AP Syllabus focus:
‘Hydrocarbon tails of phospholipids block most ions and polar molecules; only small uncharged polar molecules like water or ammonia cross in limited amounts.’
Cell membranes must regulate what enters and leaves the cell. The phospholipid bilayer’s chemistry creates strong limits on the movement of ions and most polar molecules, shaping cellular homeostasis and transport needs.
The key barrier: the phospholipid bilayer interior
Hydrophobic core and selective permeability
Phospholipids form a bilayer with hydrophobic fatty acid (hydrocarbon) tails facing inward.
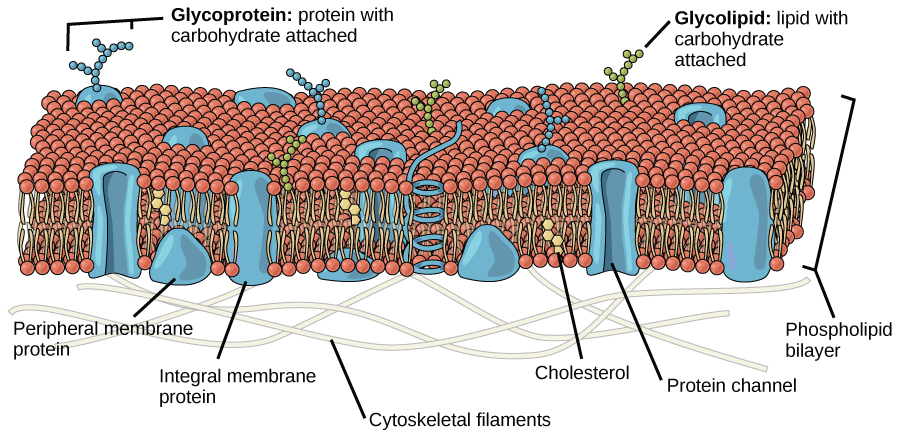
Diagram of the fluid mosaic model of the plasma membrane, highlighting the phospholipid bilayer (hydrophilic heads outward, hydrophobic tails inward) and major membrane components. The labeled protein channel reinforces why ions/polar solutes typically require protein pathways rather than diffusing through the hydrophobic core. Source
This creates a nonpolar interior that strongly resists substances that are charged or strongly polar.
Hydrophobic core: The nonpolar interior of a phospholipid bilayer formed by hydrocarbon tails, which repels charged and polar substances and limits their direct diffusion across the membrane.
This directly reflects the syllabus focus: hydrocarbon tails of phospholipids block most ions and polar molecules.
Why ions are blocked
Ions (e.g., , , , ) are charged, so they interact strongly with water molecules. In aqueous environments, ions are typically surrounded by a hydration shell (water molecules oriented around the charge). Crossing the membrane would require the ion to shed much of this stabilising water layer and move into a nonpolar region—an energetically unfavourable change.
Key ideas for AP Biology:
Charge is the strongest barrier: even very small ions diffuse extremely poorly through the hydrophobic core.
Hydration shells increase the “effective size” of an ion, further reducing permeability.
The bilayer interior lacks partial charges to stabilise ions, so ions are excluded.
Why most polar molecules are blocked
Polar molecules have partial charges and can form hydrogen bonds with water. The membrane interior cannot readily replace these favourable interactions, so diffusion across the hydrophobic core is limited.
Polar molecules that are typically blocked by the bilayer include:
Many sugars (e.g., glucose)
Many amino acids
Many other metabolites with multiple hydroxyl (), amino (), or carboxyl () groups
Even if uncharged overall, a molecule with many polar groups is still strongly excluded because it is not lipid-soluble.
What can cross (in limited amounts)
Small uncharged polar molecules
The syllabus specifies that only small uncharged polar molecules like water or ammonia cross in limited amounts.
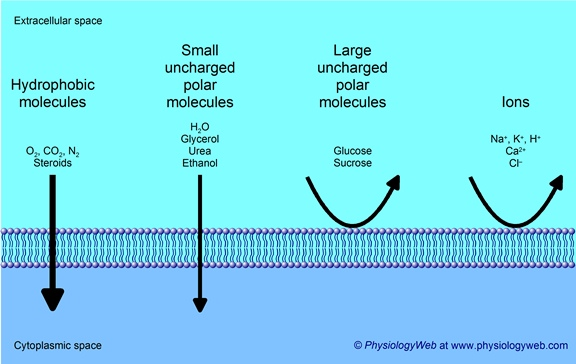
Permeability schematic for a pure lipid bilayer comparing diffusion of hydrophobic molecules, small uncharged polar molecules (e.g., ), large uncharged polar molecules (e.g., glucose), and ions. The figure emphasizes that the hydrophobic membrane interior strongly excludes ions and large polar solutes while allowing only limited passive passage of small uncharged polar molecules. Source
These molecules are polar, but small enough that occasional passage through the bilayer can occur.
Important examples:
Water (): crosses slowly because it is small; however, its polarity still makes the bilayer a significant barrier, so movement is limited without specialised pathways.
Ammonia (): small and uncharged, so it can diffuse across more readily than ions, though still not as freely as nonpolar molecules.
What “limited amounts” means biologically
“Limited” indicates that:
Diffusion is much slower than for small nonpolar molecules (which dissolve in the lipid core more easily).
The rate may be insufficient for many cellular needs, so cells often depend on additional mechanisms (covered elsewhere) to move ions and polar solutes efficiently.
Changes in membrane composition (e.g., lipid saturation and cholesterol content) can alter bilayer fluidity and slightly shift permeability, but the hydrophobic core remains a major constraint.
Factors that determine permeability of ions and polar molecules
When predicting whether an ion or polar molecule can cross the bilayer directly, prioritise:
Charge: any net charge (ions) makes diffusion across the core extremely unlikely.
Polarity and hydrogen bonding capacity: more polar groups → lower permeability.
Size: smaller uncharged polar molecules can cross slightly; large polar molecules are effectively blocked.
Lipid solubility: low lipid solubility correlates with poor passage through the hydrophobic core.
FAQ
More unsaturated tails increase fluidity and can slightly increase permeability to small uncharged polar molecules.
Higher cholesterol can reduce permeability by restricting phospholipid movement, especially at moderate temperatures, though the hydrophobic core still limits ions.
Water molecules orient around an ion’s charge, forming a stabilising layer.
This cluster behaves like a bigger particle and must be disrupted for the ion to enter the hydrophobic core, increasing the energetic cost of crossing.
Yes. Many uncharged molecules are highly polar overall (e.g., with multiple $-OH$ groups).
Strong polarity and hydrogen bonding with water keep them from dissolving in the lipid core, so they remain effectively excluded.
$NH_3$ is uncharged and can partition into the lipid core more easily.
$NH_4^+$ is charged, carries a hydration shell, and faces the same strong barrier as other ions.
One approach uses artificial lipid vesicles (liposomes) loaded with a solute and tracks net movement indirectly via:
Osmotic swelling/shrinking (light scattering)
Changes in internal pH indicators (for weak bases like ammonia)
Practice Questions
Explain why ions cannot readily diffuse directly through the phospholipid bilayer. (2 marks)
Mentions the bilayer interior is hydrophobic/nonpolar due to hydrocarbon tails (1).
Explains charge/hydration shell makes entry into the hydrophobic core energetically unfavourable (1).
Describe how the structure of the phospholipid bilayer limits the movement of polar substances, and identify which polar molecules can cross in limited amounts. (5 marks)
States hydrocarbon tails form a hydrophobic core that excludes polar/charged substances (1).
Explains ions are blocked due to charge and/or hydration shell (1).
Explains most polar molecules are blocked because they rely on hydrogen bonding with water and are not lipid-soluble (1).
Identifies small uncharged polar molecules as exceptions (1).
Gives an appropriate example such as water or ammonia crossing in limited amounts (1).

