AP Syllabus focus: ‘Unequal electronegativity produces polar covalent bonds: the more electronegative atom is partially negative, and larger electronegativity differences in single bonds give larger bond dipoles.’
Polar covalent bonding explains why many molecules have uneven electron distributions even though electrons are shared. Understanding partial charges and dipoles helps you predict molecular polarity, intermolecular attractions, and qualitative behaviour in electric fields.
Polar covalent bonds from electronegativity differences
Electronegativity compares how strongly atoms attract shared bonding electrons. When two atoms with different electronegativities form a covalent bond, electron density is pulled toward the more electronegative atom, creating a polar covalent bond.
If electronegativities are equal, the bond is nonpolar (electron density shared symmetrically).
If electronegativities are unequal, the bond is polar (electron density shared asymmetrically).
In AP Chemistry, focus on the trend: bigger electronegativity difference in a single bond → larger bond dipole (stronger separation of charge along that bond).
Partial charges and electron density
Because electrons are negative, the atom that attracts bonding electrons more strongly becomes slightly negative, while the other becomes slightly positive.
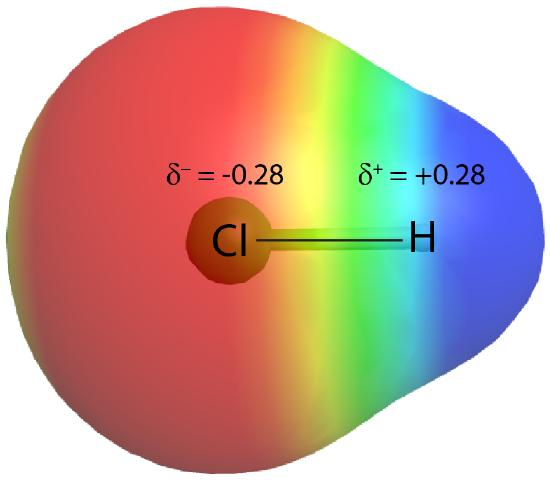
Electron density in HCl is shifted toward Cl, giving Cl a partial negative charge and H a partial positive charge. The color mapping (more negative vs. more positive regions) helps connect the idea of “electron density” to the macroscopic concept of bond polarity. Source
These are not full ionic charges; they are partial.
Partial charge ( or ). A fractional positive or negative charge on an atom caused by unequal sharing of electrons in a polar covalent bond.
Use these ideas consistently:
The more electronegative atom in a bond is labelled .
The less electronegative atom is labelled .
Partial charges reflect electron density, not a change in the number of protons; the atoms remain neutral overall unless an actual ion forms.
Partial charges are a model: they help explain attractions between polar molecules and why some bonds respond to electric fields.
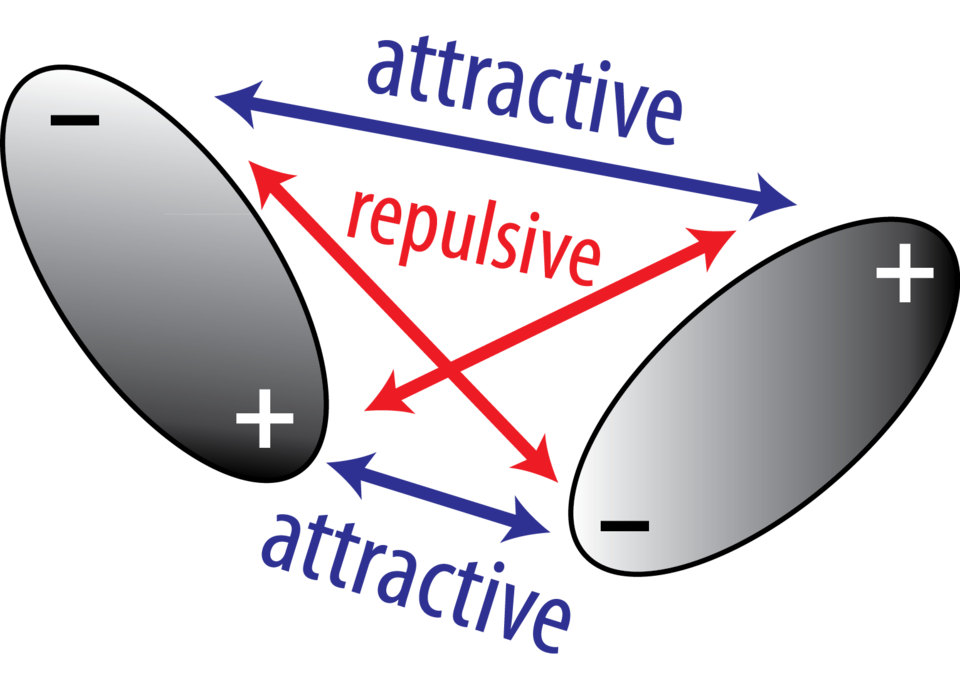
Schematic of dipole–dipole interactions between two polar molecules, showing how opposite poles (+/−) align to create an attractive electrostatic interaction. This diagram connects microscopic bond polarity to an important class of intermolecular forces that influence boiling points, solubility, and molecular organization. Source
Bond dipoles and dipole moments
A bond dipole is the separation of charge across a single bond due to a polar covalent interaction. It has:
Direction: toward the more electronegative atom (toward ).
Magnitude: increases as the electronegativity difference increases (for comparable bond lengths).
= dipole moment (commonly reported in debye, D)
= magnitude of separated charge (coulombs, C)
= distance between the charges (metres, m)
In chemistry diagrams, dipoles are often shown with an arrow pointing toward , indicating the direction of electron-density pull along the bond.
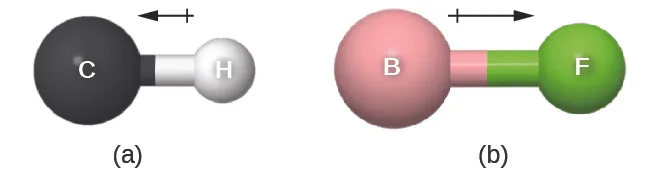
Bond dipole vectors are drawn with the arrow pointing toward the more electronegative (partially negative) end, and a plus sign marking the partially positive end. The contrast between a weakly polar bond (C–H) and a strongly polar bond (B–F) illustrates how a larger electronegativity difference corresponds to a larger bond dipole. Source
Connecting electronegativity difference to dipole size (single bonds)
For single bonds, a larger electronegativity difference generally means:
greater charge separation (larger effective )
therefore a larger bond dipole and larger dipole moment contribution from that bond
This relationship is qualitative on the AP Exam: you compare dipoles rather than calculate them.
Common pitfalls to avoid
Polar bond vs. polar molecule: A molecule can contain polar bonds yet have no overall molecular dipole if bond dipoles cancel by symmetry.
Partial charges are not oxidation states: and describe electron-density shifts, not integer electron transfer.
Bigger electronegativity difference doesn’t automatically mean “ionic”: in this subtopic, treat polarity as a continuum within covalent bonds and focus on bond dipoles in single bonds.
FAQ
If the separated charge is similar, a longer bond increases $r$, which can increase $\mu$.
In real bonds, changing bond length can also slightly change electron distribution, so the effect is not always perfectly proportional.
Polarity is continuous rather than all-or-nothing.
Even a small difference can create measurable charge separation, especially when many such bonds reinforce each other in an asymmetric structure.
A bond dipole is associated with one polar bond.
A molecular dipole is the vector sum of all bond dipoles (and lone-pair contributions), so geometry determines whether dipoles cancel.
Yes. Lone pairs are regions of high electron density and can create strong local electron-density asymmetry.
They can shift the overall charge distribution and change the direction and magnitude of the molecular dipole.
Dipole moments are inferred from how substances behave in electric fields and from spectroscopic measurements (e.g., microwave spectroscopy).
Reported values (often in debye, D) reflect the overall molecular dipole in the gas phase.
Practice Questions
(2 marks) In a single bond between atoms X and Y, Y is more electronegative than X. State (i) which atom is and (ii) the direction of the bond dipole arrow.
(i) Y is (1)
(ii) Arrow points from X towards Y / towards (1)
(5 marks) Three single bonds are listed: A–B, C–D, and E–F. The electronegativity differences are largest for A–B, intermediate for C–D, and smallest for E–F.
(a) Rank the bonds by increasing bond dipole magnitude. (2 marks)
(b) For bond A–B, describe how partial charges arise and how this relates to the idea of a dipole moment. (3 marks)
(a) E–F < C–D < A–B (2; 1 mark if one comparison correct, 2 marks if fully correct)
(b) Unequal sharing of bonding electrons due to electronegativity difference (1)
More electronegative atom becomes and the other (1)
Dipole moment arises from separated charge over a distance (mentions and idea qualitatively) (1)

