AP Syllabus focus: ‘Interactions between dissolved species and solvent can be favorable or unfavorable and contribute to the overall free energy of dissolution.’
Solute–solvent interactions are the “new” attractions formed when a solute disperses into a solvent. Their strength and structure help determine whether dissolution is thermodynamically favourable or unfavourable.
What counts as a solute–solvent interaction?
When a solute dissolves, solvent particles arrange around dissolved solute particles, creating stabilising attractions whose energetic and entropic consequences feed into the overall free energy change for dissolution.
Solvation — the stabilising interactions between solvent molecules and dissolved solute particles; in water, this is called hydration.
Common interaction types (what to look for)
Ion–dipole: between ions (e.g., , ) and polar solvents (especially water)
Hydrogen bonding: between bound to and a lone pair on
Dipole–dipole: between polar molecules (non–hydrogen-bonding cases)
Ion-induced dipole / dipole-induced dipole: ions or polar molecules polarise nearby nonpolar molecules
London dispersion: present in all substances; dominant in nonpolar solutes/solvents
Enthalpy impact: when are interactions “favourable”?
Favourable solute–solvent interactions tend to make more negative because forming strong attractions releases energy. Unfavourable interactions tend to make less negative (or positive) because the formed attractions are weak compared with those present in the separate pure substances.
Key qualitative ideas:
“Like dissolves like” is fundamentally an enthalpy argument: strong interactions form most readily when solute and solvent have compatible polarity and bonding.
Ionic solutes generally require a polar solvent to provide strong ion–dipole attractions; nonpolar solvents cannot stabilise separated ions effectively.
For molecular solutes, increasing polarity and hydrogen-bonding ability typically strengthens solute–solvent attractions in water.
In nonpolar solvents, solutes that are nonpolar and polarizable are stabilised mainly by dispersion forces; strongly polar solutes often interact poorly and remain separate.
Entropy impact: ordering vs dispersal
Solute–solvent interactions do not only change energy; they can change how “free” the particles are to arrange themselves.
Strong, highly directed interactions (especially hydration shells around small, highly charged ions) can impose local ordering of solvent molecules, which can reduce entropy (a more negative contribution to ).
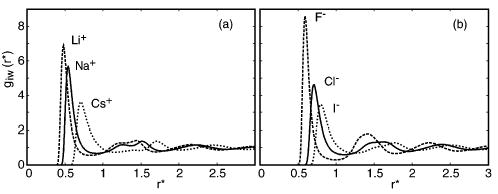
Pair correlation functions quantify how water density varies with distance from an ion, revealing structured hydration shells. Peaks correspond to preferred ion–water distances, and tighter/stronger structuring is typically associated with smaller or higher charge-density ions. This connects the microscopic ordering of solvent molecules to the macroscopic entropy term in dissolution thermodynamics. Source
Weaker, less structured interactions may allow more configurations, giving a less negative (or sometimes positive) entropy contribution.
Whether entropy increases overall depends on competing effects, but for this subtopic the key point is: forming solute–solvent interactions can either increase or decrease entropy depending on how much solvent structure is imposed.
How interactions feed into of dissolution
Solute–solvent interactions contribute to both and , and therefore to the sign and magnitude of .
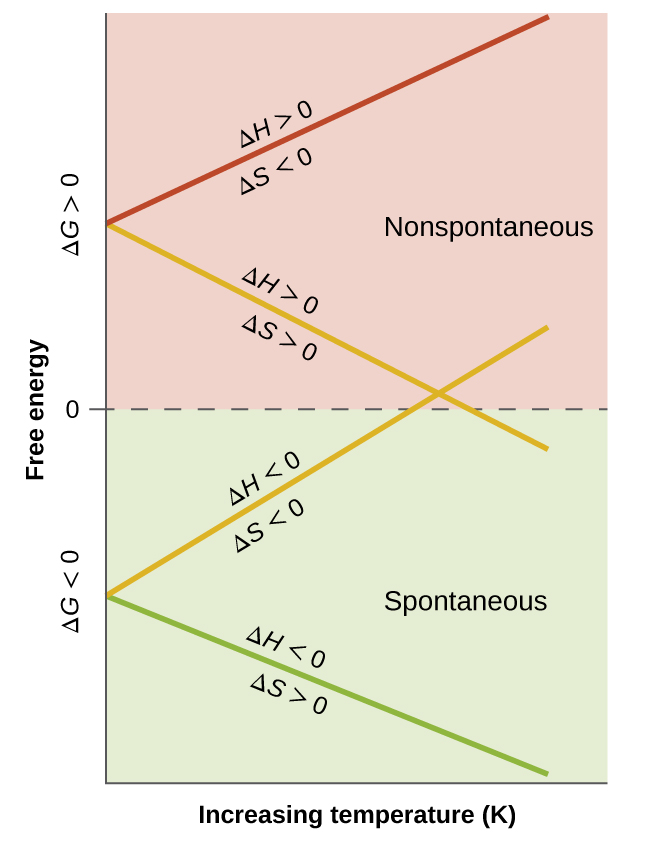
These plots show how changes with temperature for different sign combinations of and . They make clear why a process can be spontaneous only at low or only at high when enthalpy and entropy “compete,” which parallels dissolution cases where strong solvation lowers but solvent ordering lowers . Source
= standard Gibbs free energy change for dissolution (kJ·mol)
= standard enthalpy change for dissolution (kJ·mol)
= temperature (K)
= standard entropy change for dissolution (kJ·mol·K)
A process is more thermodynamically favourable when solute–solvent interactions:
make sufficiently negative (strong stabilisation), and/or
avoid making too negative (minimal ordering penalty).
High-utility reasoning cues (qualitative)
Small, highly charged ions: strong ion–dipole interactions (enthalpy favourable) but often strong solvent ordering (entropy can be less favourable).
Hydrogen-bonding solutes in water: interactions can be strongly favourable and may offset ordering effects.
Nonpolar solutes in water: weak solute–solvent attractions and significant structuring of nearby water can make dissolution unfavourable.
FAQ
Higher charge density (higher charge, smaller radius) strengthens ion–dipole attraction.
This often increases enthalpic stabilisation but can increase solvent ordering around the ion.
Very strong attractions may create rigid, structured solvation shells.
If the entropy penalty (more negative $\Delta S^\circ$) is large enough, $\Delta G^\circ = \Delta H^\circ - T\Delta S^\circ$ can remain unfavourable.
Yes. Protic solvents (e.g., water, alcohols) can donate hydrogen bonds; aprotic solvents (e.g., acetone) cannot.
This changes which solutes receive strong stabilisation and how structured the solvation environment becomes.
In a solvent mixture, a solute may be surrounded more by the component that interacts most favourably with it.
This can shift dissolution thermodynamics without changing the overall bulk solvent composition much.
More polarizable solutes experience stronger induced-dipole and dispersion interactions.
This can improve solubility in nonpolar solvents and sometimes alters how strongly a solute perturbs solvent structure.
Practice Questions
Question 1 (3 marks) Water is added separately to methanol and to hexane. State which liquid is more miscible with water and explain in terms of solute–solvent interactions.
Methanol is more miscible with water (1)
Methanol can form hydrogen bonds / strong dipole-based interactions with water (1)
Hexane is nonpolar and interacts with water mainly via weak London/induced-dipole forces, so interactions are unfavourable (1)
Question 2 (6 marks) Compare the thermodynamic impact of solute–solvent interactions when (i) is placed in water and (ii) is placed in hexane. Discuss how the interactions affect , , and the likely sign of .
In water: strong ion–dipole attractions between ions and water molecules (1)
These interactions make the enthalpy contribution more favourable (more negative relative to weakly interacting solvents) (1)
Hydration can impose ordering of water (hydration shells), making less favourable / potentially negative contribution (1)
In hexane: no significant ion–dipole interactions; solute–solvent interactions are very weak (1)
Therefore is unfavourable (positive or insufficiently negative) (1)
Overall, is much more likely negative in water than in hexane; in hexane it is likely positive (1)

