AP Syllabus focus:
‘Water and solute potential equations quantify water movement; structures like contractile vacuoles and plant central vacuoles exemplify osmoregulation.’
Water movement in biology can be predicted quantitatively using water potential. By combining solute potential and pressure potential, students can explain why water enters, leaves, or is stored by cells and organelles.
Water potential: what it measures
Water potential (): The tendency of water to move from one region to another, expressed as potential energy per unit volume; water moves from higher to lower .
Water potential is most useful when comparing two regions (inside vs. outside a cell, or cytosol vs. vacuole).
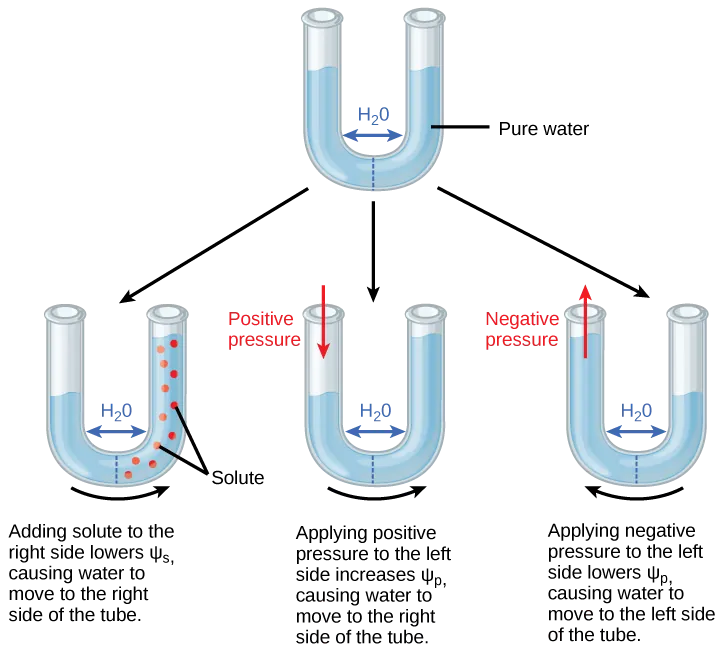
This OpenStax figure uses a U-tube separated by a semipermeable membrane to visualize how water moves from higher to lower water potential. It contrasts the effects of adding solute (making more negative) versus applying positive/negative pressure (changing ) on net water movement and equilibrium. Source
The direction of net osmosis depends on the water potential gradient, not on “where there is more water,” but where water has higher potential energy.
Core equations used in AP Biology
Water potential is typically partitioned into contributions from solutes and physical pressure. In many AP Biology contexts, additional terms (e.g., gravitational potential) are ignored because they are small at the cellular scale.
= water potential (typically in MPa)
= solute (osmotic) potential (MPa)
= pressure potential (MPa)
This relationship highlights two major ways to change : adding solute (changes ) or changing physical pressure (changes ). Pure water at atmospheric pressure is defined as MPa.
Solute potential (): The component of water potential due to dissolved solutes; adding solute lowers water potential (makes it more negative).
Solute potential is negative (or zero) because solutes reduce the free energy of water by binding water molecules and reducing the fraction of water available to move.
= ionisation constant (dimensionless)
= molar concentration (mol L)
= pressure constant (MPa L mol K)
= temperature (K)
The ionisation constant accounts for dissociation (e.g., NaCl yields more particles than glucose at the same molarity), increasing the magnitude of the negative .
Pressure potential and living cells
Pressure potential (): The component of water potential due to physical pressure on water; it is often positive in living plant cells because the cell wall resists expansion.
In animal cells, is often approximated as 0 MPa because there is no rigid wall generating sustained positive pressure. In plant cells, turgor pressure raises , which can partially offset a very negative inside the cell.
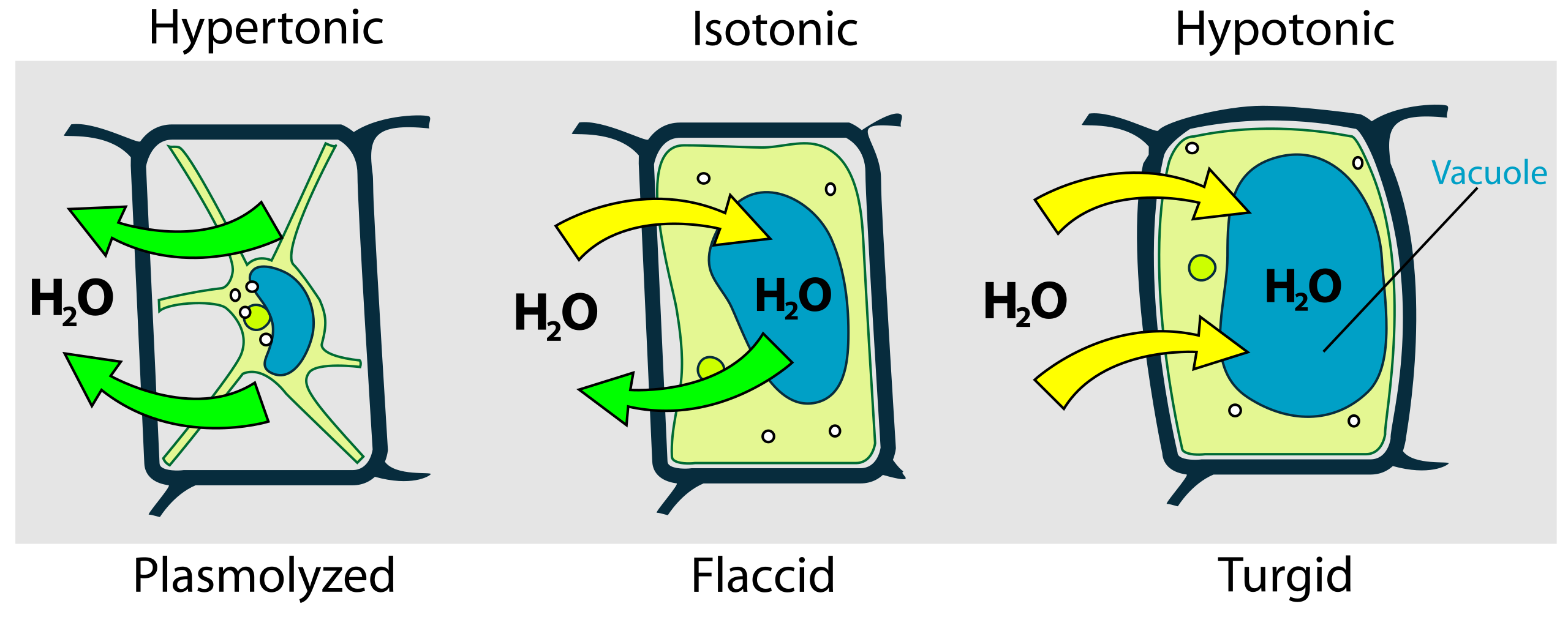
This diagram depicts how water movement changes plant cell volume and turgor, comparing flaccid/plasmolyzed versus turgid states. It makes the role of the cell wall explicit: as water enters, internal pressure builds, increasing pressure potential and opposing further influx. Source
Interpreting water movement with the equations
Water moves from higher to lower across a selectively permeable membrane.
A more negative (more dissolved particles) tends to pull water in by lowering total .
A higher tends to push water out by raising total .
At equilibrium, there is no net water movement because is equal on both sides (water can still move both directions equally).
Biological examples tied to osmoregulation
Contractile vacuoles (protists in dilute environments)
Contractile vacuoles help maintain cell integrity when the cytosol has a lower than the surrounding medium, producing a net inward water flux. Key idea using water potential:
The cell continually gains water because internal is lower (often due to solutes in the cytosol).
The contractile vacuole collects and expels water, preventing excessive swelling and maintaining a stable internal environment.
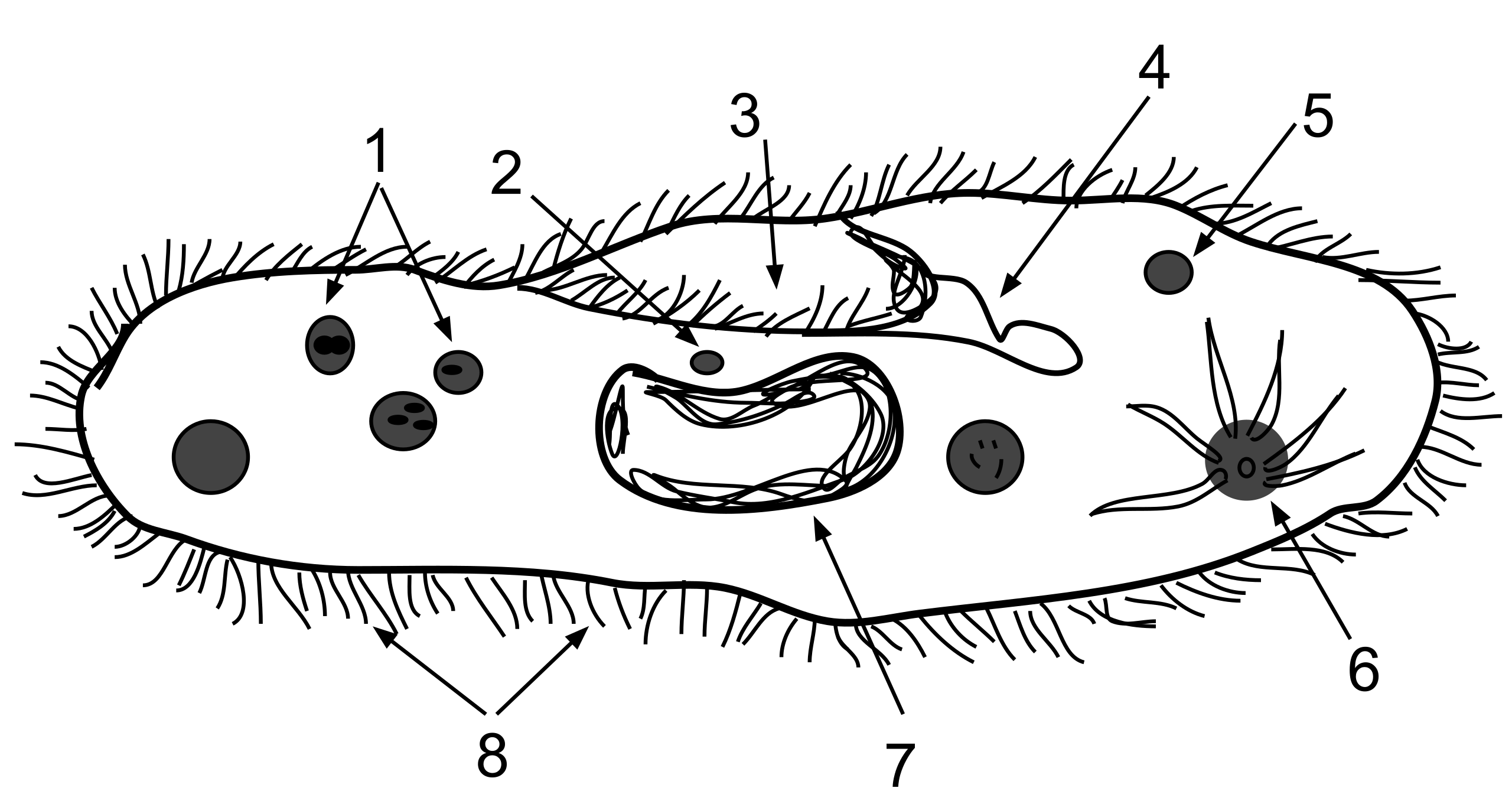
This labeled Paramecium anatomy diagram identifies the contractile vacuole within a freshwater protist. It supports the osmoregulation example by showing the structure responsible for collecting excess water and periodically expelling it to maintain cytosolic homeostasis. Source
Central vacuole (plant cells)
The plant central vacuole is a major compartment for solutes and water storage, and it strongly influences within the cell.
Solute accumulation in the vacuole lowers vacuolar , tending to draw water into the vacuole.
As water enters, the cell wall resists expansion, increasing (turgor), which can raise overall and reduce further net influx.
This balance supports rigidity in non-woody tissues and helps stabilise water relations when external conditions change.
FAQ
Solutes reduce the free energy of water by forming hydration shells and reducing the fraction of “free” water molecules. That reduction is represented as a negative contribution to $\Psi$.
It approximates how many dissolved particles a solute produces.
Non-electrolytes (e.g., sucrose): $i \approx 1$
Salts that dissociate: $i > 1$, though real values can deviate from ideal due to ion pairing.
A rigid cell wall resists expansion as water enters, generating sustained positive hydrostatic pressure. Without a wall, pressure does not build in the same controlled way.
Yes. A solution with a more negative $\Psi_s$ can be balanced by a more positive $\Psi_p$, yielding the same total $\Psi$ as another solution with fewer solutes but lower pressure.
It provides a consistent reference point, making it easier to compare biological compartments. Most real biological fluids have $\Psi < 0$ because they contain solutes, while plant cells may partly offset this with positive $\Psi_p$.
Practice Questions
Define solute potential and state how increasing solute concentration affects it. (2 marks)
Solute potential is the component of water potential due to dissolved solutes (1).
Increasing solute concentration makes more negative / lowers (1).
Explain how the equation helps account for water movement and water balance in (i) a protist with a contractile vacuole and (ii) a plant cell with a central vacuole. (5 marks)
Correct statement that water moves from higher to lower (1).
Protist: internal solutes make relatively negative, lowering total and promoting net water entry (1).
Protist: contractile vacuole expels excess water to maintain water balance (1).
Plant: solutes in central vacuole lower , drawing water into vacuole/cell (1).
Plant: water entry increases (turgor), raising total and opposing further net influx (1).

