AP Syllabus focus: ‘Polar molecules experience dipole–dipole attractions; interaction strength depends on dipole magnitude and relative orientation and acts in addition to London dispersion forces.’
Polar molecules have uneven electron density that creates permanent partial charges. These permanent dipoles attract one another in predictable ways, shaping physical properties such as boiling points, viscosity, and how molecules arrange in condensed phases.
Dipole moments (molecular polarity)
A dipole moment describes the separation of positive and negative charge in a bond or an entire molecule. Bond dipoles arise from electronegativity differences, and molecular dipoles depend on both bond polarity and 3D geometry (dipoles can reinforce or cancel).
Dipole moment — a vector measure of charge separation in a bond or molecule, with both magnitude and direction (toward the more electronegative atom).
Chemists often represent polar bonds with partial charges ( and ) and an arrow showing the direction of electron pull. For polyatomic molecules, the net dipole is the vector sum of all bond dipoles and lone-pair effects.
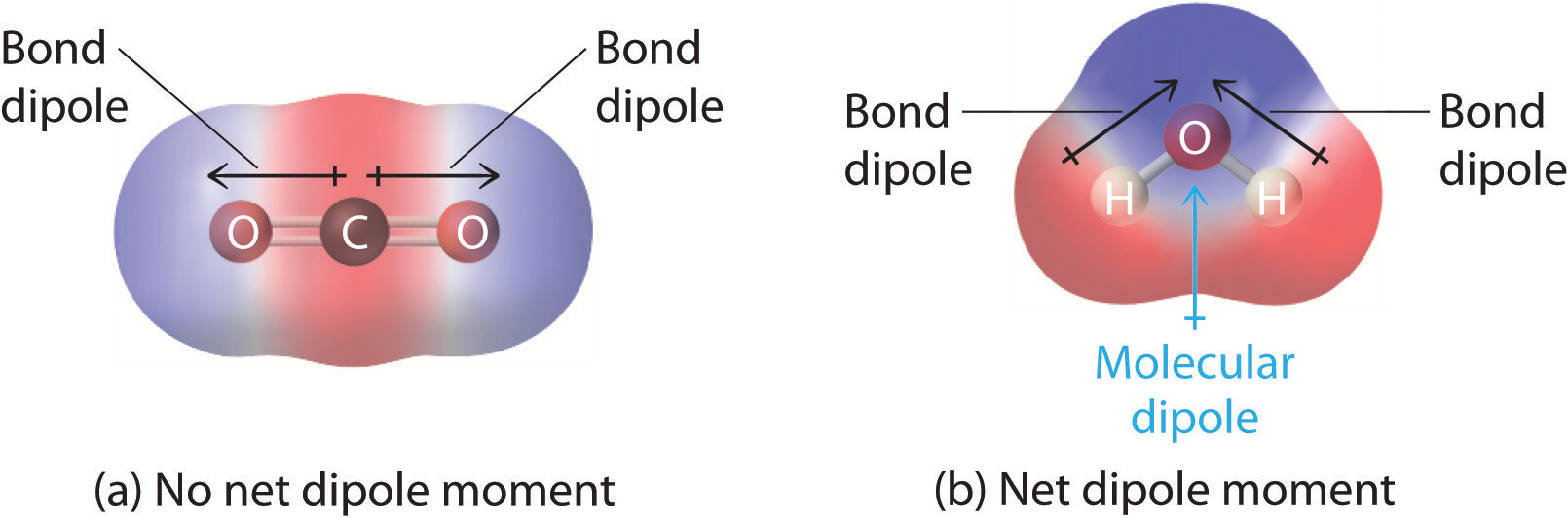
This diagram contrasts a linear molecule (CO) whose equal-and-opposite bond dipoles cancel to give zero net dipole moment with a bent molecule (HO) whose bond dipoles add to produce a nonzero molecular dipole. It reinforces that molecular polarity depends on both bond polarity and 3D geometry (vector addition), not just the presence of polar bonds. Source
= dipole moment (commonly reported in debye, D)
= magnitude of separated charge
= distance between the centres of positive and negative charge
This relationship is conceptual at the molecular level: larger charge separation and/or larger separation distance generally increase dipole moment.
Key implications for identifying polar molecules
A molecule can contain polar bonds but be nonpolar overall if its geometry is symmetric and bond dipoles cancel.
A molecule with an asymmetric shape often has a nonzero net dipole, making it polar.
The dipole moment is a vector, so orientation in space matters.
Dipole–dipole interactions
Dipole–dipole interactions are Coulombic attractions between the positive end of one polar molecule and the negative end of another.
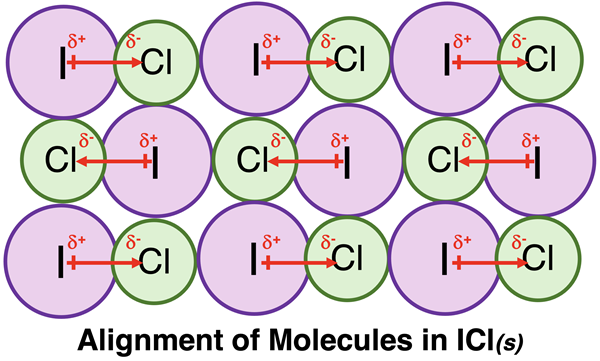
This image depicts an ordered alignment of polar ICl molecules in the solid state, with iodine ends positioned near chlorine ends on neighboring molecules. The repeated head-to-tail arrangement illustrates why dipole–dipole interactions are strongly orientation-dependent and can significantly stabilize condensed phases when favorable packing is possible. Source
These attractions are directional (unlike many non-directional interactions), meaning molecular alignment strongly affects interaction strength.
What “acts in addition to London dispersion forces” means
Even polar molecules experience London dispersion forces due to instantaneous fluctuations in electron density. Dipole–dipole attractions are an extra contribution for polar molecules, increasing the overall intermolecular attraction compared with similarly sized nonpolar molecules.
What controls dipole–dipole strength?
The syllabus emphasis is that strength depends on dipole magnitude and relative orientation.
1) Dipole magnitude
Larger molecular dipole moments tend to produce stronger dipole–dipole attractions because:
The partial charges are effectively “more intense” (greater charge separation).
The electrostatic attraction between opposite ends is stronger at a given separation.
2) Relative orientation (alignment)
Dipole–dipole forces are strongest when molecules orient so that:
The region of one molecule is close to the region of another.
They are weaker (or even partially cancelling in effect) when:
Like ends approach ( near or near ), increasing repulsion.
Thermal motion disrupts alignment, reducing the average attractive contribution over time.
Because molecules in liquids and gases rotate and translate constantly, dipole–dipole interactions should be understood as time-averaged attractions influenced by temperature.
3) Distance and molecular packing
While not unique to dipoles, distance still matters:
Closer molecules experience stronger electrostatic interactions.
Shapes that allow closer approach or better alignment can increase the effective dipole–dipole contribution.
How dipole–dipole interactions show up in observable properties
Dipole–dipole attractions help explain trends when comparing substances of similar molar mass and similar shapes:
Boiling point: stronger dipole–dipole attractions generally raise boiling points because more energy is required to separate molecules into the gas phase.
Melting point: may increase when dipoles enable efficient packing and strong attractions in the solid; however, packing effects can complicate simple comparisons.
Viscosity and surface tension: stronger intermolecular attractions (including dipole–dipole) often increase both, because molecules resist flowing past one another and resist separation at the surface.
Vapour pressure: stronger attractions tend to lower vapour pressure at a given temperature, since fewer molecules have sufficient energy to escape to the gas phase.
When comparing real substances, remember that observed properties reflect the combined effect of dipole–dipole interactions plus London dispersion forces, and the balance depends on both polarity and molecular size/shape.
FAQ
Common methods infer dipole moments from how gases or liquids respond to an applied electric field.
Techniques include microwave spectroscopy (rotational transitions) and dielectric measurements, which link molecular polarity to bulk electrical behaviour.
The debye (D) is a convenient-sized unit for molecular-scale dipoles.
$1\ \text{D} \approx 3.336 \times 10^{-30}\ \text{C·m}$, so typical molecular dipoles fall in the range of about 0–10 D.
Yes. Different conformations can change the vector sum of bond dipoles.
If conformers interconvert rapidly, the reported dipole moment may reflect an average weighted by how much each conformation is present.
In gases, molecules are far apart, so interactions are brief and weaker on average.
In liquids, molecules are close together; dipoles interact continually but are also disrupted by thermal motion, making the interaction a dynamic, time-averaged effect.
Dielectric constant reflects how well a material reduces internal electric fields by dipole alignment and polarisation.
Stronger or more easily oriented dipoles generally increase dielectric constant, but molecular mobility and specific local structure also strongly influence it.
Practice Questions
(2 marks) Explain why dipole–dipole interactions occur between molecules of chloromethane, CHCl, but not between molecules of methane, CH.
CHCl is polar / has a permanent dipole due to electronegativity difference and asymmetric charge distribution (1)
CH is non-polar / no permanent dipole, so no dipole–dipole attractions (1)
(5 marks) Molecules A and B have similar molar masses. A has a larger dipole moment than B. (a) Describe what a dipole–dipole interaction is, using and labels. (2 marks) (b) Explain how relative orientation affects the strength of dipole–dipole interactions. (2 marks) (c) State one macroscopic property that would typically differ between A and B due to dipole–dipole interactions, and give the expected direction of the difference. (1 mark)
(a) Attraction between end of one polar molecule and end of another (1)
(a) Must reference permanent dipoles in polar molecules (1)
(b) Strongest when opposite ends align close together; weaker when misaligned/like ends approach (1)
(b) Orientation dependence described as directional / alignment matters (1)
(c) Any one: higher boiling point for A / lower vapour pressure for A / higher viscosity for A (1)

