AP Syllabus focus: ‘The slopes of the linear plots for zero‑, first‑, and second‑order reactions can be used to determine the rate constant k.’
Reaction data are often messy when plotted as concentration versus time.
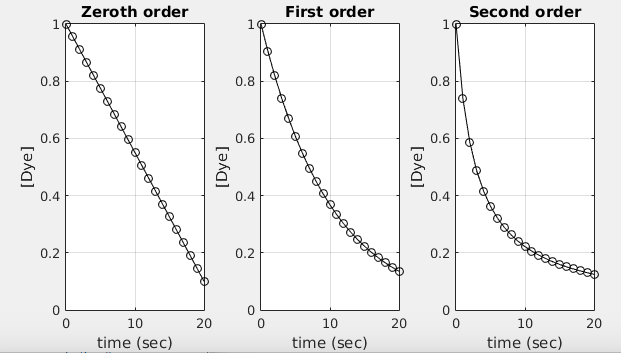
A comparison of how changes with time for different reaction orders on the untransformed vs. axes. The zero-order case appears as a straight, downward-sloping line (consistent with slope ), while first- and second-order behaviors appear curved on this plot. This helps motivate why linearization is used before extracting from a slope. Source
Linearizing the data using integrated rate laws creates straight-line graphs whose slopes directly reveal the rate constant, k, for the reaction.
What “linearized plots” mean for finding k
A linearized plot is a graphing choice that turns concentration–time data into an (approximately) straight line for a specific reaction order. When the plot is linear, the line’s slope is proportional to k, so you can determine k from the best-fit line without needing any point-by-point rate calculations.
Rate constant (k): The proportionality constant in a rate law that quantifies how fast a reaction proceeds at a given temperature; its units depend on overall reaction order.
In practice, you use linear regression (best-fit line) and read the slope.
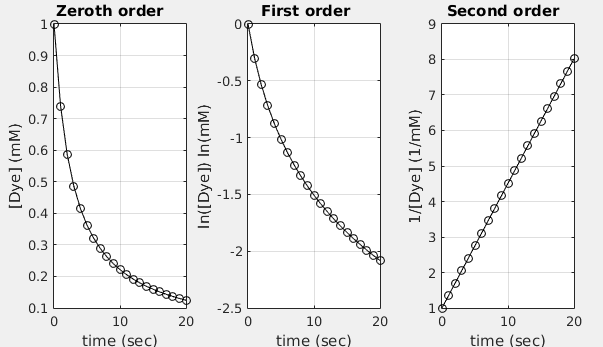
A first-order linearized plot of versus time, shown as a straight line. For first-order kinetics, the line’s slope is , so taking the negative of the best-fit slope yields the rate constant in units of time. This plot format is the standard graphical diagnostic for first-order behavior. Source
The critical skill is linking the slope from the correct linearized graph to k, including the correct sign.
Required linear plots and how slope gives k
For a single-reactant form (tracking [A] as it changes), AP Chemistry focuses on three integrated rate-law plots.
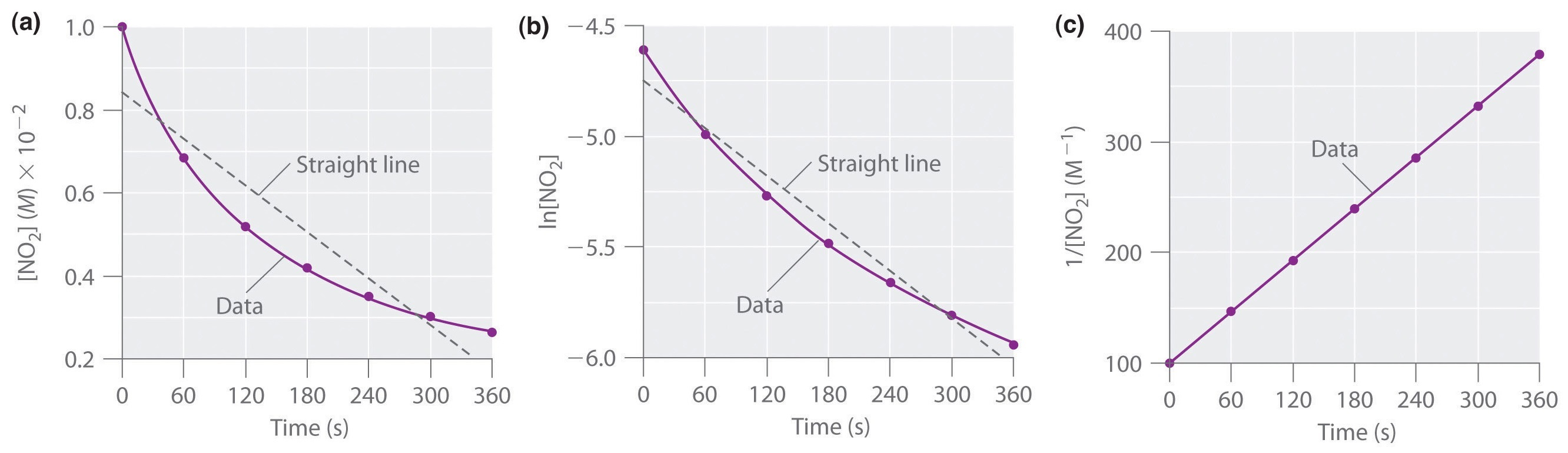
Side-by-side kinetic plots for the three standard linearizations: vs. , vs. , and vs. . For any given reaction order, exactly one of these becomes linear, and the slope of that straight line is proportional to the rate constant (either or ). Use this visual to connect “which plot is linear” to “which integrated rate law applies.” Source
Once a plot is linear, its slope corresponds to ±k as shown below.
= concentration of (M)
= time (s, min, etc.)
= rate constant (M·time)
= concentration of (M)
= time (s, min, etc.)
= rate constant (time)
= concentration of (M)
= time (s, min, etc.)
= rate constant (M·time)
The sign difference matters: for zero- and first-order linear plots, concentration (or its logarithm) decreases with time, so the slope is typically negative; for second order, increases with time, so the slope is positive.
Procedure: extracting k from a linearized graph
After you have concentration–time data for a reactant, determine k from the slope of the appropriate linearized plot.
Step-by-step workflow (graph-based)
Plot the data in the required linear forms:
Zero order: plot [A] vs t
First order: plot ln[A] vs t
Second order: plot 1/[A] vs t
Identify which graph is most linear by:
Visual straightness and
A regression measure (often ) closest to 1
Use the best-fit line slope, not a hand-drawn segment slope, to reduce random error.
Convert slope to k:
If slope , then
If slope , then
Report k with units consistent with the order implied by the linear plot:
Zero order: M·time
First order: time
Second order: M·time
Practical graphing details that affect k
Time units control k units. If time is in minutes, then k is per minute (or includes min in compound units).
Natural log is required. Use ln, not log base 10, unless explicitly instructed (AP typically uses ln).
Use reactant concentration. These linearized single-reactant plots apply to tracking a changing reactant concentration in the integrated forms given.
Treat k as positive. If experimental scatter makes a slope sign unexpected, re-check axes, transformations (ln, reciprocal), and whether the “linear” plot is truly appropriate.
Interpreting slope physically (why this works)
The integrated rate laws have the form “transformed concentration = (slope)(time) + (intercept).” The slope encodes the speed of concentration change under the model for that reaction order. Because the integrated equations place k directly in front of time, the slope is k (or −k), making k accessible from a straight-line fit to the data.
FAQ
It rescales the slope by a constant factor.
You’d need $k = -(\text{slope})\times 2.303$ when using $\log_{10}$ rather than $\ln$.
It corresponds to the transformed initial concentration (e.g., $[A]_0$, $\ln[A]_0$, or $1/[A]_0$).
A large mismatch from the known initial value can indicate timing delay or mixing error.
Early-time curvature can come from incomplete mixing, temperature drift, or instrument response lag.
Discarding obvious outliers (with justification) can improve the fitted slope.
Common issues include:
Inverting the wrong quantity (e.g., $1/t$ instead of $1/[A]$)
Swapping axes
Using product concentration rather than reactant concentration
Match the precision of the slope from regression and the raw data.
As a rule, report $k$ to 2–3 significant figures unless the dataset clearly supports more.
Practice Questions
(2 marks) A plot of versus time is linear with slope . Determine and state its units.
(1)
Units: (1)
(5 marks) Concentration–time data for reactant are tested using linearised plots. The versus graph is most linear, with best-fit slope .
(a) State the reaction order with respect to . (1)
(b) Determine and its units. (2)
(c) State the expected sign of the slope for the correct linearised plot and briefly justify. (2)
(a) Second order (1)
(b) (1); units (1)
(c) Positive slope (1); because increases as decreases with time (1)

