AP Syllabus focus: ‘Polar bonds have some ionic character, so bonding lies on a continuum; electronegativity difference is not the only factor—metal–nonmetal bonds are usually ionic and nonmetal–nonmetal bonds covalent.’
Chemical bonds are best viewed as a spectrum rather than two separate categories.
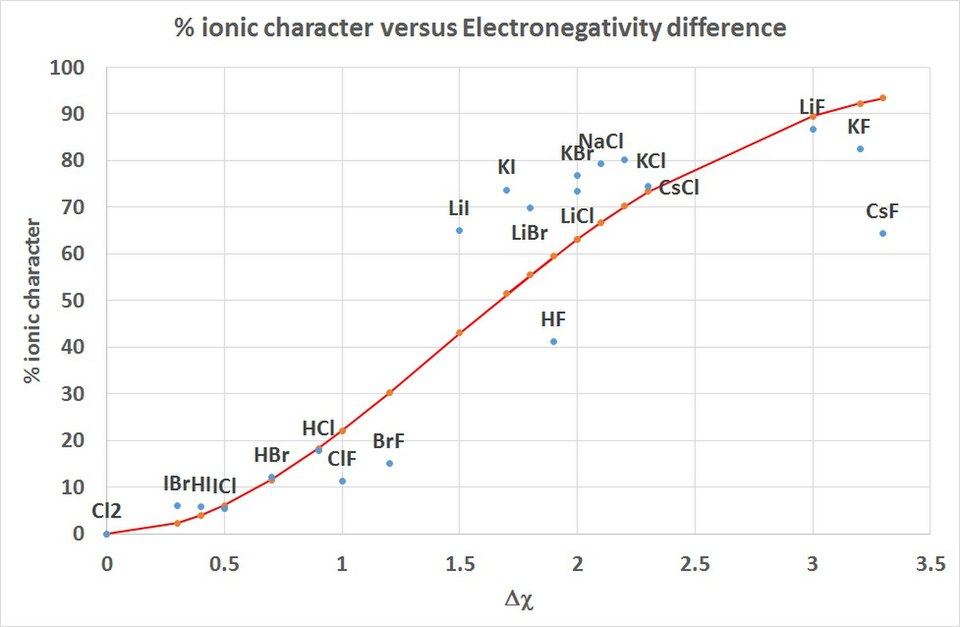
This graph shows the percent ionic character of a bond as a continuous function of electronegativity difference. As increases, the bond’s ionic character rises smoothly—illustrating why real bonds are best treated as points on a continuum rather than purely “ionic” or “covalent.” Source
This page explains why bonds can be partly ionic and partly covalent, and how to classify bonding using electronegativity and element type.
The ionic–covalent continuum
Bonding describes how electron density is distributed between atoms. In reality, most bonds are neither “perfectly ionic” nor “perfectly covalent,” because electron sharing can be unequal to varying degrees.
Ionic character: The extent to which a bond involves electron density being shifted toward one atom so strongly that it resembles ion formation (partial or full charge separation).
Nonpolar covalent is one extreme: electrons are shared about equally.
Ionic is the other extreme: electron density is effectively transferred, giving full charges (cations and anions).
Polar covalent bonds fall between these extremes: electrons are shared, but unequally, creating partial charges ( and ).
A key AP idea is that “ionic” compounds can still have some covalent character, and “covalent” molecules can have polar bonds with partial ionic character.
Electronegativity difference as a first-pass classifier
Electronegativity (EN) is a useful predictor because it reflects how strongly an atom attracts shared electrons.
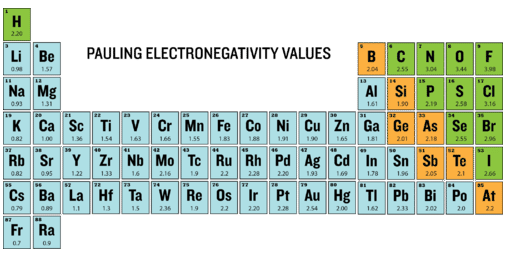
This periodic table is color-coded with Pauling electronegativity values, highlighting that EN generally increases toward the upper-right (with fluorine highest). It helps you quickly predict bond polarity (which atom is ) and why metal–nonmetal pairings often show much greater ionic character than nonmetal–nonmetal bonds. Source
Small EN difference → more equal sharing → more covalent character.
Large EN difference → more unequal sharing → greater ionic character.
However, AP Chemistry emphasises that electronegativity difference is not the only factor, so any EN-based cutoff should be treated as a guideline, not an absolute rule.
What EN difference does (and doesn’t) tell you
It helps predict direction of bond polarity (which atom is ).
It suggests relative ionic character when comparing similar types of bonds.
It does not, by itself, guarantee whether a substance forms a molecular structure or an ionic lattice, because classification also depends on the types of elements involved.
Element type matters: metal–nonmetal vs nonmetal–nonmetal
The syllabus explicitly states a strong classification rule:
Metal–nonmetal bonds are usually ionic
Metals tend to form cations (low ionisation energy, fewer valence electrons).
Nonmetals tend to form anions (higher EN, strong attraction for electrons).
The result is often best modelled as an ionic solid with ions as the key particles.
Nonmetal–nonmetal bonds are usually covalent
Both atoms have relatively high EN and tend to share electrons rather than fully transfer them.
The result is often best modelled with covalent bonds within discrete molecules or network structures (classification depends on the compound, but the bonding is still covalent in nature).
This “element-type” guideline often overrides borderline EN cases, especially on qualitative AP questions.
Why “ionic” compounds can have covalent character
Even when a compound is classified as ionic overall, individual cation–anion interactions can include electron density sharing.

This diagram illustrates polarization of an anion by a nearby cation: the anion’s electron cloud becomes distorted toward the cation. Greater polarization (often from smaller, higher-charge cations) reduces full charge separation and increases covalent character in an otherwise “ionic” interaction. Source
A small, highly charged cation can strongly attract and distort the electron cloud of an anion, increasing covalent character.
A large, low-charge cation tends to interact more purely electrostatically, increasing ionic character.
You do not need to memorise an algorithm, but you should be able to justify “more covalent character” using qualitative ideas about charge density (charge per size) and how strongly ions can attract electron density.
How to classify bonds in AP-style reasoning
Stepwise decision approach (qualitative)
Identify whether the bond is metal–nonmetal or nonmetal–nonmetal.
Use electronegativity difference to support whether the bond is “more ionic” or “more covalent.”
If needed, refine using ion characteristics:
higher cation charge and smaller cation size → more covalent character
lower cation charge and larger cation size → more ionic character
Language that earns credit
Use “continuum” wording: “This bond has significant ionic character” rather than forcing a binary label.
Distinguish:
bond polarity (unequal sharing in a bond)
compound classification (overall ionic vs covalent based on particles/element types)
Common pitfalls to avoid
Treating EN difference as a strict cutoff rather than a trend-based tool.
Calling any polar bond “ionic” without acknowledging partial charges and shared electron density.
Ignoring that metal–nonmetal combinations are typically best classified as ionic even if the EN difference seems moderate.
FAQ
Use both ideas together: EN difference for “more/less ionic character,” and element types for the overall classification. In borderline cases, “metal–nonmetal usually ionic” is often the deciding argument.
Greater covalent character is favoured by high charge density, especially:
small, highly charged cations
large, easily distorted anions
These conditions increase electron-density sharing between ions.
“Ionic” is a broad classification. Differences in ion size and charge can change how strongly electron density is pulled toward one ion, altering the degree of polarisation and therefore the relative ionic character.
Yes. Bonds within a polyatomic ion are typically covalent (often polar covalent), while the attraction between that ion and an oppositely charged ion in a salt is classified as ionic overall.
Because ionic character changes smoothly with EN difference and also depends on factors beyond EN (such as ion polarisation). Any numerical cutoff is a convention used for convenience, not a physical boundary.
Practice Questions
(2 marks) Explain why bonding is described as an ionic–covalent continuum rather than two distinct types.
States that electron density can be shared unequally to different extents / most bonds are intermediate (1)
Links to polar covalent bonds having partial ionic character (partial charges) (1)
(5 marks) A student claims: “If the electronegativity difference is large, the bond must be ionic; if it is small, the bond must be covalent.” Evaluate this claim using AP-appropriate classification ideas, including the role of metal/non-metal bonding and the concept of ionic character.
Recognises EN difference is a useful guideline for bond polarity/ionic character (1)
States bonding lies on a continuum; polar bonds have some ionic character (1)
Explains EN difference is not the only factor / not an absolute rule (1)
States metal–nonmetal bonds are usually ionic and nonmetal–nonmetal bonds usually covalent (1)
Uses correct comparative language (e.g., “more ionic character” rather than absolute) (1)

