Core quantities and constants
Pressure: , where force acts perpendicular to the surface.
Amount of substance: .
Avogadro constant: .
Use kelvin in all gas-law equations: .
Know the two forms of the ideal gas equation: and .
Universal gas constant: .
Boltzmann constant: .
Empirical gas laws and the combined law
Boyle’s law: for constant temperature, , so .
Charles’s law: for constant pressure, , so .
Pressure law / Gay-Lussac’s law: for constant volume, , so .
Combining the three gives for a fixed amount of gas.
For the same sample of gas between two states: .
In exam questions, first identify which variable is constant before choosing the equation.
On a – diagram, an isothermal change follows a curved inverse relation; a constant-volume change is vertical; a constant-pressure change is horizontal.
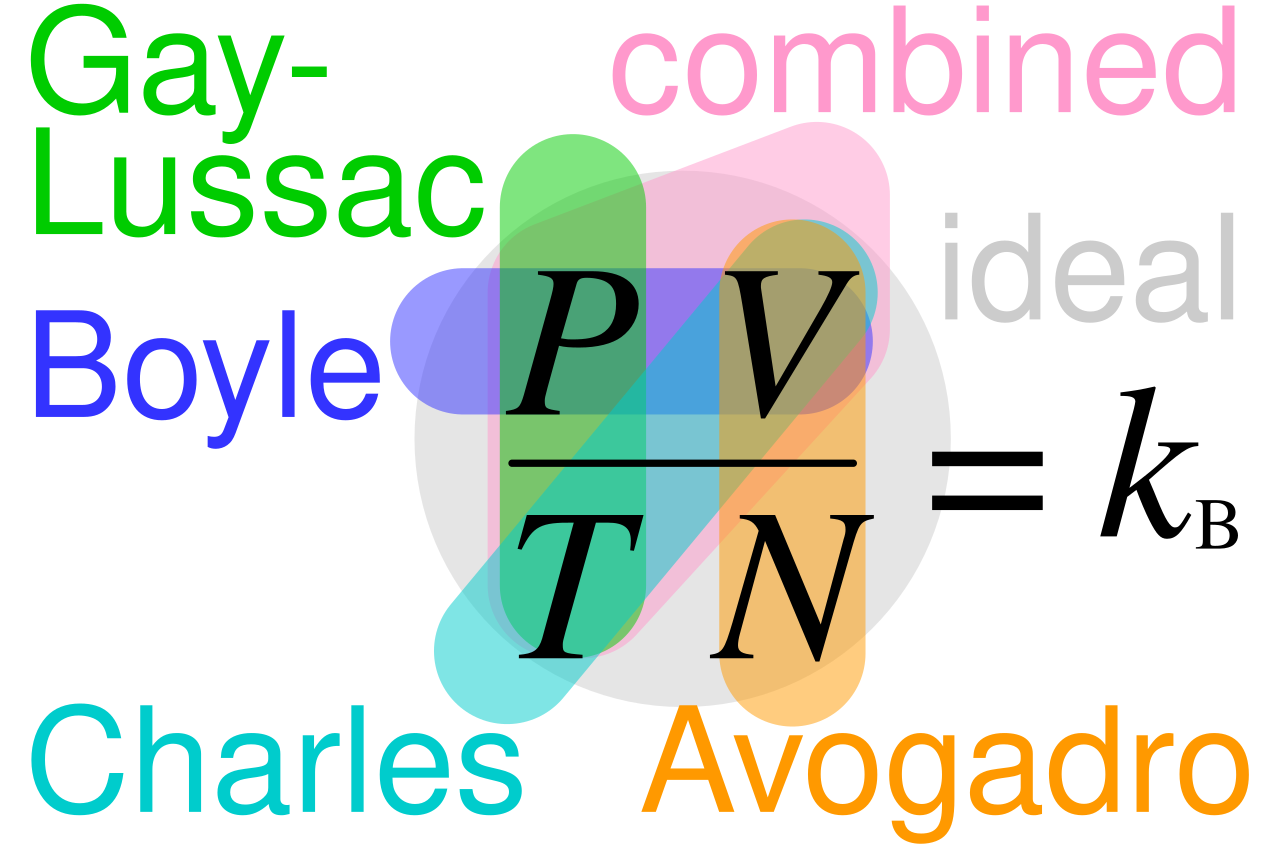
This diagram links Boyle’s law, Charles’s law, Gay-Lussac’s law, Avogadro’s law, the combined gas law, and the ideal gas law. It is useful for seeing which quantities are held constant in each relationship. It works well as a quick revision map before exam questions. Source
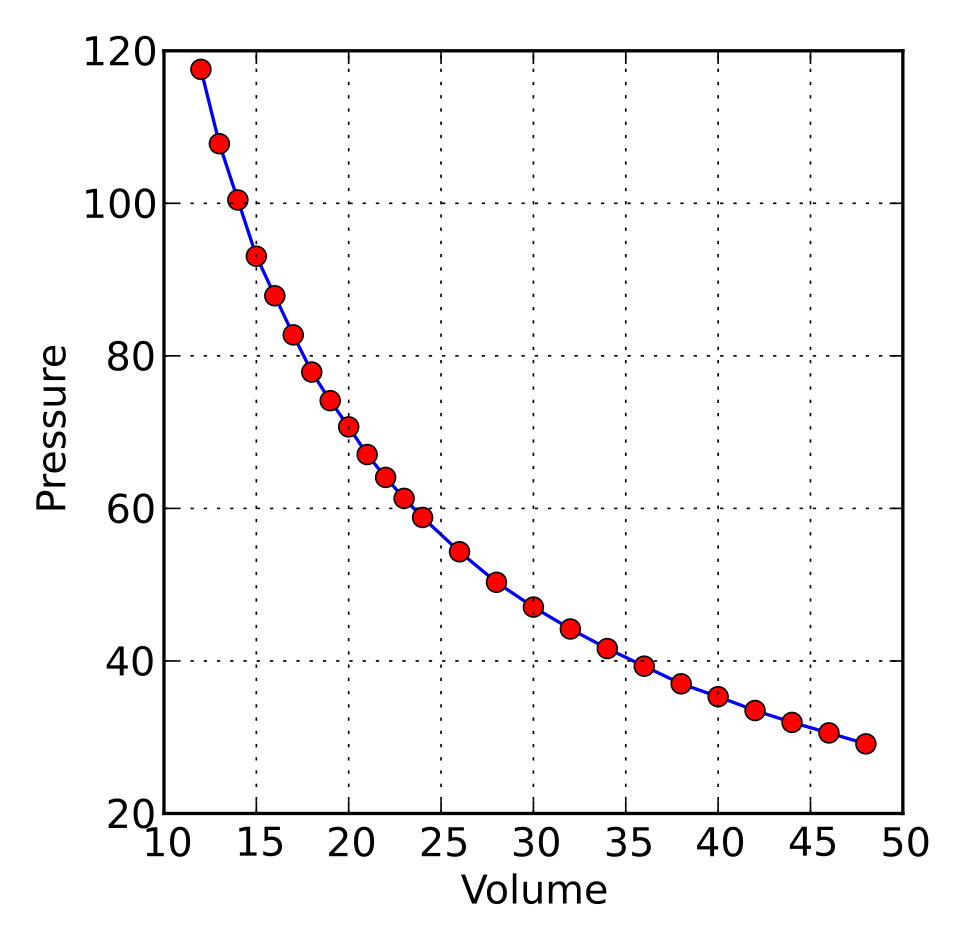
This graph shows the inverse relationship between pressure and volume for Boyle’s law. It is useful for linking the algebraic statement to the curved shape seen on graphs. Students should connect this to isothermal changes of a fixed mass of gas. Source
Ideal gas law: what it means
Ideal gas = a model used to approximate real gases.
The model treats gas particles as tiny point particles in random motion.
Particles have negligible volume compared with the container volume.
There are no intermolecular forces except during collisions.
Collisions between particles and with walls are perfectly elastic.
The ideal gas law can be written as or .
These two forms are equivalent because and .
Use when the question gives moles; use when it gives number of molecules.

This figure shows gas particles widely separated in a container, helping explain why an ideal gas can be modelled as particles with negligible size and negligible intermolecular forces. It supports the microscopic assumptions behind the ideal gas law. It is especially helpful when comparing ideal and real gas behaviour. Source
Pressure from molecular collisions
Gas pressure arises from the change in momentum of particles colliding with container walls.
More frequent collisions or harder collisions give a greater pressure.
The kinetic-theory result is , often written as .
Here is the gas density and means the mean square translational speed.
This shows pressure depends on both how much mass is packed into the volume and how fast the molecules are moving.
If temperature increases, average molecular kinetic energy rises, so collisions become more forceful.
At constant volume, this leads to higher pressure.
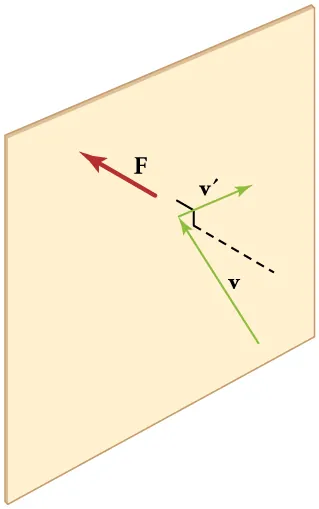
This diagram shows a gas molecule colliding with a wall and reversing its momentum component perpendicular to the surface. It helps explain how repeated molecular collisions produce pressure. This is the key microscopic picture behind the kinetic-theory expression for pressure. Source
Temperature and internal energy
For an ideal gas, temperature is linked to the average translational kinetic energy of molecules.
For a monatomic ideal gas, the internal energy is only the random kinetic energy of the molecules.
Required equations: and .
So for a monatomic ideal gas, internal energy depends only on temperature.
If temperature stays constant, internal energy stays constant.
If the number of molecules doubles at the same temperature, internal energy doubles.
Do not apply these equations to gases unless the question specifies ideal monatomic gas.
Ideal gas vs real gas
A real gas differs from an ideal gas because molecules have finite volume and can exert intermolecular forces on each other.
The ideal gas approximation works best at high temperature, low pressure, and low density.
Under these conditions, particles are far apart and interactions are less important.
Real gases deviate more from ideal behaviour at low temperature and high pressure.
In explanations, connect this to particles being closer together, so their size and intermolecular attraction matter more.
If the question asks when the model is valid, state the conditions and the particle-level reason.
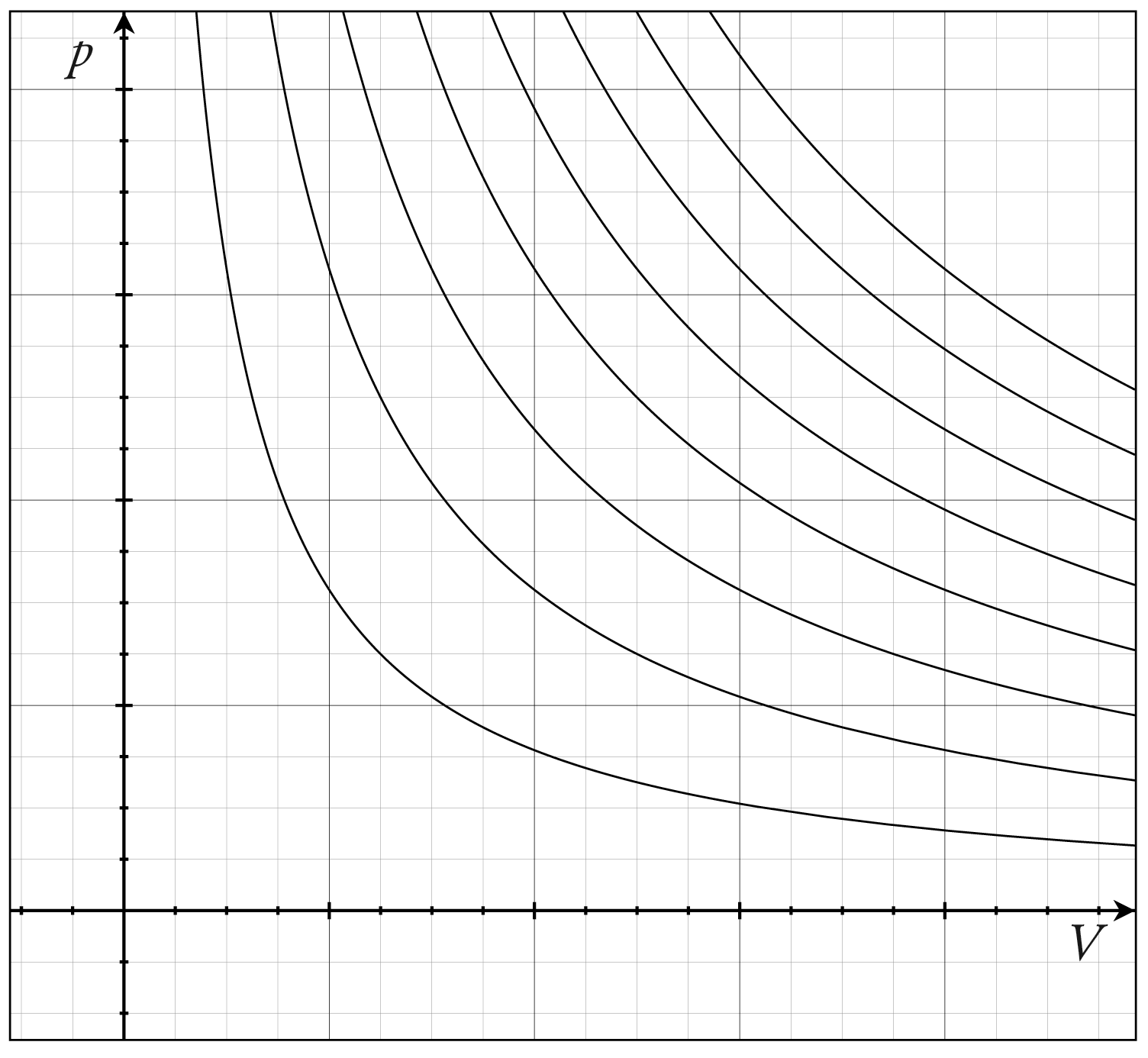
This image shows several ideal-gas isotherms on a – diagram. It helps students recognise how pressure changes with volume for different constant temperatures. It is useful for interpreting qualitative graph questions and linking them to Boyle’s law. Source
Exam traps and strategy
Always convert temperature to kelvin before substituting into gas equations.
Check whether the gas amount is given as or before choosing or .
Distinguish carefully between microscopic explanations (molecules, collisions, momentum) and macroscopic quantities ().
In proportionality questions, decide first which variables are constant.
In “explain” questions, mention collision frequency, momentum change, and molecular kinetic energy where relevant.
For graph questions, describe both the shape and the constant variable.
Remember that the syllabus limits gas laws to constant volume, constant temperature, constant pressure, and the ideal gas law.
Checklist: can you do this?
State and use Boyle’s law, Charles’s law, the pressure law, and the ideal gas law.
Convert between number of molecules and moles using .
Explain pressure microscopically using molecular collisions and change in momentum.
Use and interpret or for a monatomic ideal gas.
Identify when an ideal gas is a good approximation and justify it using particle spacing and intermolecular forces.