AP Syllabus focus:
‘Explain energy loss between trophic levels using the laws of thermodynamics, including heat loss during energy transformations.’
Energy moves through ecosystems in one direction, changing form at each step. The laws of thermodynamics explain why transfers between trophic levels are inefficient and why much energy leaves food webs as heat.
Thermodynamics as the Rulebook for Energy Flow
Ecological energy flow is constrained by thermodynamics, not by whether organisms “need” more energy.
Thermodynamics: the study of energy transformations, including how energy is conserved and how usable energy is degraded during transfers.
The First Law: Energy Is Conserved, Not Recycled
The first law of thermodynamics explains that ecosystems do not create or destroy energy; they only transform it (for example, chemical energy in biomass to kinetic energy in movement and thermal energy as heat).
First law of thermodynamics: energy cannot be created or destroyed; it can only be transferred or transformed.
This conservation idea is often expressed as an energy “budget” for consumers.
= chemical energy consumed in food (kJ per unit time)
= chemical energy converted to new biomass (kJ per unit time)
= thermal energy released during metabolism (kJ per unit time)
= energy leaving as undigested/excreted material (kJ per unit time)
Because energy is conserved, “missing” energy at higher trophic levels has not vanished; it has mostly been transformed into heat or left the organism as waste.
The Second Law: Every Transfer Degrades Usable Energy
The second law of thermodynamics explains why energy transfers are inefficient: every transformation increases entropy and converts some energy into a less usable form (usually dispersed heat).
This diagram compares particle arrangements to illustrate entropy: ordered packing (lower entropy) versus more dispersed, irregular arrangements (higher entropy). In ecological energy transfers, usable chemical energy is continually converted into more dispersed thermal energy, which is harder to harness for biological work—an application of the second law. Source
Second law of thermodynamics: every energy transfer or transformation increases entropy, so some energy becomes less available to do useful work.
In ecosystems, this means organisms cannot convert all consumed chemical energy into new biomass. Even with abundant food, metabolic processes inevitably release heat.
How Energy Is Lost Between Trophic Levels
A trophic level is an organism’s feeding position (for example, primary consumer, secondary consumer). Moving energy from one trophic level to the next is inefficient for predictable biological reasons:
Heat loss during energy transformations (core thermodynamics link)
Cellular respiration releases heat as organisms extract energy from food to make ATP for maintenance and activity.
Movement, hunting, digestion, circulation, and cellular repair all require work that ultimately dissipates as heat.
In many animals, thermoregulation (maintaining body temperature) increases respiratory heat loss.
Energy never eaten (not transferred at all)
Some biomass is not consumed (predators miss prey; grazers do not eat roots, bark, or tough stems).
Some organisms die without being eaten by the next trophic level, sending energy to decomposers rather than to higher consumers.
Energy eaten but not turned into biomass
Indigestible material (cellulose, lignin, bones, shells) is egested as feces.
Excretion removes energy-containing compounds (for example, urea).
Even digested energy is largely spent on maintenance metabolism, not growth.
Ecological Consequences of Thermodynamic Losses
Because energy is progressively degraded (second law) while total energy is conserved (first law), ecosystems show:
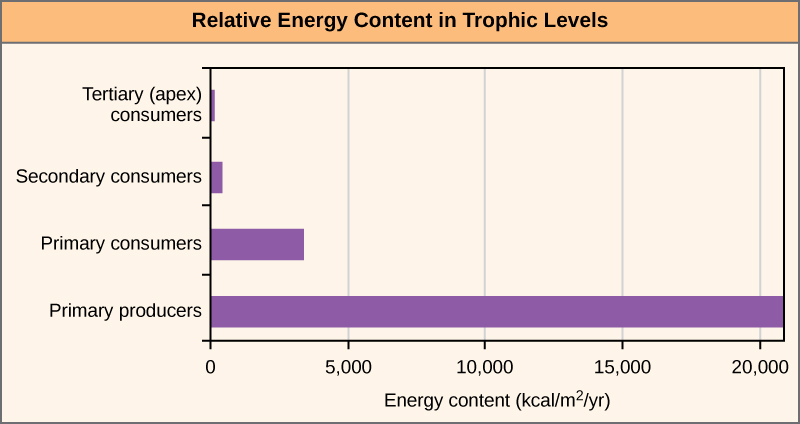
This graph shows the relative energy content available at each trophic level in a real ecosystem (Silver Springs, Florida), with a dramatic decline from primary producers to tertiary consumers. It visualizes why energy pyramids are upright: each transfer leaves much less chemical energy available to support biomass at the next level due to metabolic heat loss and other inefficiencies. Source
Less energy available to support organisms at higher trophic levels.
Shorter food chains than would be possible if transfers were perfectly efficient.
Small populations of top predators, since each step upward depends on a much larger energy base below.
FAQ
By estimating metabolic rate via oxygen consumption or carbon dioxide production, then converting to energy units using respirometry constants.
Direct calorimetry can be used in controlled settings but is less common in field ecology.
Endotherms spend more energy on thermoregulation, increasing respiratory heat loss.
Ectotherms typically allocate a larger fraction of assimilated energy to growth.
No. It remains in the environment as thermal energy, consistent with conservation.
It is simply too dispersed (high entropy) to be captured efficiently by organisms.
Edible biomass is a larger fraction of producer tissue in many aquatic systems, and producers may have less indigestible structural material.
This can reduce losses via egestion and increase the fraction assimilated.
Higher temperatures generally raise metabolic rates, increasing respiration and heat dissipation per unit biomass.
This can reduce the fraction of consumed energy stored as new biomass, especially for ectotherms.
Practice Questions
Explain, using the first and second laws of thermodynamics, why less usable energy is available at higher trophic levels. (2 marks)
First law: energy is conserved/only transformed, not destroyed (1).
Second law: transfers increase entropy so some energy becomes unusable and is released as heat (1).
Describe three distinct pathways by which energy is lost between trophic levels, and link each pathway to thermodynamic principles. (6 marks)
Any three pathways, 1 mark each (max 3):
Heat released during respiration/metabolism.
Heat associated with movement/maintenance/thermoregulation.
Biomass not consumed by the next trophic level.
Indigestible material egested as faeces.
Energy lost in excretion.
Thermodynamic links, up to 3 marks:
First law: energy accounted for as stored in biomass, dissipated as heat, or exported as waste (1).
Second law: transformations are inefficient; entropy increases; heat is less available for biological work (1).
Heat is dispersed to the environment and cannot be fully converted back to biomass (1).

