Energy balance and big picture
Earth’s temperature is set by energy balance: incoming solar power = outgoing thermal (infrared) power at equilibrium.
The natural greenhouse effect is essential because it keeps Earth warm enough for life.
The enhanced greenhouse effect is the extra warming caused by human activity increasing greenhouse gas concentrations.
In exam questions, treat the Earth–atmosphere system using conservation of energy.
A planet warms if incoming absorbed power > outgoing emitted power and cools if the reverse is true.
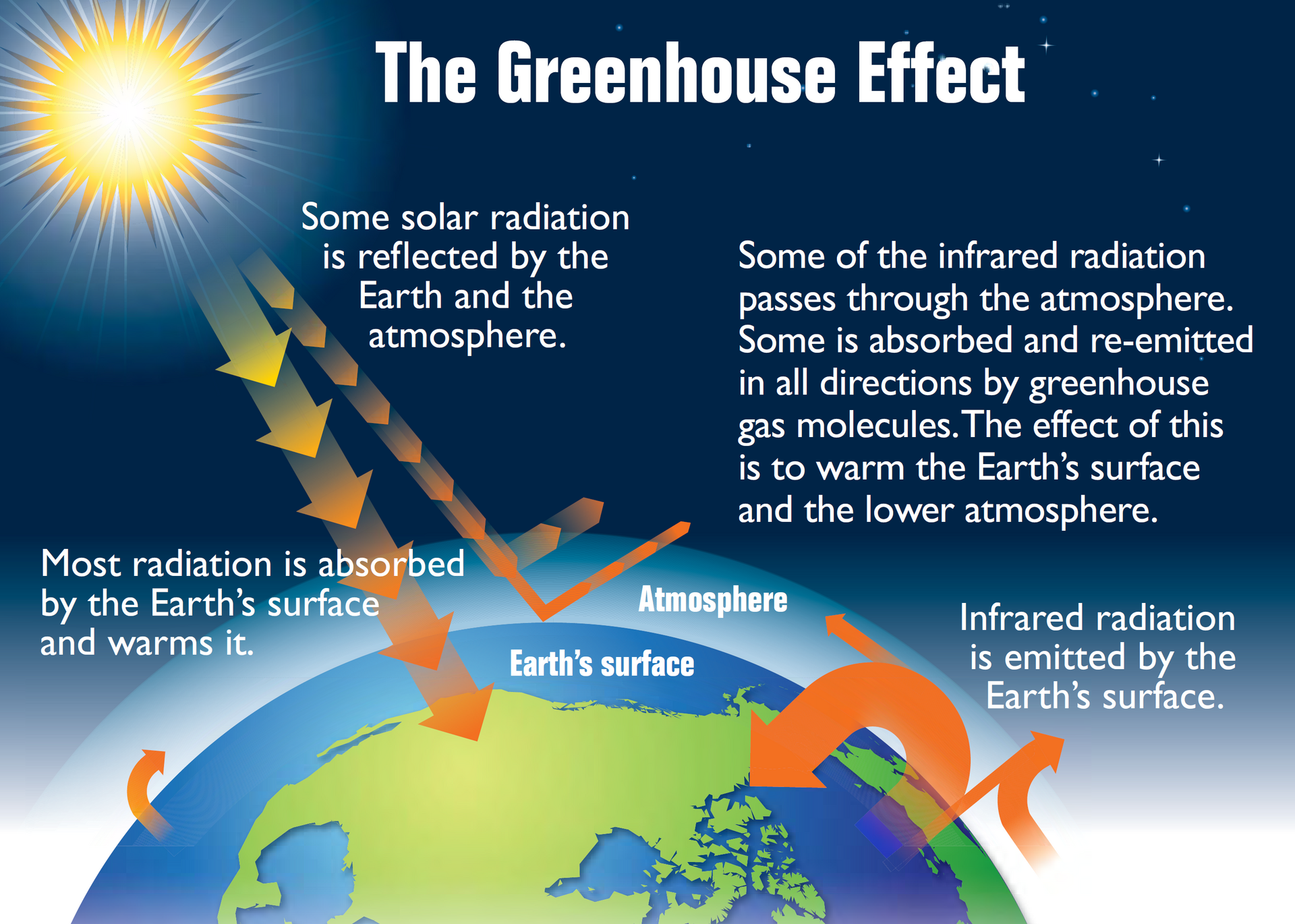
This diagram shows the basic greenhouse effect: incoming solar radiation, reflection, Earth’s infrared emission, and re-emission by greenhouse gases. It is ideal for linking the natural greenhouse effect to Earth’s energy balance. Use it to explain why some outgoing infrared escapes while some is sent back toward the surface. Source
Key quantities and equations
Emissivity measures how effectively a real surface radiates compared with an ideal black body at the same temperature: .
Albedo is the fraction of incident power that is reflected/scattered: .
Solar constant is the solar power per unit area received at the top of the atmosphere on a surface perpendicular to the Sun’s rays.
Because Earth is a sphere but intercepts sunlight over a projected circular area, the mean incoming intensity is .
For absorbed solar intensity, a common model is , where is albedo.
For emitted thermal intensity from a surface, use .
At simple radiative equilibrium for a surface or planet: absorbed intensity = emitted intensity.
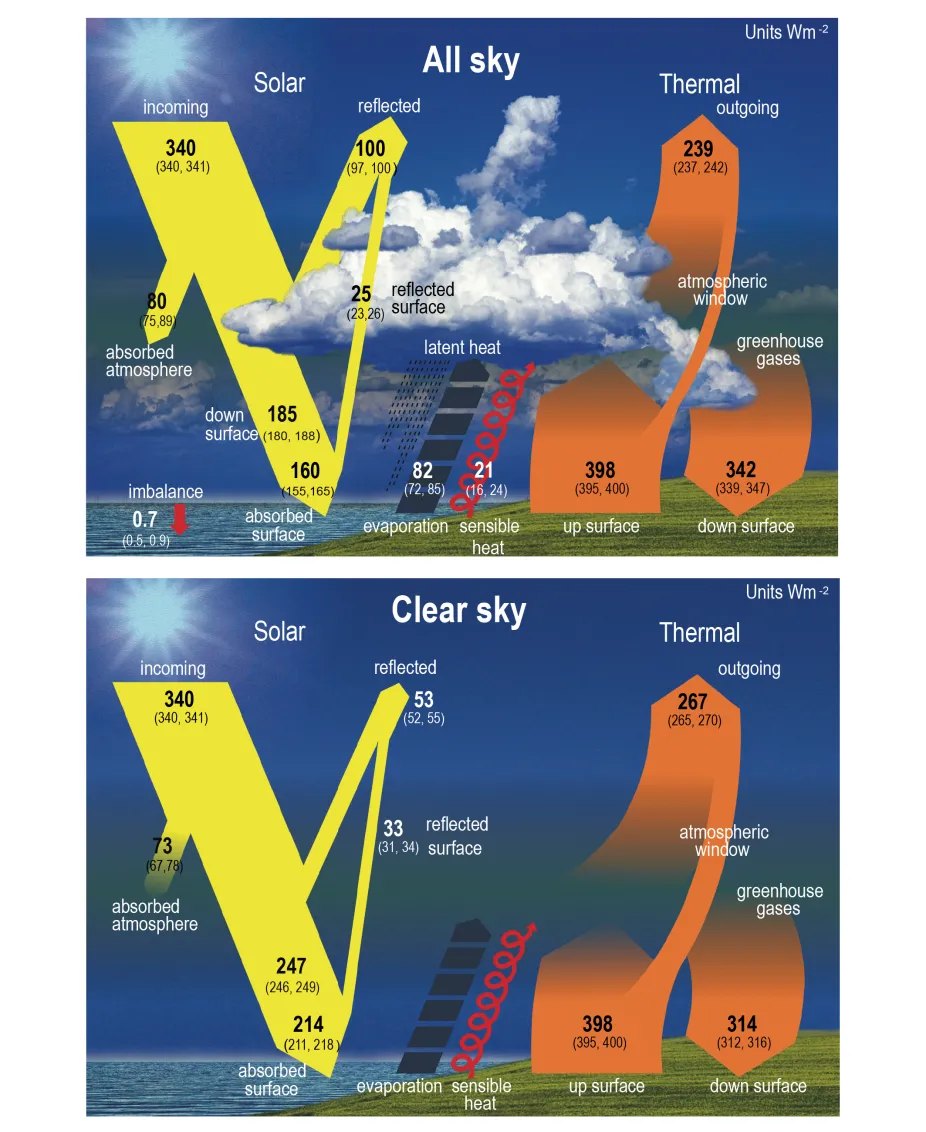
This image shows how incoming solar radiation is reflected, absorbed, and re-emitted by Earth’s surface, clouds, and atmosphere. It is useful for visualising where albedo and emissivity enter energy-balance calculations. The labeled values help connect the diagram to exam-style quantitative reasoning. Source
Albedo and its importance
High albedo surfaces reflect more solar radiation, so they absorb less energy and tend to be cooler.
Low albedo surfaces absorb more solar radiation, so they tend to be warmer.
Earth’s albedo varies daily and depends strongly on cloud cover and latitude.
Clouds, ice, and snow usually increase albedo; oceans and dark land usually lower albedo.
In calculations, a larger albedo reduces absorbed solar power.
In explanations, connect albedo changes to climate feedbacks: lower ice cover can lower albedo and increase warming.
This resource explains albedo using contrasting surface types such as forests, oceans, snow, and ice. It helps show why reflective surfaces reduce absorbed solar energy. Use it to support explanations of why cloud cover and latitude affect Earth’s average albedo. Source
How greenhouse gases warm the atmosphere
The main greenhouse gases named in the syllabus are water vapour , carbon dioxide , methane , and nitrous oxide .
These gases have both natural sources and human-made sources.
Earth’s surface absorbs solar energy and then emits mainly infrared radiation.
Greenhouse gas molecules absorb infrared radiation because their molecular energy levels match certain IR energies.
After absorption, they re-emit radiation in all directions.
This means some radiation is emitted back toward Earth’s surface/lower atmosphere, reducing the rate at which energy escapes to space.
The greenhouse effect can be explained using both a resonance model and molecular energy levels.
In exams, state clearly that greenhouse gases do not simply “trap” heat like a lid; they absorb and re-emit infrared radiation.

This visual shows a carbon dioxide molecule absorbing infrared radiation and then re-emitting it. It is directly relevant to the syllabus statement about molecular energy levels and emission in all directions. Use it to explain why greenhouse gases affect outgoing infrared more than incoming visible sunlight. Source
Natural vs enhanced greenhouse effect
The natural greenhouse effect is a normal atmospheric process and is necessary for habitability.
The enhanced greenhouse effect is the increase in this effect due to human activities.
The syllabus highlights burning fossil fuels as a primary cause of the enhanced greenhouse effect.
Human activity increases atmospheric concentrations of gases such as , , and .
More greenhouse gas molecules means a greater chance that outgoing infrared radiation is absorbed and re-emitted before escaping to space.
In long-answer questions, distinguish carefully between global warming and the mechanism causing it: the mechanism is altered radiative balance.
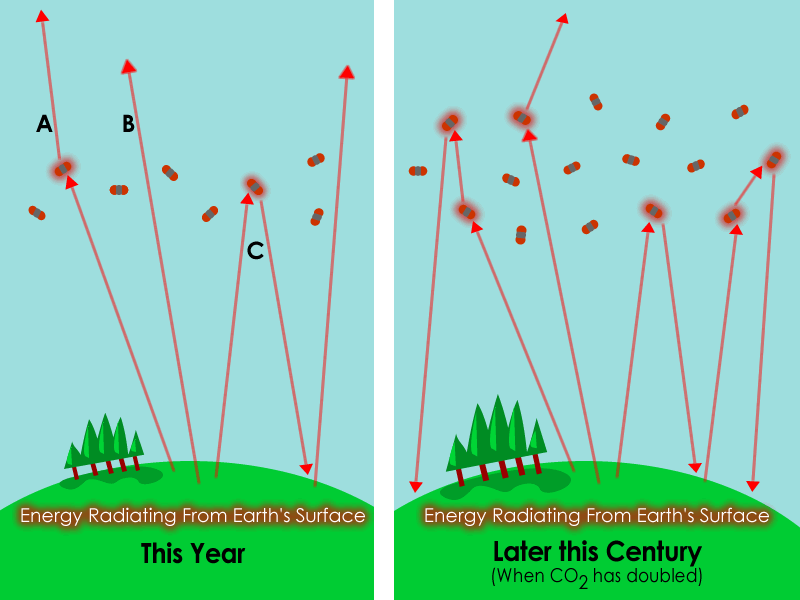
This figure compares the present greenhouse effect with a future atmosphere containing more carbon dioxide. It clearly shows more outgoing infrared being absorbed and re-emitted back downward. It is especially useful for explaining the difference between the natural and enhanced greenhouse effect. Source
Exam-style energy balance method
Identify the system first: planet only, surface, atmosphere, or surface + atmosphere.
Write the relevant incoming power/intensity terms and outgoing power/intensity terms.
Include albedo when some incoming radiation is reflected.
Include emissivity when a surface is not an ideal black body.
Use , not , when working with the global mean incoming intensity for a spherical planet.
At equilibrium, set total absorbed = total emitted and solve for .
Check whether the question refers to surface temperature or planetary effective temperature.
State units carefully: intensity in , temperature in K.
Common mistakes to avoid
Confusing albedo with emissivity.
Forgetting the factor of 4 when averaging the solar constant over the whole planet.
Forgetting the factor when some incoming radiation is reflected.
Using inside Stefan–Boltzmann expressions instead of K.
Saying greenhouse gases absorb mainly visible light rather than infrared.
Describing the greenhouse effect only qualitatively when the question requires an energy-balance equation.
Checklist: can you do this?
Define emissivity, albedo, and the solar constant correctly.
Explain how greenhouse gases absorb infrared using molecular energy levels/resonance and then re-emit in all directions.
Distinguish between the natural and enhanced greenhouse effect.
Apply energy balance using in simple equilibrium models.
Interpret whether a change in albedo, emissivity, or greenhouse gas concentration makes Earth warmer or cooler.