AP Syllabus focus: ‘Qualitatively compare dipole–dipole and ion–dipole forces by considering the signs of partial charges in dipoles and how those charges interact with ions or nearby dipoles.’
Understanding intermolecular attractions is easier when you track charge signs. This page shows how partial charges (, ) determine the geometry and relative strength of dipole–dipole versus ion–dipole interactions.
Core idea: attraction depends on charge signs
Partial charges in polar bonds
Partial charge (, ): A fractional positive or negative charge on an atom in a polar bond caused by unequal electron sharing.
A polar molecule has regions of and . Intermolecular attractions are strongest when opposite charge regions align closely and directly.
Electrostatic basis (qualitative)
= electrostatic interaction energy (relative)
= charge on region/particle 1 (full or partial)
= charge on region/particle 2 (full or partial)
= separation distance between interacting charges (relative)
This proportionality explains why interactions strengthen with larger charge magnitudes and shorter distances; it also highlights why replacing a partial charge with a full ionic charge usually increases attraction.
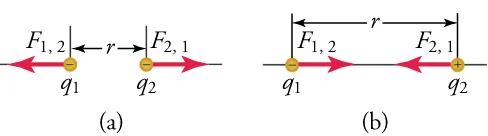
The figure shows how the sign of interacting charges controls the direction of the electrostatic force. Like charges repel and unlike charges attract, with the interaction occurring along the line connecting the charges and depending on the separation distance . This same sign-based reasoning is what you apply when you line up with (or ions with the opposite end of a dipole). Source
Dipole–dipole interactions using partial charges
What is interacting?
Dipole–dipole interaction: Attraction between the end of one polar molecule and the end of another polar molecule.
To compare dipole–dipole interactions, focus on:
Signs: must face for attraction; like signs produce repulsion.
Alignment (orientation): molecules can rotate, so attractions are strongest when dipoles line up “head-to-tail.”
Dipole magnitude: larger bond polarity and molecular geometry that produces a larger net dipole increases the possible attraction.
How to reason with partial charges
Identify the most positive H/atom (often bonded to an electronegative atom) as likely .
Identify the most electronegative atoms as likely .
Predict the lowest-energy arrangement: of one molecule nearest of another.
Recognise that a molecule can experience both attractive and repulsive contacts depending on how it approaches a neighbour.
Ion–dipole interactions using partial charges
What is interacting?
Ion–dipole interaction: Attraction between a full ionic charge (cation or anion) and the oppositely charged end of a polar molecule (its or region).
An ion has a full charge (e.g., , , ), while the polar molecule provides partial charges. Using signs:
A cation is attracted to the molecule’s end.
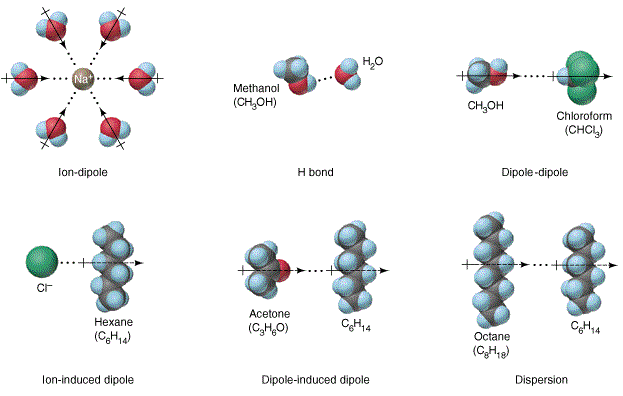
This hydration schematic depicts an ion–dipole interaction between a cation and surrounding water molecules. The water molecules orient so the oxygen end (electron-rich, ) points toward the positive ion, while the hydrogen ends () point away. The image emphasizes that ion charge strongly organizes nearby dipoles into a preferred orientation. Source
An anion is attracted to the molecule’s end.
How to compare ion–dipole vs dipole–dipole with partial charges
When comparing, keep the distance and general size in mind, then prioritise charge magnitude:
Ion–dipole often stronger because is typically larger than on an atom in a dipole.
A or ion generally interacts more strongly than a or ion with the same dipole (greater ).
Orientation still matters, but the ion’s electric field strongly biases the polar molecule to rotate so the correct end points toward the ion.
Practical comparison workflow (what AP expects you to do)
Step-by-step qualitative comparison
Label each polar molecule with / at the relevant atoms.
For dipole–dipole:
Check whether a plausible approach places near (attraction) or near / near (repulsion).
Consider whether the molecular shape allows good alignment (stronger) or forces poorer alignment (weaker).
For ion–dipole:
Pair cation → and anion → .
Compare likely strength by considering full vs partial charge and how close the ion can approach the charged end.
Language to use in explanations
“Attraction occurs between the oppositely charged regions (ion with end, or with ).”
“This interaction is stronger/weaker because the interacting charges are full vs partial, and because distance/orientation affects Coulombic attraction.”
FAQ
Use bond polarity plus molecular context: atoms with lone pairs (O, N, halogens) are commonly $\delta^-$. Atoms bonded to several electronegative neighbours become more $\delta^+$ due to electron withdrawal.
No. Treat $\delta$ qualitatively. You may reference “larger” or “smaller” partial charges based on relative electronegativity differences, without calculating exact fractions of $e$.
A neutral polar molecule has separated charge regions. The anion interacts with the $\delta^+$ region; neutrality only means the total charge sums to zero, not that charge is uniformly distributed.
Higher charge and smaller ionic radius increase charge density, strengthening attraction to a given $\delta$ end. For example, Mg$^{2+}$ typically attracts polar molecules more strongly than Na$^+$.
Partial charges describe local charge signs on atoms; dipole moment describes the molecule’s overall separation of charge (a vector). For sign-based comparisons, $\delta^+/\delta^-$ is usually the most direct.
Practice Questions
(2 marks) Molecules of chloromethane, CHCl, attract each other. Using partial charges, describe one favourable orientation for a dipole–dipole attraction.
Identifies on Cl and on the C/H end of another molecule (1)
States alignment with near (head-to-tail) to give attraction (1)
(5 marks) An Na ion is near a water molecule. Using partial charges, explain (i) which end of water is attracted to Na, and (ii) why this ion–dipole attraction is typically stronger than a water–water dipole–dipole attraction.
States O is and H atoms are in HO (1)
States Na is attracted to the oxygen end (1)
Notes ion has a full positive charge whereas water’s ends are partial charges (1)
Links larger charge magnitude to stronger Coulombic attraction (1)
Mentions orientation/rotation of water aligns toward the ion, increasing attraction (1)

