change sign.
The process has no strong thermodynamic push either way: dissolving and not dissolving are similarly favourable. Small effects (impurities, solvent composition, temperature) can then control the observed solubility.
They typically focus on one contribution (e.g., stronger attractions) while ignoring an opposing one (e.g., higher energetic cost to separate the solid). Cancellation means both must be considered together.
If the true net $\Delta G^\circ$ is small, then modest uncertainties in estimated enthalpy/entropy contributions can be comparable to the net value, making predictions unreliable without direct measurement.
Practice Questions
Q1 (2 marks): Explain, in terms of Gibbs free energy, why solubility predictions can be difficult when and are similar in magnitude.
States that and that terms can oppose (1)
Explains that near-cancellation makes net small so small changes (e.g., temperature/structure) can change favourability (1)
Q2 (5 marks): A student claims “dissolving always increases entropy, so any substance with strong solute–solvent attractions will definitely be soluble.” Evaluate this claim using the idea of competing terms and cancellations.
Identifies that for dissolution can be positive or negative due to multiple contributions (1)
Notes solvent organisation can decrease entropy and offset dispersal/mixing (1)
Explains strong solute–solvent attractions may make more negative but does not guarantee if other enthalpic costs exist (1)
Uses cancellation idea: large opposing contributions can yield small net (1)
Concludes claim is not always correct; solubility is sensitive when net is near zero (1)

