AP Syllabus focus:
‘Plasma membranes separate internal and external environments; their hydrophobic interior is the basis of selective permeability.’
Cells rely on the plasma membrane to maintain a stable internal environment while still exchanging matter and information. This depends on a simple physical principle: a water-repelling membrane core makes some substances cross easily and others not.
Plasma membranes as barriers
The plasma membrane forms the boundary between the cytosol and the extracellular environment.
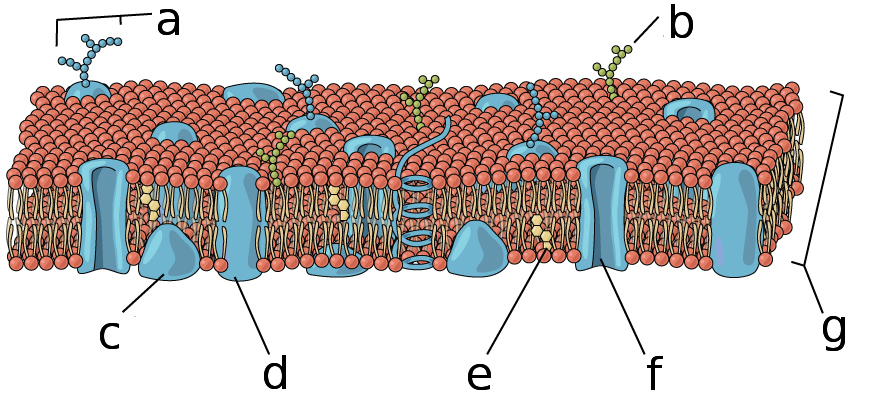
This labeled plasma membrane diagram highlights the phospholipid bilayer plus embedded proteins, cholesterol, and carbohydrate chains. It helps reinforce that selective permeability is rooted in the lipid core, while membrane proteins provide specialized pathways for substances that cannot pass through the hydrophobic interior. Source
Its most important functional property is selective permeability, meaning the membrane is not equally permeable to all substances. This allows cells to:
Keep essential solutes at useful concentrations
Exclude harmful or disruptive substances
Establish internal conditions (e.g., pH and ion composition) compatible with enzyme function
Regulate what enters and leaves, rather than equilibrating with the surroundings
Selective permeability is fundamentally a consequence of the membrane’s chemical architecture, especially its hydrophobic interior.
Selective permeability: The property of a membrane that allows some substances to cross more easily than others, enabling the cell to maintain different internal and external compositions.
The hydrophobic interior: why it matters
Amphipathic phospholipids create a nonpolar core
Membranes are built primarily from phospholipids, which are amphipathic (having both water-attracting and water-repelling regions):
Hydrophilic heads interact with water on either side of the membrane
Hydrophobic fatty acid tails cluster together away from water, forming a nonpolar interior
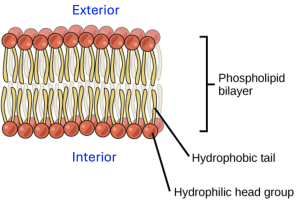
This diagram shows a phospholipid bilayer with hydrophilic head groups facing the aqueous environments and hydrophobic fatty acid tails oriented inward. The tail-to-tail arrangement creates the membrane’s nonpolar core, which underlies selective permeability by excluding most polar and charged solutes. Source
This tail-to-tail region behaves like a thin layer of oil. Because many biologically important molecules are polar or charged (and therefore interact strongly with water), moving them into the nonpolar core is energetically unfavourable. As a result, the hydrophobic interior acts as the main barrier to transport.
Permeability depends on polarity, charge, and size
Crossing the membrane requires a molecule to enter (at least briefly) the hydrophobic core. The more strongly a substance is hydrated in water (typical of polar and charged solutes), the less likely it is to partition into the membrane interior.
Key determinants of permeability:
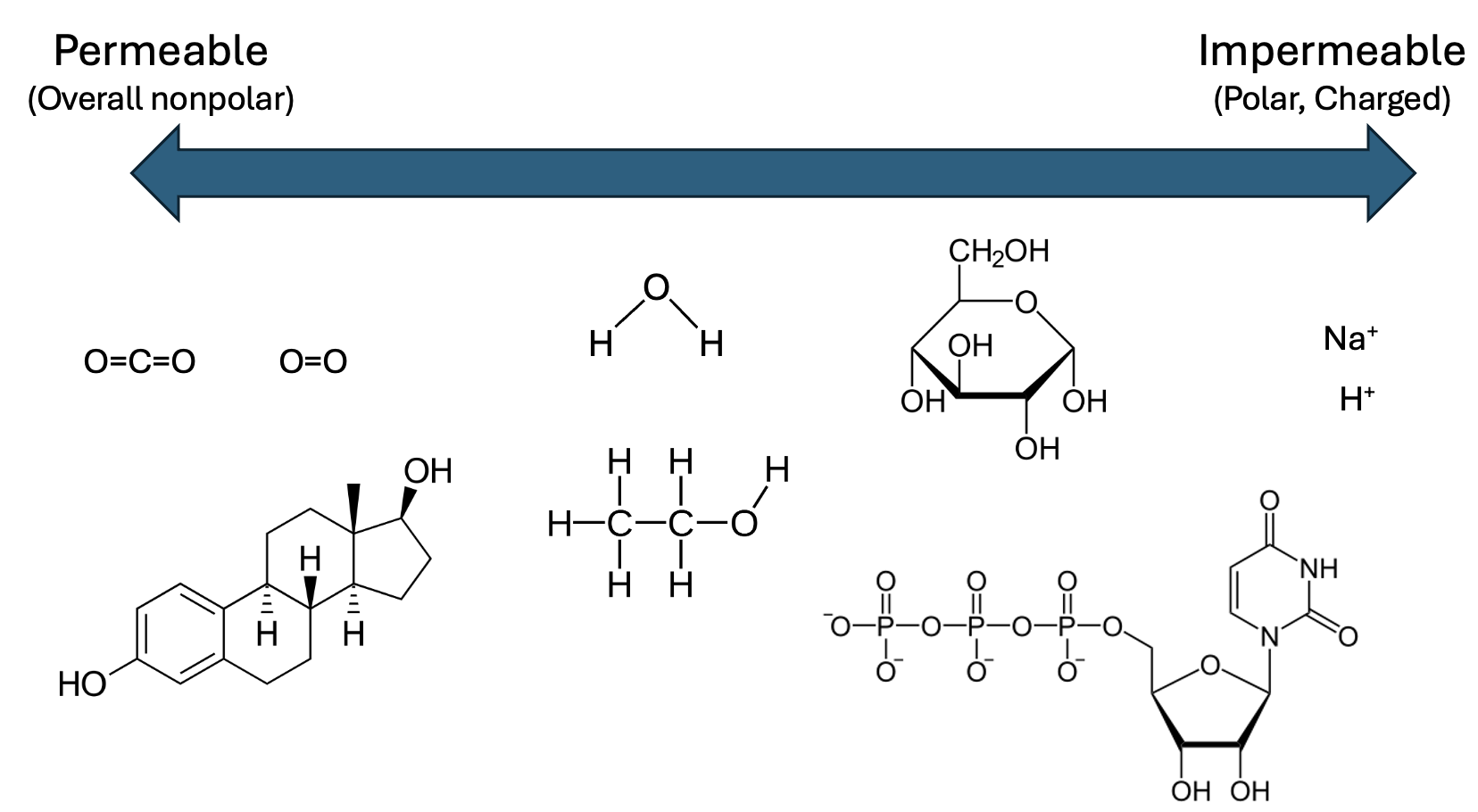
This figure summarizes how chemical properties predict membrane permeability: overall nonpolar molecules cross most readily, while polar and especially charged species are effectively blocked by the hydrophobic core. The examples (e.g., , , , glucose, and ions like ) illustrate why polarity/charge is usually a stronger barrier than size alone. Source
Nonpolarity: more nonpolar = more permeable
Charge: any ion (positive or negative) is strongly excluded
Size: smaller molecules generally cross more readily than larger ones, but polarity/charge is usually the dominant factor
What can and cannot cross the hydrophobic core
Substances that cross relatively easily
These molecules can dissolve in, and diffuse through, the hydrophobic interior:
Small nonpolar molecules (e.g., many lipids and hydrophobic compounds)
Some small uncharged polar molecules to a limited degree (their polarity still restricts passage)
Even when such molecules can cross, the membrane still functions as a selective barrier because different molecules cross at different rates.
Substances that are strongly restricted
The hydrophobic core largely blocks:
Ions (e.g., , ) due to charge and hydration shells
Large polar molecules (e.g., many sugars and amino acids) due to size and polarity
When cells need these substances to cross, they do not rely on the phospholipid core; instead, they use specific membrane components that provide alternative pathways while preserving the barrier function of the hydrophobic interior.
Biological significance of selective permeability
Maintaining distinct internal conditions
By separating internal and external environments, membranes support homeostasis at the cellular level. Selective permeability enables cells to:
Accumulate nutrients without losing them rapidly
Retain metabolites and signalling molecules
Prevent uncontrolled movement of solutes that would disrupt water balance
Supporting gradients that drive cellular processes
Because the membrane restricts many solutes, cells can maintain concentration differences across the membrane. These differences are essential for numerous cellular functions, including controlled transport and signalling, and they depend on the membrane remaining a strong barrier to most polar and charged substances.
FAQ
Ions are stabilised by a hydration shell in water. Entering the non-polar membrane core would require shedding much of that shell, which is energetically costly because charge is poorly stabilised in hydrophobic environments.
Higher temperature typically increases lipid movement, making the bilayer more fluid and slightly more permeable to some small molecules. Lower temperature reduces fluidity, tightening packing and decreasing permeability.
No. Lipid composition varies among cell types and organelles. Differences in fatty acid saturation and lipid types alter how tightly tails pack, changing how easily small molecules can pass through the hydrophobic core.
Cholesterol can reduce permeability to small polar molecules by filling gaps between phospholipid tails. It also buffers fluidity: restraining movement at higher temperatures and preventing tight packing at lower temperatures.
Some small uncharged polar molecules have limited ability to partition into the bilayer, especially if they are very small and only weakly polar. They still cross far more slowly than non-polar molecules.
Practice Questions
Explain how the hydrophobic interior of the plasma membrane contributes to selective permeability. (2 marks)
Identifies that the membrane interior is non-polar/hydrophobic due to phospholipid tails (1)
Explains that polar/charged substances are excluded while non-polar molecules cross more readily (1)
A cell is placed in a solution containing (i) a small non-polar molecule, (ii) a large uncharged polar molecule, and (iii) an ion such as . Predict and explain the relative ability of each substance to cross the plasma membrane directly through the phospholipid bilayer. (5 marks)
States small non-polar molecule crosses most readily (1)
Explains it dissolves in the hydrophobic core / is compatible with non-polar interior (1)
States large uncharged polar molecule crosses poorly/slowly (1)
Explains polarity and/or size reduces entry into hydrophobic core (1)
States ion such as crosses least/not directly (1)
Explains charge (and hydration shell) is energetically unfavourable in hydrophobic interior (1) (Max 5)

