AP Syllabus focus: ‘Electronegativity generally increases across a period and decreases down a group; relate these trends to electronic structure, the shell model, and Coulomb’s law.’
Electronegativity trends help you predict where bonding electrons will be drawn in a bond and, broadly, whether bonding is more like electron sharing or electron transfer. These trends come from atomic structure and electrostatic attraction.
Electronegativity: meaning and use
Electronegativity is a comparative property used to predict how strongly atoms attract bonding electrons in a chemical bond.
Electronegativity — the relative ability of an atom in a bond to attract the shared electrons toward itself.
Electronegativity values are typically reported on the Pauling scale, but AP Chemistry primarily uses relative comparisons and periodic trends rather than memorised numbers.
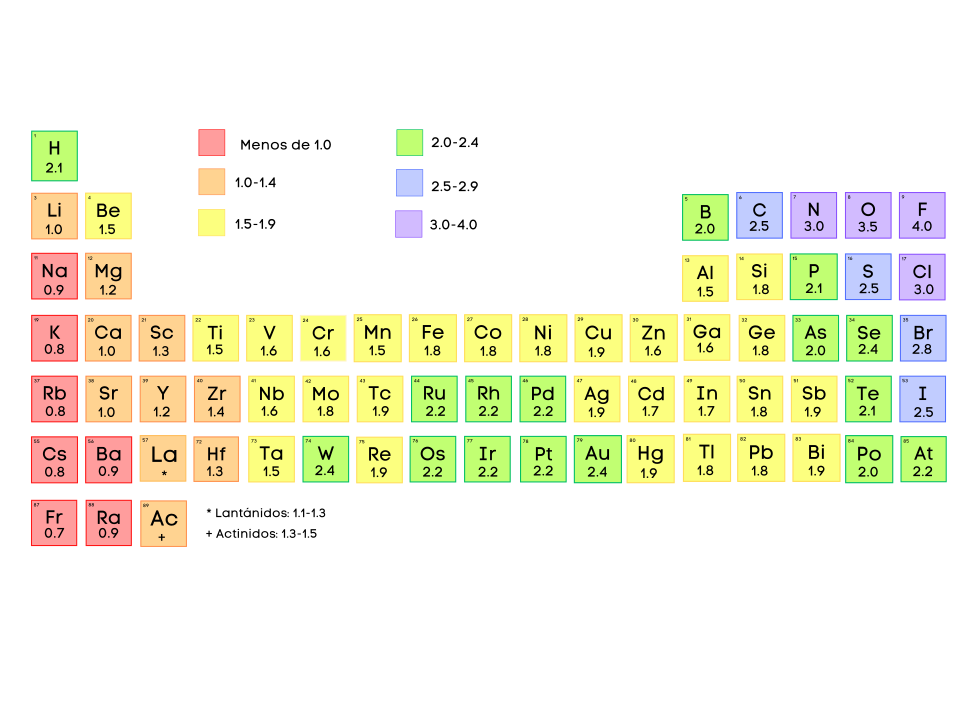
Periodic-table map of electronegativity values on the Pauling scale, showing the overall increase toward the upper right of the table and the unusually high values for elements such as fluorine. This kind of visualization reinforces that AP Chemistry cares most about relative electronegativity patterns rather than memorizing a complete list of numbers. Source
What electronegativity is (and is not)
It describes attraction for bonding electron density, not the “pull” on all electrons equally.
It is not a directly measured quantity like mass; it is inferred from bonding data and trends.
It is most useful when comparing two atoms:
The more electronegative atom attracts electron density and becomes relatively electron-rich in the bond.
Periodic trends in electronegativity
The AP trend statement is central:
Across a period (left → right): electronegativity generally increases.
Example charts of electronegativity values that rise across a period (illustrating left-to-right increase) and fall down a group (illustrating top-to-bottom decrease). Using specific element sequences helps connect the abstract trend statements to concrete data patterns students can recognize quickly. Source
Down a group (top → bottom): electronegativity generally decreases.
Across a period: why it increases
Across a period, atoms gain protons while adding electrons to the same principal energy level (same shell), so the bonding electrons are not, on average, much farther from the nucleus. Key ideas:
Nuclear charge increases (more protons).
Shielding by inner (core) electrons changes little because the number of core shells stays the same.
Atomic radius generally decreases, so bonding electrons are closer to the nucleus.
These combine to increase the nucleus–electron attraction, so the atom more strongly attracts bonding electrons.
Effective nuclear charge () — the net positive charge “felt” by an electron after accounting for shielding by other electrons.
Down a group: why it decreases
Down a group, atoms add electron shells (higher ), placing valence electrons in orbitals farther from the nucleus. Key ideas:
Distance () increases between nucleus and valence/bonding electrons.
Shielding increases because there are more inner shells of electrons.
Although nuclear charge increases, the increased distance and shielding reduce the net attraction for bonding electrons.
As a result, larger atoms lower in a group attract bonding electron density less strongly, so electronegativity decreases.
Connecting trends to the shell model and Coulomb’s law
The shell model (electrons arranged in energy levels) explains why distance and shielding change predictably across periods and down groups. Electronegativity trends follow from electrostatic attraction between:
the positively charged nucleus, and
negatively charged electrons in the bonding region.
Coulomb’s law captures the dependence of electrostatic force on charge and distance.
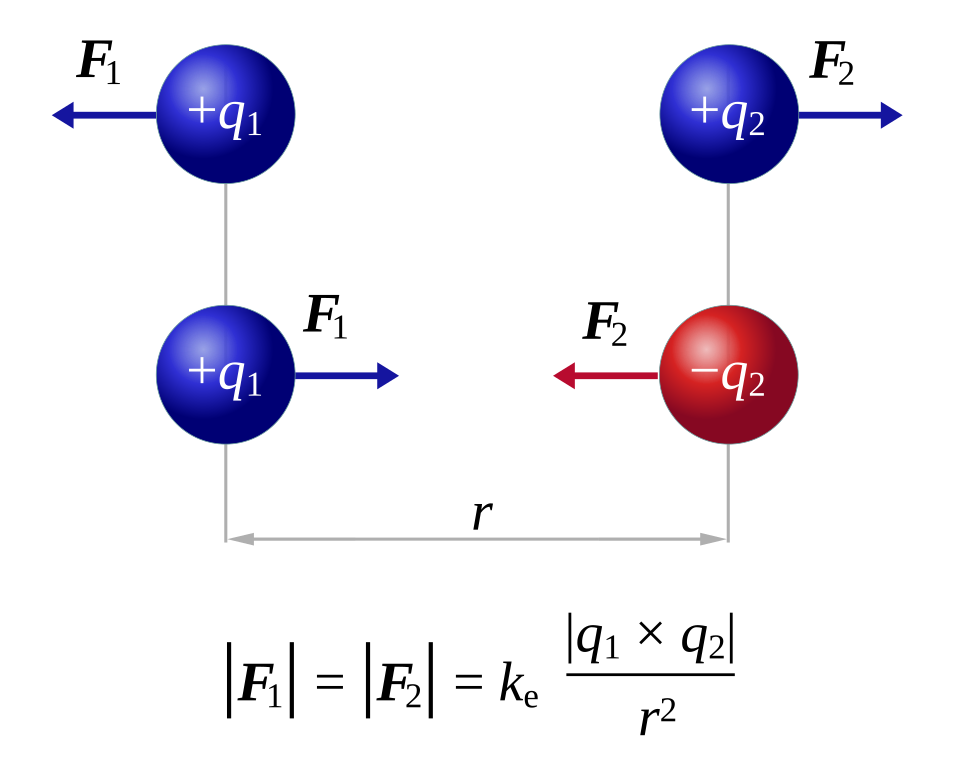
Schematic diagram of Coulomb’s law showing like-charge repulsion and opposite-charge attraction, with forces drawn along the line connecting the charges. The labels emphasize the inverse-square dependence on separation distance , aligning with the qualitative argument that increasing (down a group) weakens electrostatic attraction. Source
= electrostatic force of attraction or repulsion (newtons, N)
= charge on particle 1 (coulombs, C)
= charge on particle 2 (coulombs, C)
= distance between the charges (metres, m)
= Coulomb’s constant ()
Qualitatively applying Coulomb’s law to electronegativity:
Across a period:
effective positive charge attracting bonding electrons increases (larger “” effect via higher ),
and decreases (smaller atoms),
so attraction strengthens → electronegativity increases.
Down a group:
increases substantially (more shells),
shielding lowers the effective attraction (smaller “” effect via reduced at the valence level),
so attraction weakens → electronegativity decreases.
Bonding predictions from electronegativity trends
Electronegativity trends support quick, defensible predictions about bonding electron distribution.
Predicting direction of electron pull in a bond
Identify which atom is more electronegative using periodic trends.
Predict bonding electrons are drawn toward the more electronegative atom.
Use this to anticipate which end of a bond is relatively electron-rich vs electron-poor.
Predicting “more sharing” vs “more transfer” (broadly)
As the electronegativity difference between two bonded atoms increases, bonding tends to shift from:
more equal sharing of electrons (smaller difference), toward
stronger separation of charge (larger difference), consistent with increased electron density near the more electronegative element.
Because electronegativity is tied to periodic position:
Nonmetals (upper right, excluding noble gases) tend to be more electronegative.
Metals (left/centre) tend to be less electronegative. So bonds between a low-electronegativity element and a high-electronegativity element typically show a strong electron-density shift toward the nonmetal.
High-utility periodic cues
Electronegativity increases toward the top right; fluorine is the highest commonly referenced element.
Electronegativity is low toward the bottom left (alkali metals are among the lowest).
When two elements are in the same group, the higher (smaller) element is typically more electronegative due to less shielding and smaller .
FAQ
Electronegativity depends on several competing factors (subshell energies, electron–electron repulsions, and orbital penetration), not just proton number.
Small deviations can occur when a new subshell begins filling or when electron pairing changes repulsion within an orbital.
Many noble gases form few stable covalent bonds under typical conditions, so there is limited bonding data to define a meaningful Pauling electronegativity.
Some modern scales assign values for certain noble gases, but AP trend reasoning usually excludes them.
Actual nuclear charge is the number of protons, $Z$.
Effective nuclear charge, $Z_\text{eff}$, is the net attraction experienced by a particular electron after shielding by other electrons is considered, so it is always less than $Z$.
No. Ionisation energy concerns removing an electron from a gaseous atom; electron affinity concerns adding an electron to a gaseous atom.
Electronegativity describes attraction for electron density in a bond, but it often correlates with those properties because all reflect nucleus–electron attraction.
Orbitals with greater penetration (electron density closer to the nucleus) experience higher $Z_\text{eff}$.
For example, $s$ electrons penetrate more than $p$ electrons, influencing how strongly valence electrons are attracted and contributing to subtle variations in electronegativity across and within periods.
Practice Questions
(2 marks) State the general trend in electronegativity (i) across a period and (ii) down a group. Then state which is more electronegative, oxygen or sulfur.
Across a period electronegativity increases (1)
Down a group electronegativity decreases (1)
Oxygen is more electronegative than sulfur (allow as part of either trend mark if clearly linked)
(6 marks) Using electronic structure, the shell model, and Coulomb’s law ideas, explain why electronegativity increases from sodium to chlorine across Period 3. Your answer should refer to effective nuclear charge, shielding, and distance from the nucleus.
Nuclear charge/proton number increases across Na → Cl (1)
Electrons are added to the same shell/energy level in Period 3 (shell model reference) (1)
Shielding by inner shells is approximately constant across the period (1)
Effective nuclear charge felt by bonding/valence electrons increases across the period (1)
Atomic radius/distance to bonding electrons decreases across the period (1)
Therefore nucleus–electron attraction increases (Coulomb’s law: larger effective charge and/or smaller increases attraction), so electronegativity increases (1)

