AP Syllabus focus: ‘In metallic solids, valence electrons are delocalized (a ‘sea’ of electrons) and are not associated with any single metal atom; use this model to explain metallic properties.’
Metals display a distinctive set of physical properties that can be explained by a single bonding model. Metallic bonding focuses on how valence electrons behave collectively in a solid and how that behavior controls conductivity, strength, and shape change.
Core model of metallic bonding
Metal atoms have relatively low ionization energies, so their valence electrons are not held tightly compared with nonmetals. In a metallic solid, those valence electrons are best modeled as belonging to the entire crystal rather than to individual atoms.
Metallic bonding: the electrostatic attraction between a lattice of positive metal ions and a mobile “sea” of delocalized valence electrons in the solid.
This model treats the metal as two interpenetrating parts:
An ordered array of metal cations (often described as metal “ions” or “kernels,” meaning nucleus + core electrons).
A continuous, mobile population of delocalized valence electrons that can move throughout the crystal.
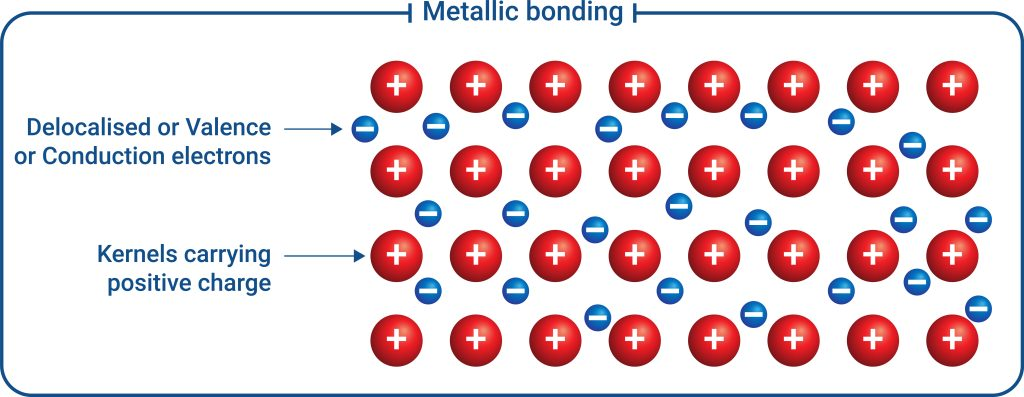
Diagram of the electron-sea model of metallic bonding: fixed positive metal kernels (cations) are surrounded by a mobile population of delocalized (conduction) electrons. This visual reinforces that the bonding is an electrostatic attraction between the cation lattice and the shared electron cloud rather than localized, directional bonds. Source
Delocalized electrons and “electron sea”
The key feature is electron mobility: valence electrons are not confined to one bond or one atom, so they can respond to electric fields and redistribute as the metal is stressed.
Delocalized electrons: valence electrons that are not associated with any single atom or single bond and are free to move through the entire metallic solid.
Because metallic bonding is non-directional (unlike many covalent bonds), the solid can often change shape without breaking a specific set of localized bonds.
Explaining metallic properties with the model
The AP focus is using the “sea of electrons” model to connect microscopic structure to macroscopic properties. When you explain a property, name the feature of the model (mobile electrons; cation lattice; non-directional attraction) and then link it to what is observed.
Electrical conductivity
Metals conduct electricity well in the solid (and molten) state because:
Delocalized electrons are mobile charge carriers.
When an electric potential is applied, electrons drift through the lattice, producing current.
The cations remain in fixed positions in the solid, but they do not need to move for charge to flow.
A common language cue for AP responses is: “mobile electrons move through the lattice under an applied field.”
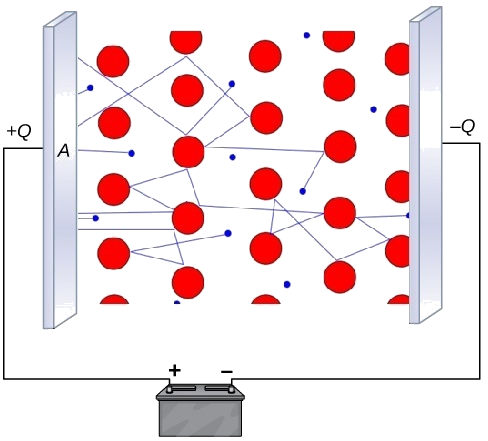
Schematic showing delocalized electrons moving through a metal when a potential difference is applied (battery connected), producing an electric current. The positive metal centers remain in place while the mobile electron sea responds to the electric field, which is the microscopic basis of metallic electrical conductivity. Source
Thermal conductivity
Metals are also strong thermal conductors because:
Delocalized electrons can transfer kinetic energy rapidly through the solid.
Collisions between electrons and the cation lattice distribute energy efficiently, helping heat spread quickly.
Malleability and ductility
Metals can be hammered into sheets (malleable) and drawn into wires (ductile) because metallic bonding does not depend on maintaining specific, directional bonds between fixed pairs of atoms.
When layers of cations shift under force, the electron sea redistributes to maintain attraction to the cations.
The crystal can deform while remaining held together by the overall electrostatic attraction between cations and delocalized electrons.
This explains why many metals bend rather than shatter: the bonding can “follow” the new arrangement.
Luster (metallic shine)
Metallic luster arises because:
Delocalized electrons can oscillate collectively in response to incoming light.
These oscillations lead to strong reflection (rather than light passing through), producing the characteristic shine of many metals.
Strength and melting point trends (qualitative)
The strength of metallic bonding depends on how strongly the electron sea attracts the cation lattice. Qualitatively, metallic bonding tends to be stronger when:
The metal forms more highly charged cations in the lattice (greater positive charge increases attraction).
There are more delocalized valence electrons per atom contributing to the electron sea.
The cations are smaller (higher charge density increases attraction for a given charge).
These factors help explain why some metals are harder and have higher melting points than others: stronger attraction between cations and the electron sea requires more energy to disrupt the lattice arrangement.
What to emphasise in AP-style explanations
When asked to “use the model,” anchor your reasoning in these statements:
Valence electrons are delocalized and not associated with any single metal atom.
The metal consists of a lattice of positive ions immersed in a sea of electrons.
The attraction is electrostatic and non-directional, enabling conductivity and deformation without fracture.
FAQ
In the model, electrons are shared by the whole crystal rather than assigned to one atom.
This is a solid-state description, not a claim about isolated gaseous atoms.
Conductivity depends on how easily electrons move through the lattice.
Electron scattering from defects, vibrations, and impurities can reduce mobility.
Higher temperature increases lattice vibrations.
More vibrations cause more electron scattering, typically lowering conductivity in metals.
In a solid, cations vibrate about fixed lattice positions.
They become mobile only when the metal melts.
Many metals reflect a broad range of visible wavelengths due to collective electron response.
A few (e.g., copper, gold) absorb specific wavelengths more strongly, giving colour.
Practice Questions
(1–3 marks) Explain, using the electron-sea model, why solid copper conducts electricity.
Mentions delocalised/mobile electrons in copper (1)
States electrons move through the lattice when a potential is applied (1)
Links moving electrons to current/electrical conduction (1)
(4–6 marks) Magnesium metal is harder and has a higher melting point than sodium metal. Use the metallic bonding model to explain both observations.
Describes metallic bonding as attraction between positive ions and delocalised electrons (1)
States Mg provides a greater positive charge on ions than Na (Mg vs Na) (1)
States Mg contributes more delocalised electrons per atom than Na (1)
Links greater charge and/or more electrons to stronger electrostatic attraction (1)
Links stronger attraction to greater hardness (ions held more firmly; deformation more difficult) (1)
Links stronger attraction to higher melting point (more energy needed to overcome bonding) (1)

