AP Syllabus focus: ‘Estimate enthalpic and entropic effects for disrupting the interactions that hold the solid together during dissolution.’
Dissolving a solid requires separating its particles before they can mix with the solvent. This “solid-breaking” step has predictable enthalpy and entropy trends that strongly influence whether dissolution is energetically costly.
What “breaking the solid apart” means
When a solid dissolves, one contribution to the overall thermodynamics is the energy and disorder change associated with separating the solid’s particles (ions, molecules, or atoms) from their ordered lattice or network.
Types of solids and what must be disrupted
Ionic solids: separation of oppositely charged ions held by strong electrostatic attractions.
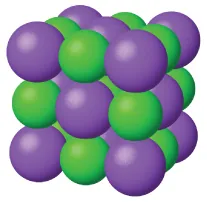
An ionic crystal consists of cations and anions arranged in a repeating, ordered lattice. The alternating charges visually emphasize why separating ions requires energy input: electrostatic attractions are strong at the short ion–ion distances found in the solid. Source
Molecular solids: separation of neutral molecules held by intermolecular forces (IMFs) such as dispersion forces, dipole–dipole forces, and hydrogen bonding.
Covalent-network solids: separation requires breaking covalent bonds; this step is usually so energetically costly that dissolution is negligible.
Enthalpy contribution (ΔH) from disrupting the solid
Breaking the solid’s internal attractions requires an input of energy in almost all cases, so this step is typically endothermic.
Lattice enthalpy (ionic solids) — the enthalpy change associated with separating one mole of an ionic solid into widely separated gaseous ions; it reflects the strength of ionic attractions.
A larger-magnitude lattice enthalpy (stronger attractions) implies a larger enthalpy cost to pull ions apart during the “break the solid” step.
What makes the enthalpy cost larger (more positive)
For ionic solids, stronger ion–ion attractions increase the energy required to separate ions:
Higher ionic charge (e.g., / vs. /) → much stronger attraction → more endothermic to disrupt.
Smaller ionic radii → ions are closer together → stronger attraction → more endothermic to disrupt.
Crystal packing/structure can change attraction strength, but charge and size dominate most AP-level comparisons.
For molecular solids, the enthalpy cost depends on IMF strength:
Strong hydrogen bonding networks (e.g., many donors/acceptors) → more energy required to separate molecules.
Larger, more polarizable molecules may have stronger dispersion forces, increasing the energy needed to separate them.
Interpreting ΔH for this step (not the whole dissolution)
If the solid’s attractions are strong, the solid-breaking contribution to tends to be strongly positive.
A dissolution process can still occur overall, but that requires other contributions not covered on this page.
Entropy contribution (ΔS) from disrupting the solid
Separating particles from a crystal increases the number of accessible arrangements (microstates).
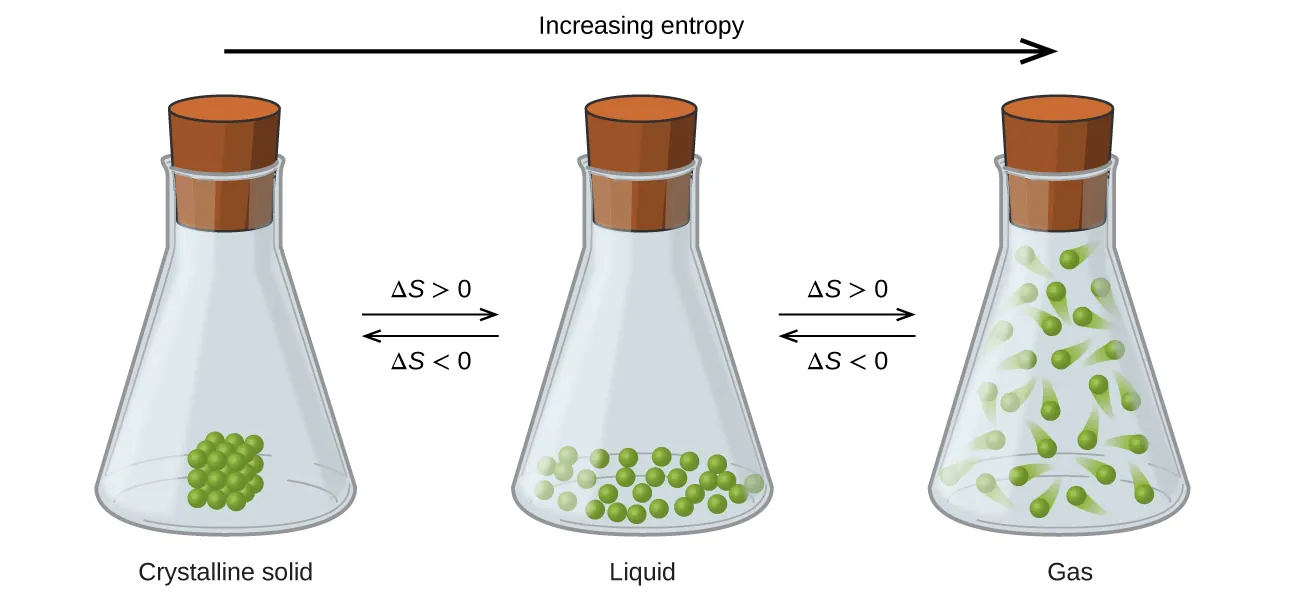
The diagram compares particle arrangements in a crystalline solid, a liquid, and a gas, showing increasing positional freedom across phases. It provides a particle-level rationale for why entropy rises when particles become less constrained, consistent with for separating particles from an ordered crystal. Source
Therefore, the “break the solid” step generally has .
Why ΔS tends to be positive
A crystalline solid is highly ordered: particles occupy fixed positions and have limited freedom.
Once separated (conceptually, before considering solvent effects), particles can occupy many more positions and configurations, increasing dispersal of matter and thus entropy.
When the entropy gain is relatively small vs. large
Ionic solids often show a significant entropy increase upon separation into independent ions (more distinguishable particles and positional possibilities).
Molecular solids may show a smaller entropy increase if the molecules were already somewhat mobile (e.g., softer solids) or if the solid is not highly ordered.
Using these ideas on AP-style qualitative reasoning
When asked to “estimate enthalpic and entropic effects” for breaking the solid apart, focus on the sign and relative magnitude:
Enthalpy (ΔH): usually positive; larger for higher charges, smaller ions, stronger IMFs, or covalent networks.
Entropy (ΔS): usually positive because separation increases disorder and dispersal.
Be careful to attribute these statements to the solid-disruption step, not automatically to the total dissolution process.
FAQ
Measure a thermal property linked to separation, such as enthalpy of fusion or sublimation for molecular solids. For ionic solids, lattice enthalpy is commonly inferred indirectly from thermochemical cycles using measurable formation enthalpies.
Hydrogen bonding can form extensive, directional networks. Disrupting many O–H···O interactions across a crystal can require substantial energy, sometimes comparable to (but typically less than) strongly charged ionic lattices.
No. That description is a conceptual reference state used to discuss separation energetics. It isolates the cost of overcoming solid–solid attractions before considering what the solvent does.
Different polymorphs pack particles differently, changing distances and coordination. Tighter packing and higher coordination generally strengthen attractions, increasing the energy required to separate particles.
Because the “attractions” are covalent bonds throughout the lattice. Separating particles requires bond breaking across an extended network, giving a very large positive enthalpy that typically prevents ordinary dissolution.
Practice Questions
Question 1 (3 marks) A student compares the energy required to disrupt the solids in the following before any interactions with water are considered: NaCl(s) and MgO(s).
(a) State the sign of the enthalpy change for disrupting each solid.
(b) Identify which solid requires more energy to disrupt and justify your choice in terms of ionic charge and/or ionic size.
Correct sign: endothermic / for disrupting NaCl(s) (1)
Correct sign: endothermic / for disrupting MgO(s) (1) (b)
MgO requires more energy due to higher ionic charges (/) and/or smaller ionic radii leading to stronger electrostatic attraction / larger lattice enthalpy (1)
Question 2 (6 marks) Consider three solids: KBr(s), CaF(s), and sucrose(s) (a molecular solid with many O–H groups). For the step “breaking the solid apart” during dissolution:
(a) Predict the relative enthalpy cost of disrupting these solids (rank from smallest to largest) and justify each comparison.
(b) State the expected sign of the entropy change for disrupting a solid and give one reason.
Correct overall ranking with KBr smallest among ionic solids (1)
Justification: KBr has / ions with relatively larger radii → weaker attractions than CaF (1)
CaF higher due to cation and relatively small ions → stronger attractions / larger lattice enthalpy (1)
Sucrose justification: molecular solid; disruption breaks multiple hydrogen bonds/strong IMFs, so can be energetically significant (1) (Allow rankings that place sucrose between KBr and CaF if justified by “strong H-bonding but not ionic-charge level”; full credit requires coherent IMF vs ionic reasoning.) (b)
(1)
Reason: particles become less ordered/more dispersed with more possible arrangements upon separation (1)

