AP Syllabus focus:
‘Osmosis moves water from high to low water potential, often described as movement from hypotonic to hypertonic regions.’
Osmosis explains how cells gain or lose water across selectively permeable membranes. Water potential provides a quantitative framework for predicting the direction of net water movement in biological systems, especially when solutes and pressure are involved.
Core ideas: osmosis and water potential
Osmosis depends on a selectively permeable membrane
Osmosis: Net movement of water across a selectively permeable membrane from higher water potential to lower water potential.
Osmosis is “net” movement because water molecules move randomly in both directions; a difference in water potential creates an overall bias in one direction.
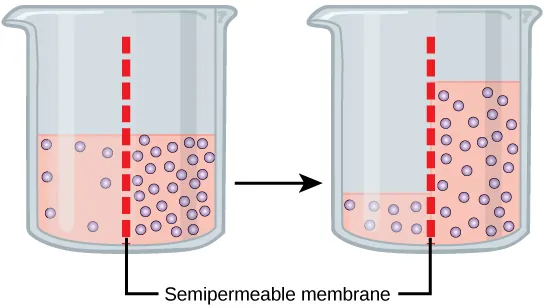
Osmosis across a semipermeable membrane: solute is unable to cross, but water diffuses until the gradient is reduced. The diagram makes the “net movement” idea explicit by showing water shifting toward the side with higher solute (lower free-water concentration). Source
Water potential predicts direction of water movement
Water potential (): The potential energy of water in a system, relative to pure water, that determines the direction of water movement; water moves from higher to lower .
In cells, is lowered mainly by dissolved solutes and can be increased by physical pressure against a cell wall. This makes water potential especially useful for understanding osmosis in plant tissues and other pressurised systems.
Linking “hypotonic/hypertonic” to water potential
Tonicity language as a shortcut
The syllabus framing is that osmosis moves water from high to low water potential, and this is often described as water moving from hypotonic to hypertonic regions.
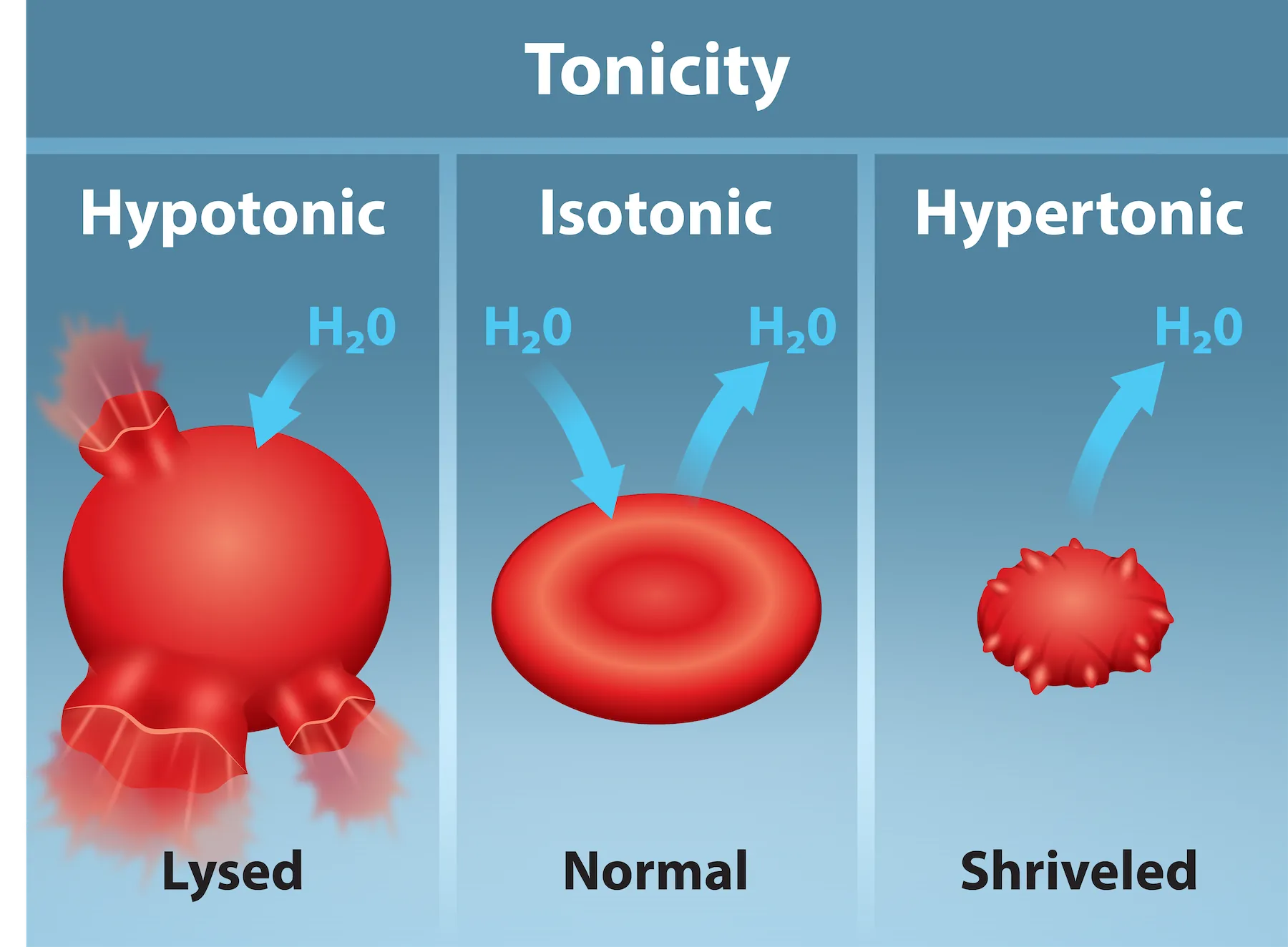
Red blood cells in hypotonic, isotonic, and hypertonic solutions show how tonicity predicts water movement and resulting cell shape changes. The figure highlights swelling/lysis in hypotonic conditions, no net change in isotonic conditions, and shrinking (crenation) in hypertonic conditions. Source
Hypotonic region: lower solute concentration (effectively higher for water), so water tends to leave it.
Hypertonic region: higher solute concentration (effectively lower for water), so water tends to enter it.
These terms are most reliable when comparing solutions separated by a membrane that is permeable to water but not to the solutes creating the gradient; otherwise, tonicity can change as solutes redistribute.
Components of water potential in biology
Solutes lower water potential
Dissolving solute reduces the “free” water molecules available to move, lowering . This component is typically called solute potential and is always negative (or zero in pure water).
Pressure can raise water potential
Physical pressure (for example, in walled cells) increases the tendency for water to move out of that compartment. In plants, turgor pressure can make the cell’s less negative, reducing further water uptake.

Plant water status under different water-potential conditions: when external is lower, water exits cells and the plant wilts; when external is higher, water enters cells and builds turgor pressure. This visual ties pressure potential () to whole-plant rigidity and explains why walled cells behave differently from animal cells. Source
= overall water potential (typically in MPa)
= change in water potential due to dissolved solutes (MPa; )
= change in water potential due to physical pressure (MPa; can be )
Because water moves from higher to lower , a compartment with more negative can still fail to gain water if it also has sufficiently high positive .
What actually happens at the membrane
Direction and “net” movement
Across a selectively permeable membrane:
If the outside solution has lower than the cytosol, net water movement is out of the cell.
If the outside solution has higher , net water movement is into the cell.
If is equal on both sides, there is no net osmosis (dynamic equilibrium).
Biological significance for cells
Osmosis affects cell volume and thus cell function because membranes are flexible:
Water influx can increase membrane tension.
Water efflux can reduce cytoplasmic volume and disrupt normal biochemical conditions.
In walled cells, pressure potential can build as water enters, helping stabilise cell volume and limiting further net uptake once the internal and external values match.
FAQ
It is a reference point that makes comparisons straightforward.
Any dissolved solute decreases the free energy of water relative to this standard, so $\Psi$ becomes negative, allowing direction of movement to be predicted consistently.
Higher temperature increases molecular kinetic energy and can increase the rate at which equilibrium is reached.
However, temperature can also change solute behaviour and membrane properties, so experimental comparisons of $\Psi$ should be made at controlled temperatures.
Aquaporins are water channel proteins that increase membrane permeability to water.
They increase the rate of water movement without changing the direction, which is still determined by the water potential gradient.
Common approaches include pressure chamber methods and psychrometers.
These techniques estimate $\Psi$ by determining the pressure or vapour conditions needed to balance water movement, translating physical readings into MPa values.
If a membrane is permeable to solutes, solutes can move and change the gradient over time.
Also, tonicity ignores pressure effects; two compartments may have similar solute concentrations but different $\Psi$ due to differences in $\Psi_p$.
Practice Questions
Define osmosis and state the direction of net water movement in terms of water potential. (2 marks)
Defines osmosis as net movement of water across a selectively permeable membrane (1)
States water moves from higher water potential to lower water potential (1)
Explain how water potential accounts for osmosis between a cell and its external solution, including the roles of solute potential and pressure potential. (6 marks)
Water moves by osmosis from higher to lower (1)
is affected by solutes and pressure, e.g. (1)
Solute potential () becomes more negative as solute concentration increases, lowering (2)
Pressure potential () increases when positive pressure builds, opposing further water entry (1)
Net movement stops at equilibrium when is equal across the membrane (1)

