AP Syllabus focus: ‘Do not use “London dispersion forces” as a synonym for “van der Waals forces”; distinguish LDF from broader, general intermolecular attractions.’
Intermolecular-force vocabulary is often used loosely, but AP Chemistry expects precision. This page clarifies what London dispersion forces (LDF) are, what van der Waals forces means, and how to avoid common wording mistakes.
Core Terminology: What Each Phrase Means
London dispersion forces (a specific force)
London dispersion forces (LDF): Attractions arising when momentary (instantaneous) electron-density fluctuations create a temporary dipole that induces a dipole in a nearby particle, leading to a net Coulombic attraction.
LDF are a single, specific type of intermolecular attraction. They occur between all particles (because all particles have electrons), but the phrase “London dispersion forces” should be reserved for this temporary-dipole/induced-dipole mechanism.
A key language point: saying “the molecule has van der Waals forces” is vague; saying “the dominant attraction is LDF” is specific.
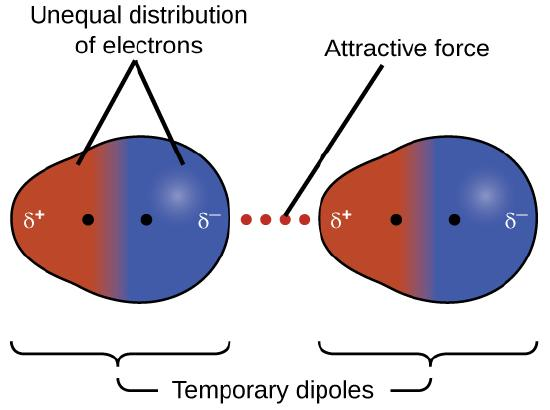
London dispersion forces arise when an instantaneous (temporary) dipole forms in one particle and induces a complementary dipole in a neighboring particle. The diagram explicitly labels the unequal electron distribution, the resulting temporary dipoles ( and ), and the net attractive force between them. Source
van der Waals forces (a broader umbrella term)
van der Waals forces: A broad label for weak, noncovalent attractions between neutral particles, commonly used as an umbrella term that includes London dispersion forces and other intermolecular attractions (depending on the course/text context).
Because van der Waals is broader, it is not interchangeable with LDF. The syllabus requirement is essentially a terminology rule: do not use “London dispersion forces” as a synonym for “van der Waals forces.”
How to Use the Terms Correctly (AP-Appropriate Precision)
Acceptable statements
“LDF are present between all atoms and molecules.”
“The intermolecular attractions can be described as van der Waals forces, including LDF.”
“In this nonpolar substance, the relevant intermolecular attraction is London dispersion forces.”
Statements to avoid (and better replacements)
Avoid: “This substance has van der Waals forces, meaning London dispersion forces.”
Better: “This substance has van der Waals attractions, primarily London dispersion forces.”
Avoid: “Van der Waals forces are the same as LDF.”
Better: “LDF are one type of van der Waals attraction.”
Why the Distinction Matters in Explanations
Preventing overgeneralisation
If you call all weak intermolecular attractions “LDF,” you lose the ability to communicate which attraction is being discussed. AP Chemistry often rewards identifying the correct category of interaction rather than using a generic label.
Communicating mechanism vs category
LDF communicates a mechanism (temporary dipole induces another dipole).
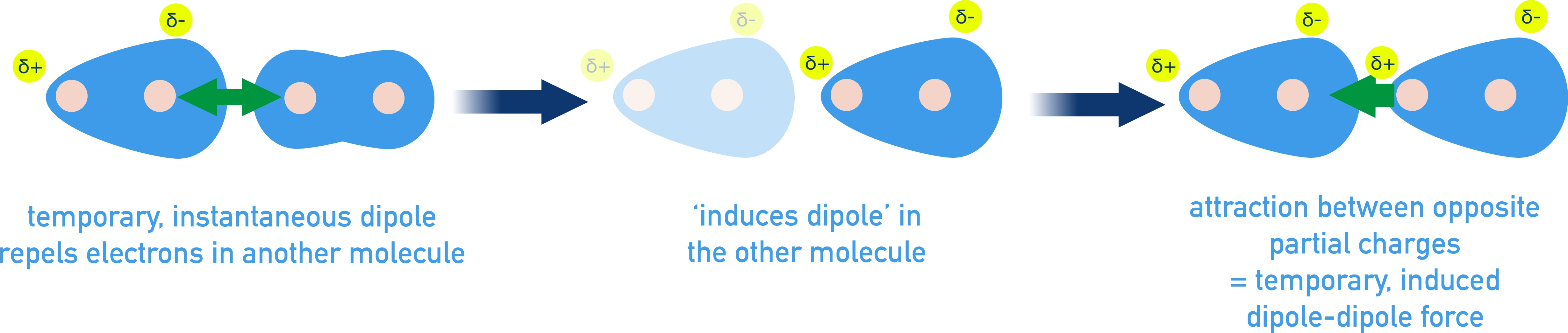
This sequence diagram shows how a momentary electron-density fluctuation produces an instantaneous dipole, which then induces a dipole in a neighboring molecule. The final panel highlights that the attraction is between opposite partial charges, reinforcing that LDF is a specific mechanism rather than a catch-all term. Source
van der Waals communicates a category (a collection of weak attractions between neutral particles).
When writing explanations, choose the term that matches what the question asks:
If asked for the type/mechanism, state London dispersion forces.
If asked for a general description of weak attractions, van der Waals may be acceptable, but specifying LDF is usually clearer.
Common Exam Pitfalls and How to Avoid Them
Pitfall: treating “van der Waals” as a single force
Students sometimes write as if “van der Waals” is one distinct interaction with one cause. On AP-style items, it is safer to treat van der Waals as an umbrella and then name LDF when you mean temporary-dipole attractions.
Pitfall: using LDF to mean “any weak intermolecular force”
In AP Chemistry, LDF is not a synonym for “weak forces” or “intermolecular forces in general.” Use intermolecular forces (IMFs) as the general term, and use LDF for the specific temporary-dipole attraction.
FAQ
Not always. Some sources use “van der Waals” very broadly (nearly all weak intermolecular attractions), while others define narrower subsets.
For AP-style clarity, treat it as an umbrella term and specify LDF when you mean temporary-dipole attractions.
If a question asks for a general description of weak attractions, it may be acceptable.
If a question asks for the specific attraction responsible, naming London dispersion forces is typically more precise and safer.
“Intermolecular forces (IMFs)” is the broadest classroom term for attractions between particles.
“Van der Waals” commonly refers to a subset of weaker, noncovalent attractions between neutral particles, so it’s not always identical in scope to “IMFs.”
Because chemical explanations often depend on identifying the relevant interaction mechanism.
Saying “LDF” signals temporary-dipole interactions specifically, rather than vaguely indicating “some weak attraction.”
Ask: am I describing a mechanism or a category?
Mechanism (temporary dipoles inducing dipoles) → write London dispersion forces.
Category (general weak attractions) → write van der Waals attractions, optionally followed by the specific type.
Practice Questions
(1–3 marks) State whether the term “London dispersion forces” should be used as a synonym for “van der Waals forces”, and justify your answer.
1 mark: Correct statement that they should not be used as synonyms.
1 mark: States that LDF are a specific type of intermolecular attraction.
1 mark: States that van der Waals refers to a broader set/umbrella of weak attractions (may include LDF).
(4–6 marks) A student writes: “Between these molecules there are van der Waals forces, i.e. London dispersion forces.” Explain what is imprecise about this statement and rewrite it in two scientifically accurate ways, each with different emphasis.
1 mark: Identifies the imprecision: equating/using LDF as a synonym for van der Waals.
1 mark: States that van der Waals is an umbrella term (broader category).
1 mark: States that LDF are one type within that umbrella (specific mechanism).
1 mark: Provides rewrite emphasising category then type (e.g., “van der Waals attractions, including LDF”).
1 mark: Provides rewrite emphasising the specific interaction (e.g., “the dominant attraction is LDF”).
1 mark: Uses clear, chemically appropriate language (neutral particles/weak noncovalent attraction) without introducing incorrect force types.

