AP Syllabus focus: ‘For a first‑order reaction, ln[A] versus time is linear; the half‑life is constant and t1/2 = 0.693/k.’
First-order kinetics links how concentration changes over time to a simple exponential model. These ideas let you recognise first-order behaviour from graphs, extract the rate constant, and interpret a constant half-life.
What “first-order” means
In a first-order reaction, the instantaneous rate depends linearly on the reactant concentration.
First-order reaction: A reaction for which the rate is directly proportional to the concentration of a single reactant, so doubling that concentration doubles the rate.
This model is commonly written for a single reactant as “rate is proportional to ,” which implies that decreases exponentially rather than by a fixed amount each second.
Integrated rate law (first-order)
The integrated rate law connects concentration and time for a first-order process. It is the key tool for using concentration–time data to test first-order behaviour and to relate a measured concentration at time to the initial concentration.
A defining graphical feature from the AP syllabus is that versus time is linear.
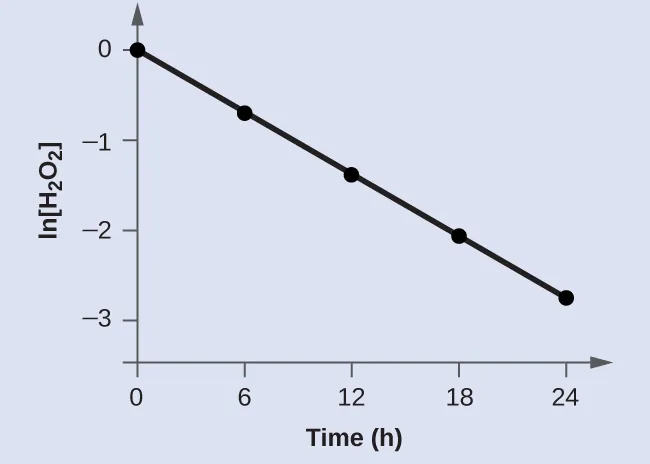
Plot of versus time showing a straight-line trend for a first-order process. The linearity supports the integrated rate-law form , where the slope is negative and equal to . Source
Linearity here means points fall on a straight line (within experimental uncertainty), supporting first-order kinetics under the same conditions.
= natural log of concentration at time (dimensionless)
= time elapsed (s, min, etc.)
= first-order rate constant ()
= natural log of initial concentration (dimensionless)
= time for to decrease to half its current value (same units as )
= (dimensionless)
Because the equation is in the form “,” a plot of versus gives:
Slope (so is the positive magnitude of the slope)
Intercept
What the straight-line test tells you
When vs. is linear:
The reaction is consistent with first-order dependence on under those conditions.
The rate constant is constant across the dataset (same temperature, catalyst status, solvent, etc.).
A larger corresponds to a steeper negative slope and faster decay of .
Half-life for first-order reactions
The AP syllabus emphasises that the half-life is constant for first-order kinetics. “Constant” means it does not depend on the starting concentration; it depends only on (and thus the conditions that affect ).
How to interpret “constant half-life”
For a first-order process:
The time to go from to is always .
The next halving, from to , takes the same again.
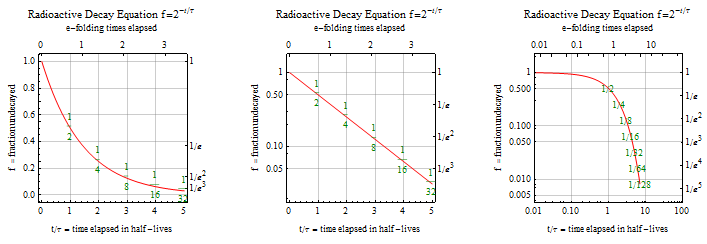
Three coordinate plots of exponential decay illustrating how halving occurs at equal time intervals (constant half-life behavior). The figure also highlights how using a semilog-style plot makes exponential decay appear linear, mirroring the “straight-line test” used for first-order kinetics. This helps connect the constant- concept to the graphical cue that vs. is linear. Source
After half-lives, (useful for reasoning, even when you are not doing full calculations).
What changes half-life
Since :
Increasing decreases (faster reaction, shorter half-life).
Decreasing increases (slower reaction, longer half-life).
Common AP Chemistry graph-reading cues
When analysing experimental outputs for a single reactant :
If vs. curves downward (exponential-looking), that can be consistent with first-order behaviour.
If vs. is a straight line, that is the syllabus-aligned diagnostic.
The line’s negative slope indicates depletion of reactant over time; a perfectly horizontal line would imply no reaction under those conditions.
FAQ
The derivation naturally produces the natural logarithm from calculus.
You can use $\log_{10}$ if you convert: $\log_{10}[A]t = -\dfrac{k}{2.303}t + \log{10}[A]_0$.
The mean lifetime $\tau$ is the average time a molecule survives before reacting.
For first-order processes, $\tau = 1/k$, and it relates to half-life by $t_{1/2} = (\ln 2)\tau$.
Taking a logarithm can magnify relative error when $[A]$ is very small.
Good practice is to avoid late-time points near detection limits and check whether residuals are randomly scattered about the fitted line.
Yes, if the gas behaves ideally and the rate law is written in terms of partial pressure.
Because $P_A \propto [A]$ at constant $T$ and volume, the same linear form applies with $\ln P_A$ versus $t$.
It comes from $\ln 2$, which appears when you set $[A]_t = \tfrac{1}{2}[A]_0$ in $\ln[A]_t = -kt + \ln[A]_0$.
It is a constant conversion between “halving” and natural-log decay.
Practice Questions
(2 marks) A student claims a reaction is first order in reactant . State the graph that should be linear to support this claim, and state how the rate constant is obtained from that graph.
versus time is linear. (1)
is the magnitude of the slope (slope ). (1)
(5 marks) A first-order reaction has . (a) Write the integrated rate law in a linear form that matches . (2 marks) (b) Write the half-life expression and state whether the half-life depends on . (3 marks)
. (2) (b)
. (1)
Half-life is constant for first order / depends only on . (1)
Does not depend on . (1)

