AP Syllabus focus:
‘External solutions may be hypotonic, hypertonic, or isotonic relative to cells, affecting net water movement across membranes.’
Tonicity describes how an external solution influences a cell’s volume by driving net water movement across the plasma membrane. In AP Biology, focus on hypotonic, hypertonic, and isotonic solutions and their predictable effects on cells.
Core idea: tonicity depends on “effective” solutes
Tonicity is not just “how concentrated” a solution is; it specifically predicts the direction of net water movement and resulting cell swelling or shrinking. The key is whether solutes can cross the membrane.
Tonicity: The ability of a solution to cause a cell to gain or lose water, determined by the concentration of nonpenetrating solutes relative to the cell.
A selectively permeable membrane allows water to move more freely than many solutes. If a solute cannot cross (or crosses extremely slowly), it behaves as a nonpenetrating solute and has a strong effect on tonicity because it maintains a persistent concentration difference.
Tonicity vs. concentration language
Students often mix up these related terms:
Solute concentration describes how much solute is present.
Tonicity predicts what happens to cell volume.
Two solutions can have similar total solute concentration but different tonicity if their solutes differ in membrane permeability.
The three solution types relative to a cell
When you compare an external solution to the cell’s cytosol, you classify it as hypotonic, hypertonic, or isotonic. These terms are always relative (they require a reference cell and its internal solute conditions).
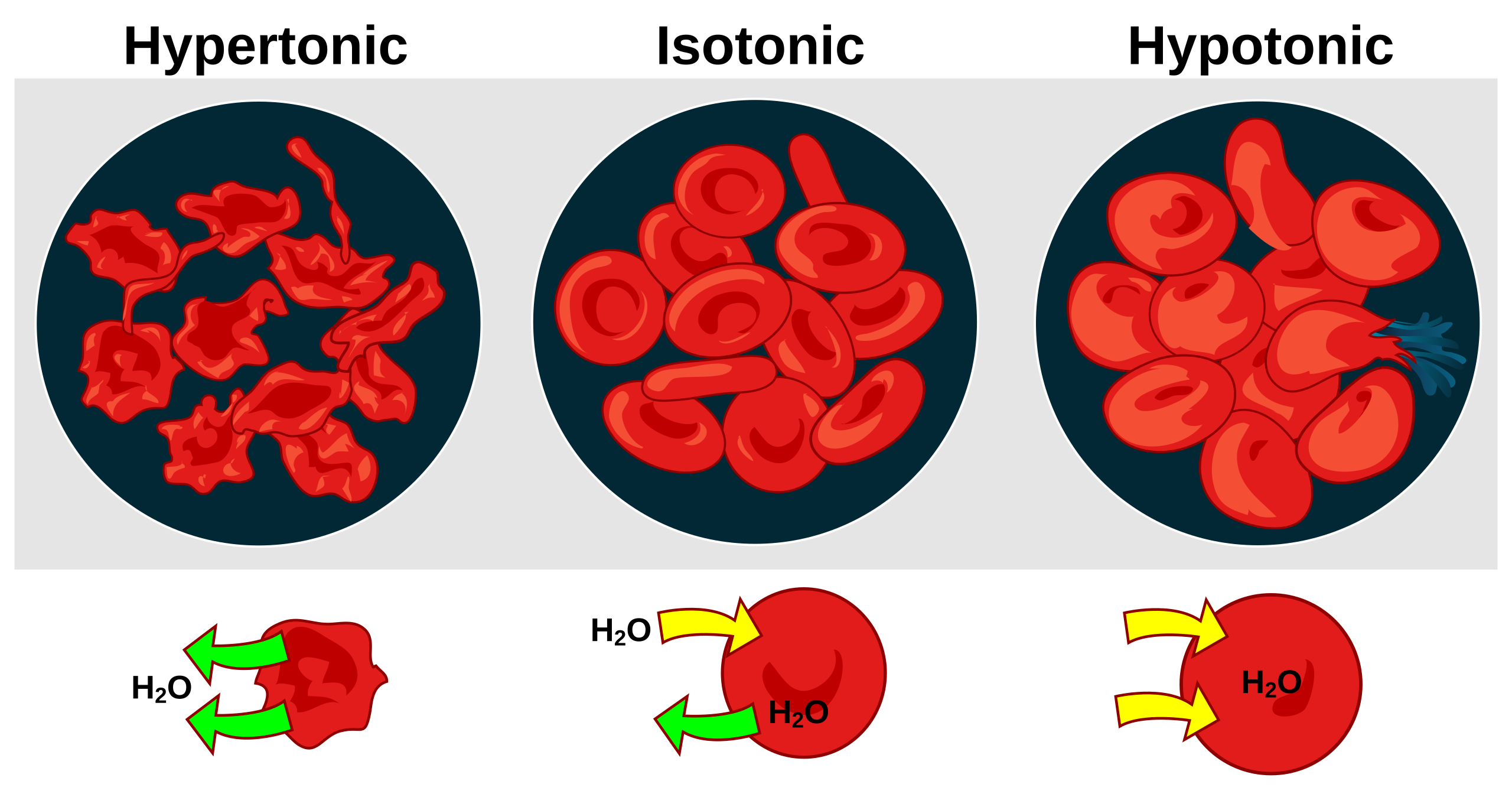
This schematic summarizes the relationship between extracellular tonicity and red blood cell volume: hypertonic solutions cause cells to shrink, isotonic solutions maintain shape, and hypotonic solutions can cause swelling and rupture. Using RBCs makes the direction of net water movement visually obvious, helping you connect terminology to predicted outcomes. Source
Hypotonic solution
In a hypotonic environment, the solution outside the cell has a lower concentration of nonpenetrating solutes than inside the cell.
Hypotonic: A solution with a lower concentration of nonpenetrating solutes than the cell; water tends to enter the cell, increasing cell volume.
Key outcomes:
Net water movement: into the cell
Animal cells: may swell; if excessive, can undergo lysis (bursting) because they lack a rigid cell wall.
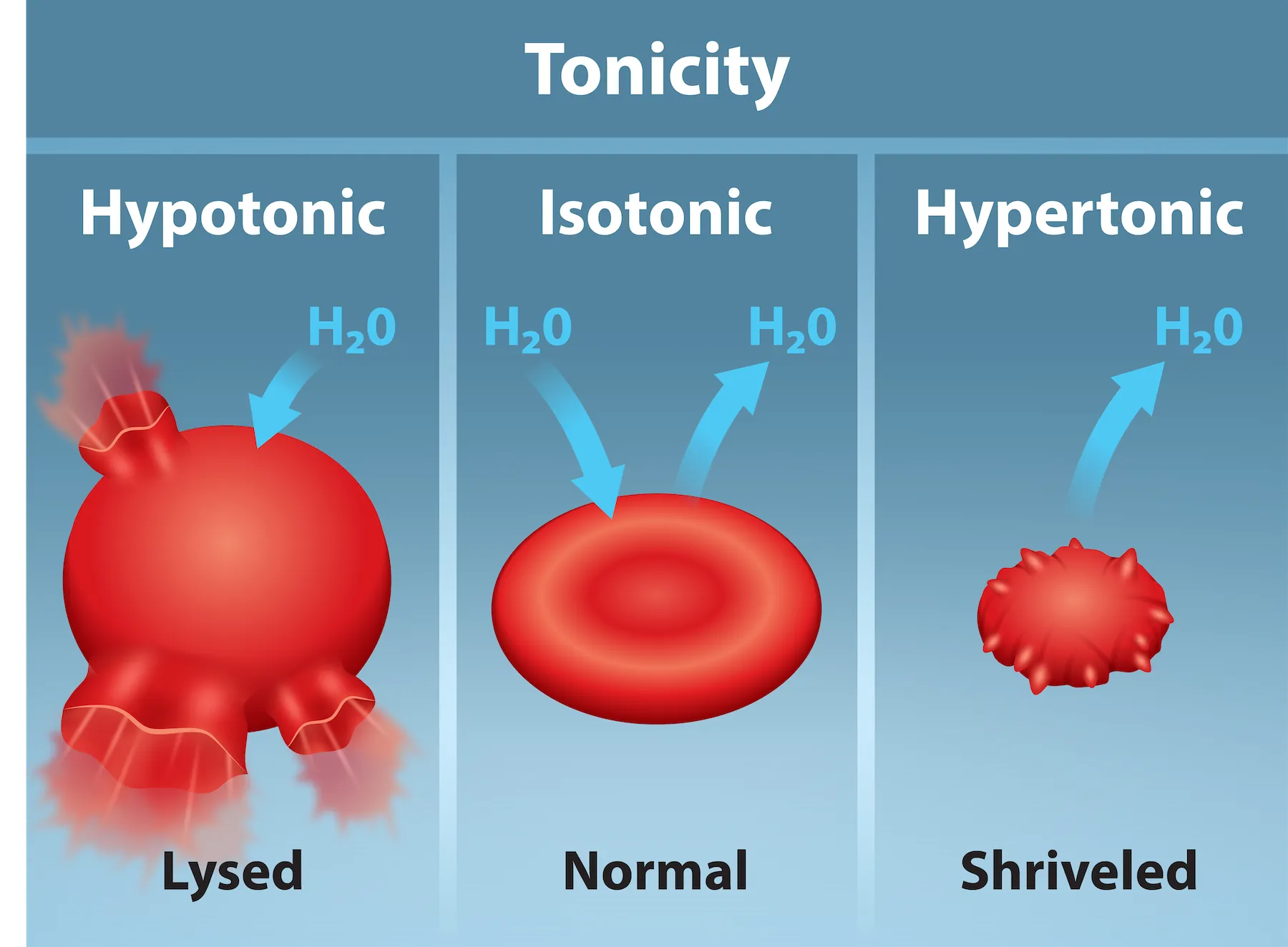
Red blood cells provide a classic visual readout of tonicity: in a hypotonic solution water enters and can cause lysis, in an isotonic solution cells retain normal volume, and in a hypertonic solution water leaves and the cells shrink. The arrows emphasize that tonicity is about net water movement driven by nonpenetrating solute differences. Source
Plant cells: become turgid as the central vacuole expands and the cell wall resists further expansion; this supports plant structure.
Hypertonic solution
In a hypertonic environment, the solution outside the cell has a higher concentration of nonpenetrating solutes than inside.
Hypertonic: A solution with a higher concentration of nonpenetrating solutes than the cell; water tends to leave the cell, decreasing cell volume.
Key outcomes:
Net water movement: out of the cell
Animal cells: shrink (often described as crenation in red blood cells).
Plant cells: lose water; the plasma membrane may pull away from the cell wall, a condition called plasmolysis, reducing turgor and causing wilting.
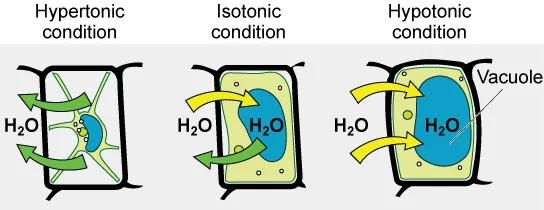
This diagram contrasts plant cell responses across tonicities: hypertonic conditions drive water out (promoting plasmolysis), isotonic conditions reduce turgor (flaccid), and hypotonic conditions drive water in (turgid). The large central vacuole is highlighted because vacuolar expansion is the main contributor to turgor pressure in plant cells. Source
Isotonic solution
In an isotonic environment, the solution outside the cell has an approximately equal concentration of nonpenetrating solutes compared with inside.
Isotonic: A solution with an equal concentration of nonpenetrating solutes as the cell; there is no net water movement and cell volume remains stable.
Key outcomes:
Net water movement: water moves both directions, but no net change in cell volume
Animal cells: maintain normal volume
Plant cells: often become flaccid (less rigid) because turgor pressure is not maximized, though the cell does not necessarily collapse.
Why “relative to cells” matters
The same solution can be:
Isotonic to one cell type but hypertonic to another if their internal solute concentrations differ.
Hypotonic at one time and isotonic later if cell solute content changes (for example, after solute transport or metabolism alters cytosolic composition).
Common pitfalls and how to avoid them
Do not label a solution as hypo-/hyper-/isotonic without stating relative to what cell.
Focus on nonpenetrating solutes when predicting volume change; a solute that readily crosses the membrane does not sustain the gradient that drives lasting water movement.
Remember that tonicity predicts cell volume change, not the rate of water movement (which also depends on membrane permeability and surface area).
FAQ
Iso-osmotic means equal total solute concentration, but isotonic depends on non-penetrating solutes.
If some solutes can cross the membrane, they may not maintain an effective gradient, so cell volume can still change.
Penetrating solutes can diffuse into/out of the cell, reducing the concentration difference over time.
Non-penetrating solutes remain separated by the membrane, sustaining the imbalance that drives lasting net water movement.
They can change whether a solute behaves as “penetrating” in practice.
More channels/carriers for a solute can make it effectively penetrating
Fewer transport pathways can make it effectively non-penetrating
Isotonic fluids minimise net water movement into or out of red blood cells.
This helps prevent:
cell swelling and haemolysis in hypotonic conditions
cell shrinkage (crenation) in hypertonic conditions
They assess membrane permeability to that solute by measuring how quickly it crosses the membrane.
Practical indicators include the time course of cell volume change and whether the volume change persists (suggesting non-penetrating behaviour).
Practice Questions
Define an isotonic solution and state its effect on an animal cell’s volume. (2 marks)
Correct definition: equal concentration of non-penetrating solutes relative to the cell (1)
States no net movement of water / cell volume remains unchanged (1)
A plant cell is placed into a hypertonic solution. Explain what happens to the movement of water and describe two resulting changes to the plant cell. (5 marks)
Identifies the external solution has higher concentration of non-penetrating solutes than the cell (1)
States net movement of water out of the cell (1)
Describes loss of turgor / cell becomes flaccid leading towards wilting (1)
Describes plasmolysis: membrane pulls away from the cell wall as the cytoplasm shrinks (1)
Links changes to decreased cell volume due to water loss (1)

