AP Syllabus focus: ‘Atoms with similar electronegativity share valence electrons to form nonpolar covalent bonds; many C–H bonds are effectively nonpolar despite a small electronegativity difference.’
Nonpolar covalent bonding explains why many molecules have bonds with shared electrons but little to no charge separation. This page focuses on how similar electronegativity values lead to essentially equal sharing in a covalent bond.
Core idea: similar electronegativity means equal sharing
Electronegativity and electron sharing
Electronegativity: a relative measure of how strongly an atom attracts shared (bonding) electrons in a covalent bond.
When two bonded atoms have similar electronegativities, neither atom can “pull” the bonding electrons much more strongly than the other. As a result, the bonding electron density is approximately symmetric between the nuclei, and the bond is considered nonpolar covalent.
To discuss “similar” quantitatively, chemists often compare electronegativity values on the same scale (typically Pauling).
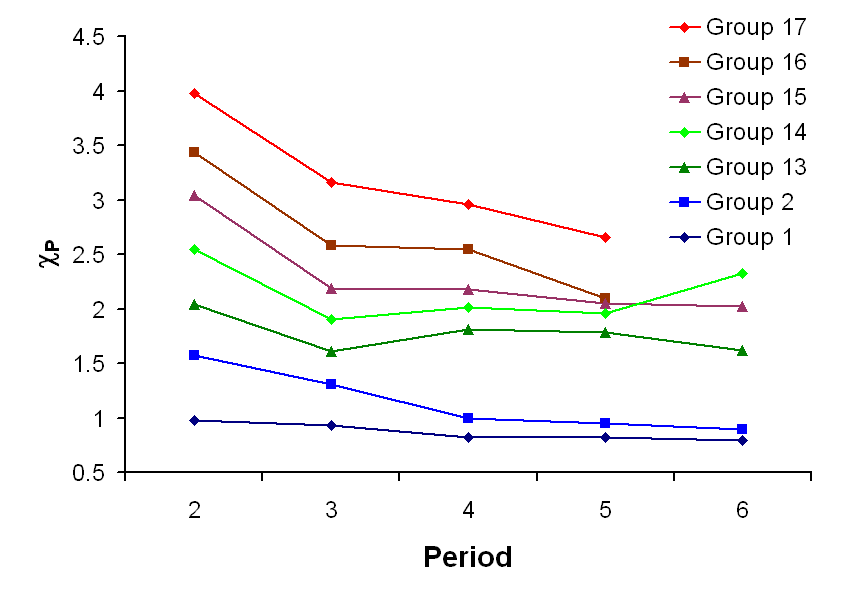
Periodic trends in Pauling electronegativity across the first six periods. The plot highlights the systematic rise toward the upper-right region of the periodic table, which explains why atoms in similar regions often have comparable electronegativity values (and therefore more equal electron sharing). Source
The key comparison is the electronegativity difference.
= electronegativity difference (unitless)
= electronegativities of the bonded atoms on the same scale (unitless)
A small supports the prediction of nonpolar covalent bonding, but AP Chemistry emphasises the conceptual link: similar attraction strengths produce shared electrons without meaningful partial charges.
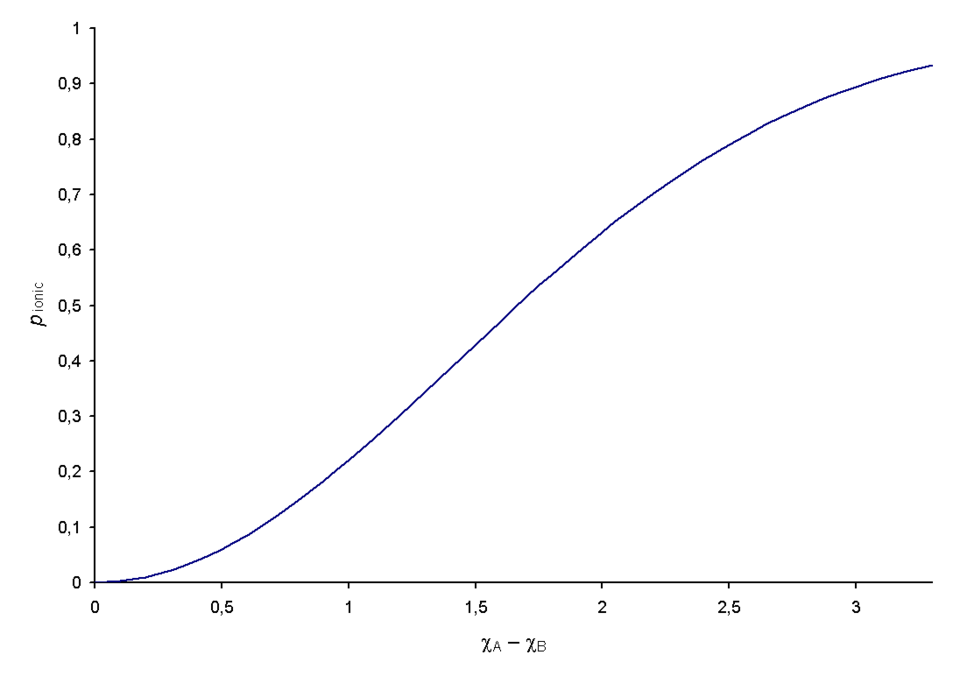
Graphical representation of Pauling’s relationship between electronegativity difference and estimated ionic character of a bond. The curve makes the modeling point explicit: as increases, the bond gains a larger ionic contribution (greater charge separation), whereas small stays largely covalent. Source
What “nonpolar covalent” means in bonding terms
Nonpolar covalent bond: a covalent bond in which bonding electrons are shared approximately equally, producing negligible partial charges on the bonded atoms.
In a nonpolar covalent bond:
The bonding pair is located roughly midway between the atoms (on average).
Any partial charges ( and ) are absent or extremely small.
The bond is best modelled as equal electron sharing, not electron transfer.
Recognising nonpolar covalent bonds on the AP level
Common cases where electronegativities are identical or very close
Bonds are strongly predicted to be nonpolar covalent when:
The atoms are the same element (homonuclear diatomics), so electronegativity values are identical (e.g., , , ).
The atoms are different but have very similar electronegativities, so the shared electrons are not significantly displaced toward either atom.
Because electronegativity values are comparative and depend on scale and context, AP Chemistry typically expects you to use:
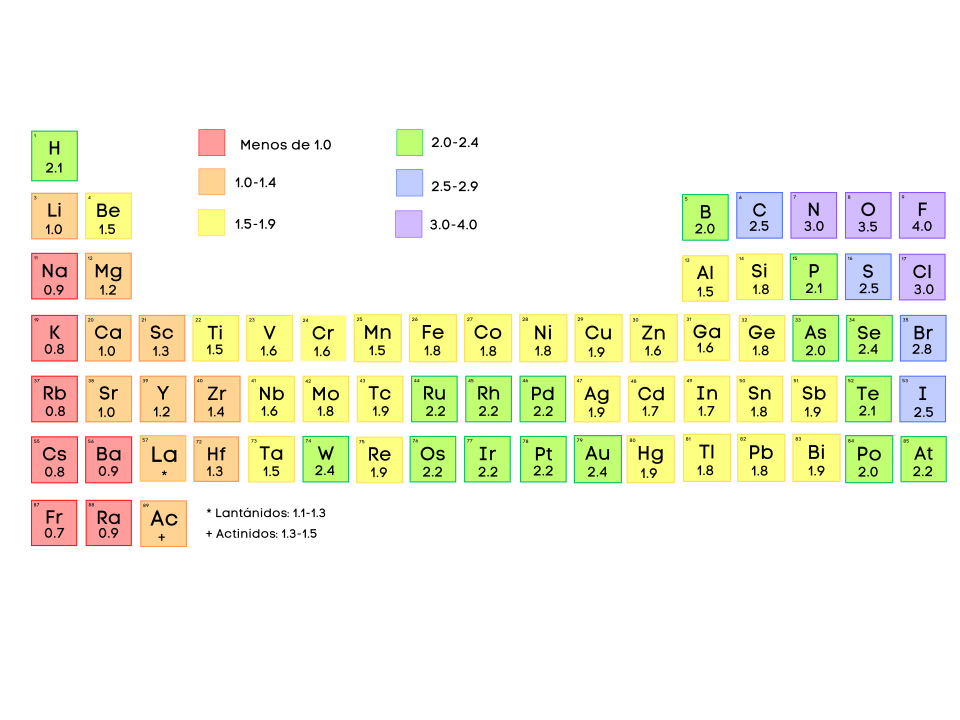
Reference diagram summarizing Pauling-scale electronegativity values alongside common interpretive ranges used in introductory chemistry. It helps students translate “similar electronegativities” into a practical decision-making framework for classifying bonds as nonpolar covalent versus increasingly polar/ionic as grows. Source
The relative electronegativity relationship (similar vs. different), and
The bond description (equal sharing vs. unequal sharing), rather than strict numerical cutoffs.
The important exception-style idea: C–H bonds are effectively nonpolar
The syllabus highlights that many C–H bonds are effectively nonpolar despite a small electronegativity difference. This reflects how AP Chemistry treats bond polarity as a model with practical thresholds.
Key points for C–H:
Carbon is slightly more electronegative than hydrogen, so is not zero.
Nevertheless, the electron sharing in a C–H bond is close enough to equal that the bond is commonly treated as nonpolar for predicting many molecular properties.
This “effective” nonpolarity is especially useful in recognising that many hydrocarbons consist largely of nonpolar bonds, so their electron distribution is not strongly separated by individual C–H bonds.
Using the model correctly (and avoiding common mistakes)
What you should and should not claim from “similar electronegativity”
From similar electronegativity, you should conclude:
The bond is covalent (electrons are shared).
The bond is nonpolar covalent (sharing is approximately equal).
The bond will not create a strong separation of charge within that bond.
You should avoid overextending the idea by:
Treating as a single rigid rule that guarantees all properties.
Assuming a tiny always implies perfectly identical electron density; “nonpolar” is a simplified but useful model.
Language that matches AP scoring expectations
High-utility phrasing for explanations:
“Because the atoms have similar electronegativities, they share the bonding electrons approximately equally, so the bond is nonpolar covalent.”
“Although C–H has a small electronegativity difference, it is often treated as effectively nonpolar, so it does not contribute strongly to charge separation.”
FAQ
Different scales assign slightly different numerical values, so $\Delta EN$ can shift.
In practice, classification relies more on the qualitative idea (“similar” vs “different”) than an exact cutoff, so conclusions usually stay the same.
Bond polarity is a model describing electron density, not an all-or-nothing physical switch.
C–H has a small asymmetry, but it is often too minor to matter for many predictions, so it is treated as effectively nonpolar.
Yes. Surrounding atoms can change electron distribution through inductive effects.
This does not change the bond’s basic classification from electronegativity alone, but it can change how important that bond’s polarity is in a larger molecule.
No single universal cutoff is required.
What matters is linking the idea correctly: small $\Delta EN$ implies nearly equal sharing, supporting a nonpolar covalent description.
Small bond polarities can add up or be outweighed by other bonds.
Also, molecular geometry and substituents can shift electron density, creating regions of slight partial charge even when many individual C–H bonds are treated as effectively nonpolar.
Practice Questions
Explain why the bond in is classified as nonpolar covalent.
States that the two atoms have identical (or very similar) electronegativity because they are the same element. (1)
Concludes that electrons are shared equally, so the bond is nonpolar covalent (no significant partial charges). (1)
A student claims that any bond with a non-zero electronegativity difference must be polar. Use the example of a C–H bond to evaluate this claim.
Identifies that C and H have a small but non-zero electronegativity difference. (1)
States that similar electronegativities lead to approximately equal sharing of bonding electrons. (1)
Explains that “nonpolar covalent” is an effective classification when charge separation is negligible. (1)
Applies this to C–H: despite the small difference, the bond is often treated as effectively nonpolar. (1)
Directly evaluates the claim as incorrect/overgeneralised, because non-zero can still correspond to effectively nonpolar sharing. (1)

