Particle model, density and temperature
Solids: particles are closely packed, vibrate about fixed positions, and have strong intermolecular forces.
Liquids: particles are close together but move past each other; forces are weaker than in solids.
Gases: particles are far apart, move randomly and rapidly, and have negligible intermolecular forces except during collisions.
Density is mass per unit volume: .
Temperature is measured in kelvin (K) or degrees Celsius (C).
Convert with .
A temperature change has the same numerical value in kelvin and Celsius: .
Kelvin temperature is proportional to the average kinetic energy of particles: .
Higher temperature means greater average random kinetic energy of particles.
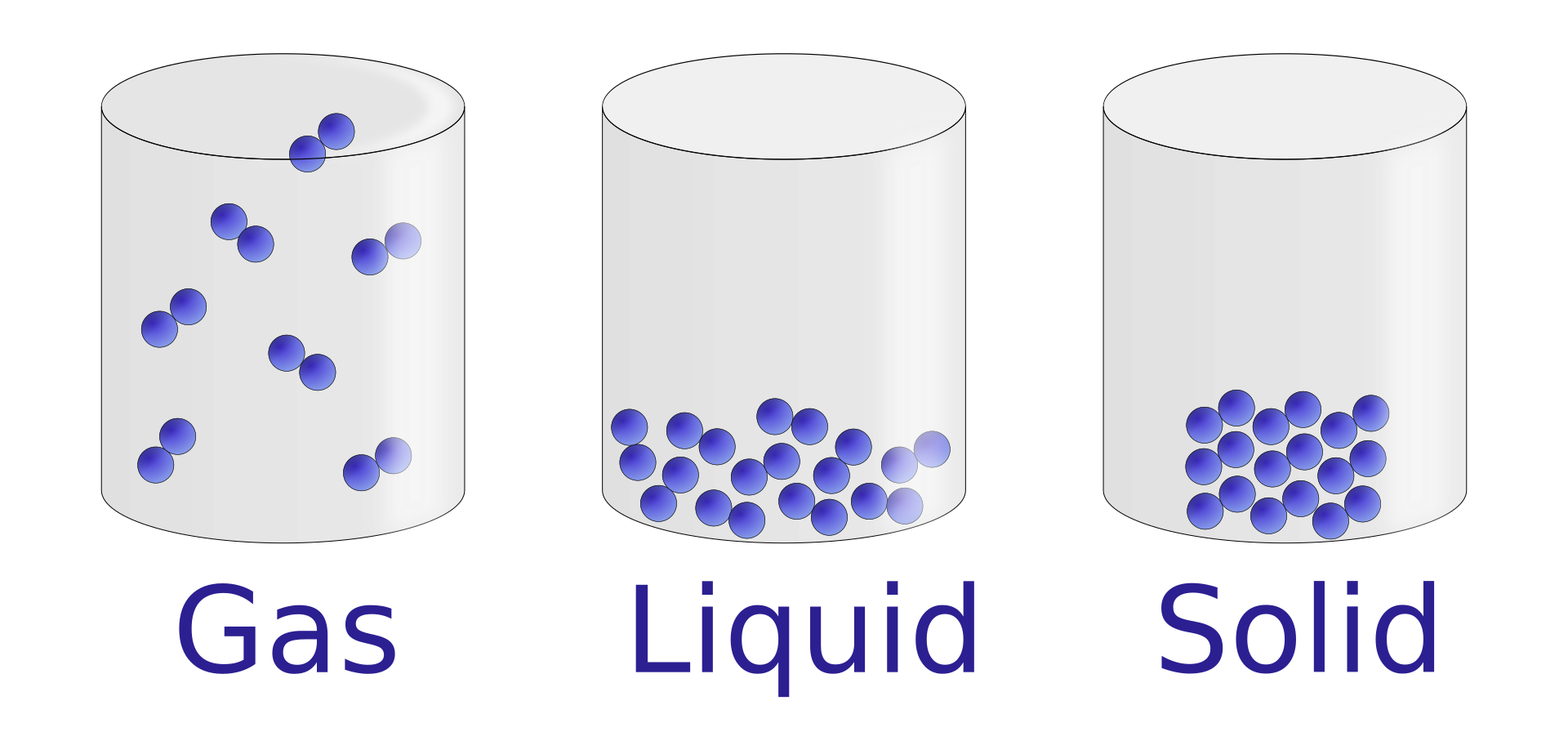
This diagram compares particle spacing and arrangement in a solid, liquid and gas. It is useful for linking the microscopic model to macroscopic properties such as shape, volume and compressibility. Source
Internal energy and phase changes
Internal energy is the total random kinetic energy of molecules plus the total intermolecular potential energy of the system.
If temperature increases, average kinetic energy increases.
If a substance changes state at constant temperature, the added energy changes intermolecular potential energy, not kinetic energy.
During melting and boiling, energy is used to overcome intermolecular forces.
During freezing and condensing, energy is released as intermolecular forces become more significant.
Know these changes of state: melting, freezing, boiling, condensing, evaporation.
Evaporation can happen below boiling point because faster molecules escape from the surface.
A phase change occurs at constant temperature for a pure substance under fixed conditions.
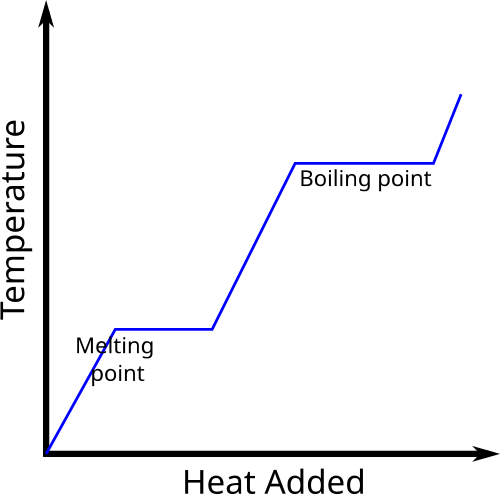
This heating-curve graph shows temperature rising within a phase, then staying constant during phase changes. The flat sections represent energy being used as latent heat rather than to raise temperature. Source
Specific heat capacity and latent heat
Thermal energy transferred by heating without a change of state: .
Specific heat capacity is the energy needed to raise the temperature of 1 kg of a substance by 1 K.
Use when the substance stays in the same phase.
Thermal energy transferred during a phase change: .
Specific latent heat is the energy needed to change the state of 1 kg of a substance at constant temperature.
Specific latent heat of fusion: for melting/freezing.
Specific latent heat of vaporization: for boiling/condensing.
In multi-step problems, split the process into sections and add all energy changes.
Always check whether the question involves temperature change, state change, or both.
Use kg, K or C for temperature change, J, J kg, and J kg K consistently.
Direction of thermal energy transfer
Thermal energy is transferred from higher temperature to lower temperature.
The temperature difference determines the direction of the resultant thermal energy transfer.
Thermal equilibrium is reached when there is no net thermal energy transfer.
In calorimetry-style ideas, energy lost by the hotter body equals energy gained by the colder body if losses to surroundings are negligible.
Conduction and convection
Conduction is thermal energy transfer through a material by particle interactions.
In conduction, faster particles transfer energy to neighbouring particles by collisions/vibrations.
Metals conduct well because of free electrons and lattice vibrations.
Poor conductors are thermal insulators.
Rate of conduction: .
Larger thermal conductivity , larger area , and larger temperature difference increase the rate of transfer.
Greater thickness decreases the rate of transfer.
Convection occurs in fluids because warmer fluid becomes less dense and rises, while cooler fluid becomes more dense and sinks.
Convection transfers energy by the bulk movement of the fluid.
Convection does not occur in solids because the particles cannot flow.
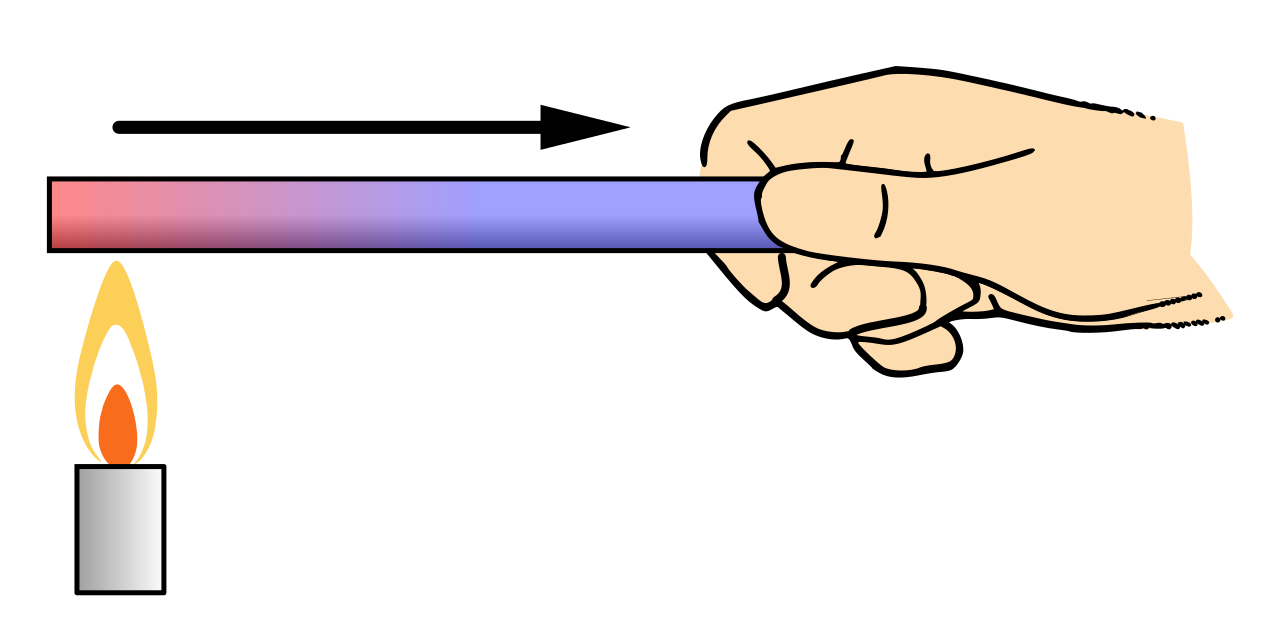
This diagram shows heat conduction along a rod from the hotter end to the colder end. It helps connect the conduction equation to temperature gradient, area, and direction of energy flow. Source
This image shows a convection current driven by density differences in a fluid. Warm fluid rises, cool fluid sinks, and energy is transferred by the resulting circulation. Source
Thermal radiation, black bodies and stars
Thermal radiation is energy transfer by electromagnetic waves, mainly infrared for ordinary temperatures.
Radiation can travel through a vacuum; conduction and convection cannot.
All bodies emit thermal radiation; hotter bodies emit more and at shorter peak wavelengths.
A black body is an ideal emitter and absorber of radiation.
For a black body, luminosity is given by the Stefan–Boltzmann law: .
This means total power emitted depends strongly on absolute temperature.
Apparent brightness is power received per unit area at distance : .
This is an inverse-square law: if distance doubles, brightness becomes one quarter.
Wien’s displacement law: .
As temperature increases, the peak wavelength decreases.
Use the peak wavelength of a black-body spectrum to determine surface temperature of a star.
In exam questions, distinguish clearly between luminosity (total power emitted) and apparent brightness (power per unit area received).
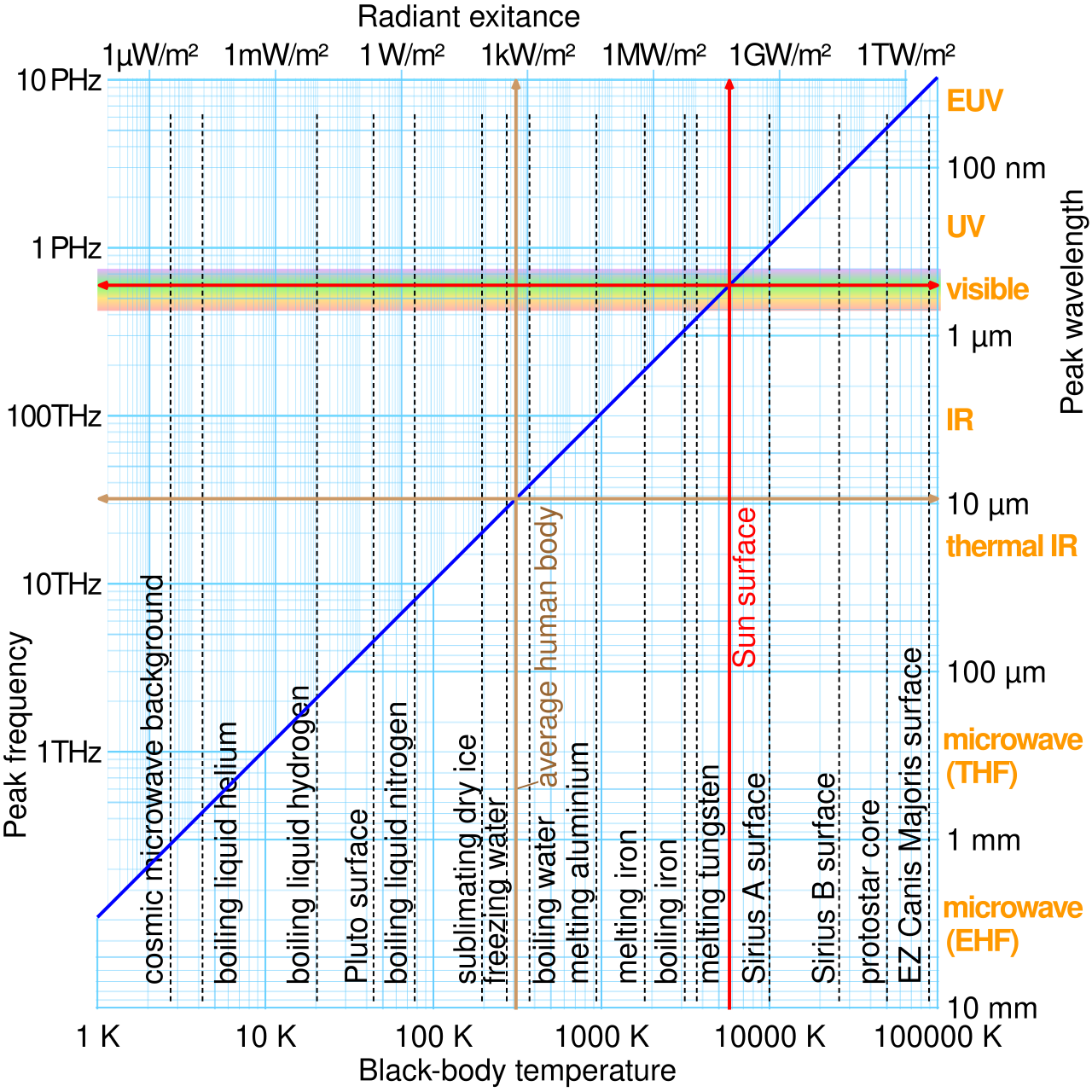
This graph combines Stefan–Boltzmann and Wien’s displacement law ideas. It shows that hotter bodies emit much more power and that their peak wavelength shifts to shorter wavelengths. Source
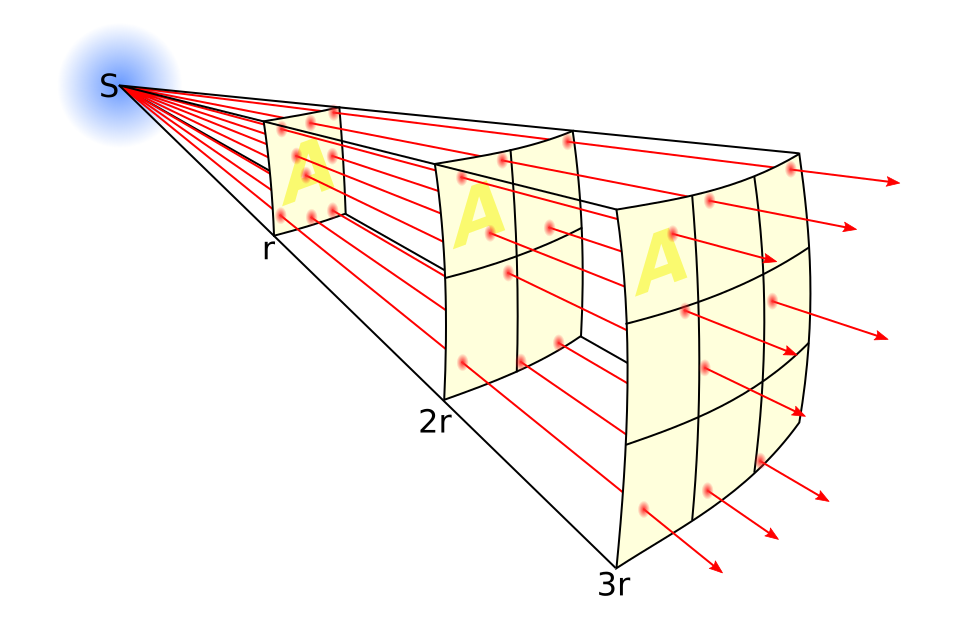
This diagram illustrates why apparent brightness falls with distance as . The same emitted power is spread over a larger spherical area as distance increases. Source
Common exam traps
Do not confuse temperature with internal energy.
Do not use Celsius in Stefan–Boltzmann or Wien’s law; use kelvin.
Do not use during a phase change.
Do not use when temperature is changing within a single phase.
Do not say heat rises; it is the warmer, less dense fluid that rises in convection.
Do not mix up luminosity and apparent brightness.
In conduction questions, remember the role of , , and temperature gradient.
Checklist: can you do this?
Explain solids, liquids and gases using the particle model.
Calculate density, specific heat problems, and latent heat problems with correct units.
Identify whether a situation involves conduction, convection, or radiation.
Apply Stefan–Boltzmann law, Wien’s displacement law, and apparent brightness correctly.
Interpret a heating curve or black-body spectrum and state what is happening to particle energy.