AP Syllabus focus: ‘London dispersion forces arise from Coulombic attraction between temporary, fluctuating dipoles; for large molecules, LDF can be the strongest net intermolecular force.’
London dispersion forces are universal intermolecular attractions. They arise even in nonpolar substances because electrons are always moving, briefly creating dipoles that induce dipoles in nearby particles and generate net attraction.
What London Dispersion Forces Are
London dispersion forces (LDF) are attractions caused by electron motion rather than permanent charges. Because they depend on temporary, fluctuating dipoles, LDF occur between all atoms and molecules, including noble gases and nonpolar molecular substances.
London dispersion forces (LDF): Intermolecular attractions arising from Coulombic attraction between temporary, fluctuating dipoles in adjacent particles.
Even though the dipoles are short-lived, enormous numbers of them form and vanish continuously in any sample, producing measurable cohesive forces.
How Temporary Dipoles Form
At any instant, electrons in a particle are not perfectly evenly distributed. A momentary imbalance creates a temporary dipole with a partial negative region (electron-rich) and a partial positive region (electron-poor). That temporary dipole can then induce a dipole in a nearby particle by distorting its electron cloud.

Schematic showing how an instantaneous (temporary) dipole in one neutral atom/particle polarizes a neighboring atom to create an induced dipole. The diagram emphasizes that even without permanent polarity, transient charge separation can create an electrostatic attraction between adjacent particles—this is the microscopic origin of London dispersion forces. Source
Key steps at the particle level:
Random electron motion produces a brief, uneven electron distribution in particle A.
Particle A develops a temporary dipole.
The electric field from particle A distorts particle B’s electron cloud.
Particle B develops an induced dipole oriented to maximize attraction.
The opposite partial charges attract via Coulombic forces, and the interaction persists until the fluctuations change.
Because the direction and magnitude of these dipoles change constantly, LDF are best viewed as an average attractive effect over time.
Why LDF Can Be Strong (Especially in Large Molecules)
The syllabus emphasis that “for large molecules, LDF can be the strongest net intermolecular force” reflects how dispersion forces scale with electron cloud size and contact area. Larger molecules generally have:
More electrons
Larger, more easily distorted electron clouds
More opportunities for simultaneous induced-dipole interactions along the molecule
As a result, the cumulative LDF between two large molecules can exceed other intermolecular attractions present in smaller substances, even if those other attractions are individually strong.
Factors That Increase LDF Strength
Two closely related ideas control LDF magnitude: polarizability and surface contact.
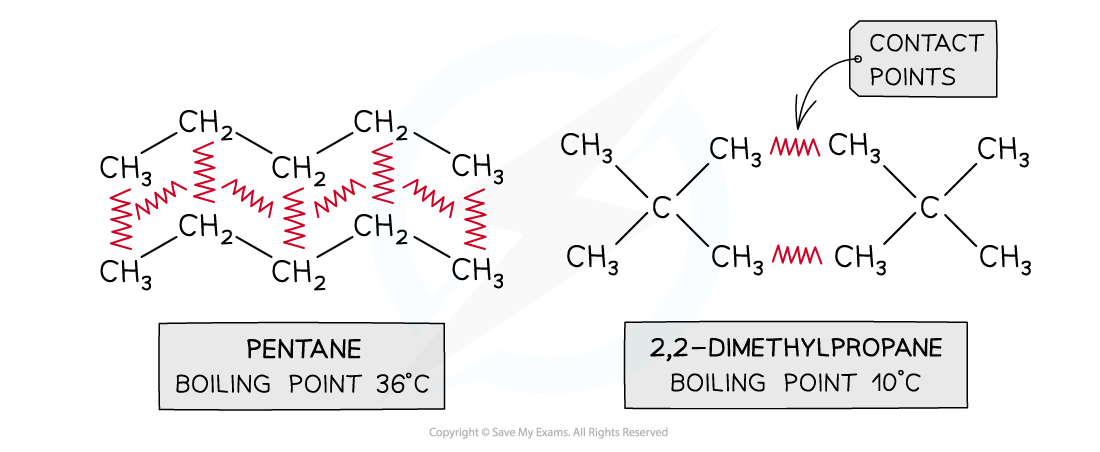
Diagram comparing isomers with the same electron count but different shapes, highlighting how greater surface area increases the number of intermolecular “contact points.” More contact points allow more simultaneous induced-dipole interactions, strengthening London dispersion forces and typically raising boiling points relative to more compact, branched isomers. Source
Polarizability: The ease with which a particle’s electron cloud can be distorted to form an induced dipole.
A more polarizable particle forms larger induced dipoles, which increases the Coulombic attraction.
Main trends AP Chemistry expects you to reason about:
More electrons → stronger LDF
Heavier atoms (down a group) are typically more polarizable.
Larger molecular molar mass often correlates with stronger LDF (within a related set of substances).
Larger electron cloud volume → stronger LDF
Valence electrons farther from the nucleus are held less tightly and distort more easily.
Greater surface area of contact → stronger LDF
Long, relatively linear molecules can align to maximize contact, strengthening attractions.
Compact or highly branched shapes reduce effective contact area, weakening attractions (all else equal).
Closer proximity → stronger LDF
Like all electrostatic attractions, shorter intermolecular distances increase interaction strength; efficient packing can therefore enhance LDF in condensed phases.
Interpreting “Temporary” Correctly
“Temporary” does not mean “negligible.” In a macroscopic sample, the constant formation of many induced dipoles leads to a persistent average attraction that helps explain why nonpolar substances can exist as liquids or solids at accessible temperatures. LDF are therefore a foundational explanation for cohesion in nonpolar matter and for trends that grow more pronounced as particle size increases.
Common Pitfalls to Avoid
Do not describe LDF as requiring a polar molecule; all particles experience LDF.
Do not treat dispersion as a rare event; it is continuous, arising from ongoing electron motion.
Do not assume small nonpolar molecules have strong LDF; size and polarizability are the dominant drivers of strength.
FAQ
Temporary dipoles reflect instantaneous electron probability distributions. Even in a ground state, electron density is not a static “shell”; it is a changing distribution described by a wavefunction.
Electron correlation (how electrons avoid each other) contributes to momentary charge imbalances that can synchronise between nearby particles.
Branching makes molecules more compact, reducing surface contact when molecules approach.
Less contact means fewer areas where induced dipoles can align favourably at the same time, lowering total dispersion attraction.
Yes. Different regions of a flexible molecule can approach closely enough for temporary dipoles to induce attractions between segments.
These intramolecular dispersion attractions can subtly influence preferred conformations, especially in large, electron-rich structures.
Polarity refers to a permanent separation of charge (a permanent dipole moment). Polarizability is about how easily the electron cloud can be distorted to create an induced dipole.
A molecule can be nonpolar but highly polarizable.
The underlying ability to polarise is not primarily temperature-controlled, but temperature affects average separation and motion.
At higher temperatures, particles spend less time close together in condensed phases, reducing the effectiveness of dispersion attractions in keeping substances together.
Practice Questions
(1–3 marks) Explain, at the particle level, how London dispersion forces arise between two atoms of argon.
States that electron distribution fluctuates to create a temporary dipole in one atom (1).
Explains that this dipole induces a dipole in the neighbouring atom by distorting its electron cloud (1).
States that Coulombic attraction occurs between opposite partial charges of the temporary/induced dipoles (1).
(4–6 marks) Substance X and substance Y are nonpolar molecular substances. X has a much higher boiling point than Y. Using only London dispersion forces, give two structural or particle-based reasons why X could have stronger intermolecular attractions than Y, and explain each reason.
Identifies that X could have more electrons / higher molar mass / larger electron cloud than Y (1).
Explains this increases polarizability and leads to larger induced dipoles / stronger dispersion attraction (1).
Identifies that X could have a less branched / more extended shape giving greater surface contact than Y (1).
Explains greater surface contact allows more simultaneous dispersion interactions / stronger net attraction (1).
Identifies that larger size can make LDF the strongest net intermolecular force in large molecules (1).
Overall explanation links both reasons explicitly to stronger LDF and hence higher boiling point (1).

